Welcome to Our Company -
-
Our Products
- Laboratory Chemicals
- 2 -Amino-6-bromo-4-methylbenzo[d]thiazole
- O-Bromoaniline Chemicals
- Solar Glycol
- Formic Acid
- 1-Chlorobutane For Liquid Chromatography
- 3-3-Dimethylbutyraldehyde Chemicals
- Ethyl--D-Glucuronide Chemicals
- Pure N-Heptane Chemical
- Nitric Acid
- Liquid Nitric Acid
- Tert Butyl Methyl Ether
- HACH Chemicals
- 1-Butanol Chemical
- HMB Calcium
- Zinc Sulphate Solution
- Acetonitrile Hypergrade For LC-MS
- Acetonitrile Sigma Aldrich
- Silver Iodide
- 1-Chlorobutane Chemicals
- Neutragel Neo Chemical
- Acetone Chemicals
- 3-Octanone Chemicals
- Ginkgolide C Chemicals
- Goldbio Chemicals
- 4-Aminophenol Acetaminophen RCK
- 1-Amino-3-(benzyloxy)cyclobutanecarboxylic acid hydrochloride
- 4-Aminopyrrolo[2,1-f][1,2,4]triazine-5-carboxylic acid
- 2-Amino-2- methylpropanenitrile
- Specialty Chemicals and Research Chemicals
- Hazardous and Non Hazardous Bulk Chemicals
- Aldehydes
- 3-Allylsalicylaldehyde 95%
- 1-Allyl-1h-pyrrole-2-carbaldehyde 95%
- 1-Allyl-1h-pyrazole-4-carbaldehyde 95%
- 3-Allyl-4-(2-propyn-1-yloxy)benzaldehyde 95%
- 4-(Allyloxy)-3-methoxybenzaldehyde 95%
- 4-(Allyloxy)-3-iodo-5-methoxybenzaldehyde 95%
- 4-(Allyloxy)-3-ethoxybenzaldehyde 95%
- 4-(Allyloxy)-3-chloro-5-methoxybenzaldehyde 95%
- 4-(Allyloxy)-2-chloro-5-methoxybenzaldehyd 95%
- 4-(Allyloxy)-3-chloro-5-ethoxybenzaldehyde 95%
- 2-(2-Hydroxyethoxy) Acetaldehyde 95%
- 4 - Acetamidobenzaldehyde 95%
- Gossypol-acetic Acid 95%
- Acetic acid 2-formyl-quinolin-8-yl Ester
- 3-Formylphenyl Acetate 98%
- 5-Bromo-4-formyl-2-methoxyphenyl Acetate 98%
- 4-Acetoxy-3,5-dimethoxybenzaldehyde 97%
- Acetylferulic Acid 95%
- 5-Acetoxymethyl-2-furaldehyde 98%
- 4-Acetoxy-4-methyl-1-pentanal 95%
- 2 - Acetylbenzaldehyde
- 3-Acetylbenzaldehyde 98%
- 4 Acetylbenzaldehyde 96%
- 4 Acetyl-biphenyl-4-carbaldehyde 95%
- N-Acetyl-D-Galactosamine 95%
- N-Acetyl-D-glucosamine 96%
- N-Acetylindole-3-carboxaldehyde 98%
- N-Acetyl-D-Mannosamine 97%
- 5-Acetyl-2-methoxybenzaldehyde 95%
- 4-Acetyl-1-methyl-1h-pyrrole-2-carbaldehyde 95%
- N-Acetyl-Muramic Acid 95%
- 1-(3-Acetylphenyl)-2,5-dimethyl-1h-pyrrole-3 Carbaldehyde
- 5-(4-Acetyl-phenyl)-furan-2-carbaldehyde 95%
- 1-(4-Acetylphenyl)-1h-pyrrole-2-carbaldehyde 95%
- 4-(4-Acetylpiperazin-1-yl)benzaldehyde 95%
- 1-Acetylpiperidine-4-carbaldehyde 95%
- 4-Acetyl-1h-pyrrole-2-carbaldehyde 98%
- 1-acetyl-1H-pyrrolo[2,3-b]pyridine-3 Carbaldehyde
- Acetylspiramycin 95%
- 5-Acetylthiophene-2-carbaldehyde 95%
- 5-Acetylthiophene-3-carbaldehyde 95%
- 3-Acetyl-2,4,6-trimethylbenzaldehyde 95%
- 3-(1-Adamantyl)-4-hydroxy-5-methoxybenzaldehyde 95%
- 3-(1-Adamantyl)-2-hydroxy-5-methylbenzaldehyde 95%
- 3-(1-Adamantyl)-1-phenyl-1h-pyrazole-4- carbaldehyde
- Adipaldehyde 1M in Water
- Alcaftadine 95%
- Ald-ch2-peg8-azide 95%
- Ald-ch2-peg10-t-butyl ester 95%
- Ald-ch2-peg4-ch2co2tbu 95%
- Aldehydo-D-glucose; keto-D-fructose 95%
- Ald-peg3-azide 95%
- Ald-peg1-t-butyl ester 96%
- Ald-peg4-t-butyl ester 95%
- Ald-peg5-t-butyl ester 95%
- Ald-peg5-t-butyl Ester
- Ald-peg4-nhs ester 95%
- Ald-peg4-pfp 95%
- Ald-ph-peg2-acid 95%
- Ald-Ph-PEG4-acid 95%
- Ald-ph-peg6-acid 95%
- Ald-ph-peg3-amine 95%
- Ald-ph-peg2-amine tfa salt 95%
- Ald-ph-peg2-t-butyl Ester
- Ald-ph-peg2-t-butyl Ester
- Ald-ph-peg4-t-butyl Ester
- Ald-ph-peg6-t-butyl Ester
- Ald-ph-peg2 nhboc
- Ald-ph-peg3-nh boc
- Ald-ph-peg3-o-nh boc
- Ald-ph-peg2 nhs
- All-trans Retinal
- 3-Allyl-4-(allyloxy)benzaldehyde 95%
- 3-Allyl-4-(allyloxy)-5 ethoxybenzaldehyde
- 3-Allyl-4-(allyloxy)-5 methoxybenzaldehyde
- 3-Allyl-4-butoxy-5 methoxybenzaldehyde
- -[(2E)-3-Allyl-4-chloro-5-formyl-1,3-thiazol-2(3h)-ylidene]thiophene-2-sulfonamide 95%
- 3-Allyl-4-(allyloxy)-5-methoxybenzaldehyde 95%
- 3-Allyl-4-butoxy-5-methoxybenzaldehyde 95%
- N-[(2E)-3-Allyl-4-chloro-5-formyl-1,3-thiazol-2(3h)-ylidene]thiophene-2-sulfonamide 95%
- 2-(Allyloxy)-5-chlorobenzenecarbaldehyde 95%
- 4-(Allyloxy)-2-chloro-5-ethoxybenzaldehyde 95%
- 3-Allyl-4,5-diethoxybenzaldehyde 95%
- 1-Allyl-2,3-dihydro-1h-indole-5 carbaldehyde
- 3-Allyl-4,5-dimethoxybenzaldehyde 95%
- 1-Allyl-3,5-dimethyl-1h-pyrazole-4 carbaldehyde
- 3-Allyl-4-ethoxybenzaldehyde 95%
- 3-Allyl-5-ethoxy-4-hydroxybenzaldehyde 95%
- 3-Allyl-4-ethoxy-5-methoxybenzaldehyde 95%
- 3-Allyl-5-ethoxy-4 methoxybenzaldehyde
- 3-Allyl-5-ethoxy-4 propoxybenzaldehyde
- 3-Allyl-5-ethoxy-4-(2-propyn-1-yloxy) benzaldehyde
- 2-Allyl-3-hydroxybenzaldehyde 95%
- 3-Allyl-4-hydroxybenzaldehyde 95%
- 2-Allyl-3-hydroxy-4-methoxybenzaldehyde 95%
- 3-Allyl-4-hydroxy-5-methoxybenzaldehyde 95%
- 5-Allyl-2-hydroxy-3-methoxybenzaldehyde 95%
- 1-Allyl-1h-indole-3-carbaldehyde 95%
- 3-Allyl-4-isopropoxybenzaldehyde 95%
- 3-Allyl-4-isopropoxy-5-methoxybenzaldehyde 95%
- 3-Allyl-4-methoxybenzaldehyde 95%
- 3-Allyl-5-methoxy-4-propoxybenzaldehyde 95%
- 1-Allyl-3-methyl-1h-pyrazole-4-carbaldehyde 95%
- 1-Allyl-5-methyl-1h-pyrazole-4 carbaldehyde
- 2-Allyloxybenzaldehyde 98%
- 3-(Allyloxy)benzaldehyde 95%
- 4-Allyloxybenzaldehyde 95%
- 2-(Allyloxy)-5-bromobenzaldehyde 95%
- 4-(Allyloxy)-3-bromobenzaldehyde 95%
- 4-(Allyloxy)-3-bromo-5-methoxybenzaldehyde 95%
- Fluorides
- 2-(Benzyloxy)-1-chloro-3 -fluorobenzene
- (R)-1-(Azetidin-3-yl) -3-fluoropiperidine
- 4-(benzyloxy)-2, 3-difluorophenol
- (1-Benzyl-4-fluoropiperidin-3-yl)methyl acetate
- 1-(Azetidin-3-yl)-4-fluoropiperidine dihydrochloride
- 2-Acetamido-4-fluorobenzoic acid
- 4- Acetylbenzotrifluoride
- 2-Amino-5-bromo-3-trifluoromethylpyridine 98%
- 2-Amino-6-fluorobenzoic acid 98%
- 2-(Benzyloxy)-3,5-difluorophenylboronic acid
- Benzyl 5-fluoropiperidin-3-ylcarbamate hydrochloride
- 5-Amino-2-methoxy-3-(trifluoromethyl) pyridine
- 1-Benzyl-3-bromo-5-(trifluoromethyl) pyridin-2(1H)-one
- ((2R,3R,5R)-3-(Benzoyloxy)-4,4-difluoro-5-((methylsulfonyl)oxy)tetrahydrofuran-2-yl)methyl benzoate
- 1-Acetamido-4-bromo-2- (trifluoromethoxy)benzene
- 1-(Benzyloxy)-2,4-dibromo-5- fluorobenzene
- 2-Amino-5-(4-fluorophenyl)isonicotinic acid 97%
- 2-(benzyloxy)-1,4-dichloro- 3-fluorobenzene
- 2-Benzoxazol-2-yl-1-(3-trifluoromethylphenyl) ethanol
- 3-Amino-4-fluorobenzoic acid 98%
- [3-Acetyl-5-(4-fluorophenyl)-2-methyl-1h-pyrrol-1-yl]acetic acid 95%
- 5-Amino-2-fluorobenzonitrile 97%
- Benzyl N-(4-chloro-2-fluorophenyl)carbamate
- 2-(benzyloxy)-1-chloro-4- fluorobenzene
- N-Benzyl-4-bromo-5-fluoro-2- nitroaniline
- 2-Amino-5-fluoropyridine 97%
- [2-Amino-1-(4-fluorophenyl)ethyl]dimethylamine 96%
- 4-Amino-2-fluoro-5-methoxybenzonitrile 98%
- 6-Amino-3-Bromo-2-trifluoromethylpyridine 98%
- 2-Amino-5-methoxybenzotrifluoride 98%
- N-Acetyl-3-(2,5-difluorophenyl) -d-alanine
- N-(2-Amino-3-fluorophenyl)methanesulfonamide 96%
- 1-(Benzyloxy)-4-bromo-2-(trifluoromethyl) benzene
- 4-Amino-5-bromo-2-(trifluoromethyl)benzonitrile 97%
- 2-Amino-5-fluorobenzonitrile 97%
- 2-Amino-4-fluorobenzamide 98%
- 3-(4-Benzyl-3-oxo-7-trifluoromethyl-3,4-dihydro-quinoxalin-2-yl)-propionic acid
- 1-Acetoxy -2-fluorobenzene
- 4-Acetoxy-2,3,5,6-tetrafluorobenzoic acid
- (2S,3R,4S,5S,6R)-2-(3-(Benzo[b]thiophen-2-ylmethyl)-4-fluorophenyl) -6-(hydroxymethyl)-2-methoxytetrahydro-2h-pyran-3,4,5-triol
- 3-Amino-4-fluorophenylboronic acid, pinacol ester 96%
- 6-Amino-2-fluoropyridine-3-boronic acid pinacol ester 95%
- 3-(1H-Benzoimidazol-2-yl) -1,1,1-trifluoro-propan-2-one
- 3-(benzyloxy)-1-chloro-2,4-difluoro- 5-methylbenzene
- 2-(Benzofuran-2-yl) -4-fluorobenzonitrile
- benzyl N-(5-chloro-2-fluorophenyl)carbamate
- 6-Benzyl-2,4-dichloro-8,8-difluoro-5,6,7,8-tetrahydropyrido[4,3-d]pyrimidine hydrochloride
- 1-(Benzyloxy)-3-bromo-5- fluorobenzene
- (2R,3R,4S,5R)-2-[(Benzoyloxy)methyl]-5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-4-fluorooxolan-3-yl benzoate
- Benzyl N-[3,5-bis(trifluoromethyl)phenyl]carbamate
- Benzenepropanoic acid, 2,3,6-trifluoro-
- Benzyl (2,4-difluorophenyl)carbamate
- 2-Amino-5-chloro-3-fluoropyridine 98%
- 2-(benzyloxy)-5-chloro-1,3- difluorobenzene
- 1-(Azetidin-3-yl)pyrrolidin-2-one trifluoro acetate
- Acetamide, n-[4-[2,2,2-trifluoro-1-hydroxy-1-(trifluoromethyl)ethyl]phenyl]-
- 5-(Benzyloxy)-2 -bromobenzotrifluoride
- 2-Acetamido -5-fluorotoluene
- 1-Benzyl-4,4- difluoropiperidine
- N-Benzyl-4- fluorobenzenesulfonamide
- Benzyl (2-(4-((4-fluorobenzyl)carbamoyl)-5-hydroxy-6-oxo-1,6-dihydropyrimidin-2-yl)propan-2-yl)carbamate
- 1-(benzyloxy)-5-chloro-2, 4-difluorobenzene
- Benzyl 2-(benzyloxy)-4-fluorobenzoate
- 2-Amino-5-fluoronicotinic acid 97%
- Benzyl N-[1-(2-fluorophenyl)cyclopropyl]carbamate
- 2-Amino-3-bromo-5-fluorobenzoic acid 96%
- 1-(4-Azepan-1-yl-3-fluorophenyl) ethanone
- 5-Amino-3-(2-fluorophenyl)-1-methylpyrazole ...96%
- 2-Benzyloxy-1-bromo-4- fluorobenzene
- 5-Amino-3-(2-fluorophenyl)-1-methylpyrazole .....96%
- 1-Benzoyl-3-(2,5-difluorophenyl) thiourea
- 3-(2-Amino-5-fluoropyridin-3-yl)prop-2-yn-1-ol 95%
- Benzoic acid, 3-(3-fluoropropoxy)-
- N-Benzyl-2-chloro-n-(4-fluorophenyl) acetamide
- [1-(Azetidin-3-yl)-4-piperidyl]methanol di trifluoro acetic acid salt
- Benzyl (3,3-difluoro-2-hydroxypropyl)carbamate
- 4-Amino-2-ethoxy-5-fluorobenzonitrile 96%
- 2-Aminomethyl-3-chlorophenylboronic acid, pinacol ester
- 4-(Benzyloxy)- 2-fluorobenzonitrile
- 3-Amino-4-mercaptobenzotrifluoride hydrochloride 95%
- Azetidin-3-yl(3-fluorophenyl) methanone
- Benzyl 3,3-difluorocyclobutylcarbamate
- 2-(Benzyloxy)-3-bromo-5- fluoroaniline
- 2-Amino-2-(4-fluoro-phenyl)-propionic acid 95%
- 2-Amino-3,5-difluoropyridine 97%
- 4-Acetamido-2-fluoro-5-methylbenzenesulfonyl chloride
- 4-(Benzyloxy)-3-bromo-5- fluoroaniline
- N-(4-Acetylphenyl)-2,2,3,3,4,4,4- heptafluorobutanamide 95%
- 2-Amino-4,6-difluoropyrimidine 96%
- 4-(benzyloxy)-1-chloro-2-(trifluoromethyl) benzene
- 4-Amino-2-fluoro-N-methylbenzamide 98%
- Benzyl[(5-fluoropyridin-2-yl) methyl]amine
- 4-Benzyloxy-2-fluoroaniline hydrochloride
- 5-Amino-2-fluorobenzoic acid 95%
- [3-(Benzyloxy)-2,4-difluoro-5-methylphenyl]boronic acid
- 2-Amino-4-fluorobenzoic acid 98%
- 5-Amino-8-fluoroquinoline 98%
- 1-Benzyl-5-bromo-6-fluoro-2-methyl-1, 3-benzodiazole
- 5-Amino-2-fluoro-3-picoline 98%
- 2-Amino-6-fluorobenzothiazole 98%
- 2-(benzyloxy)-1-chloro-4 -fluorobenzene
- 1-(1,3-Benzodioxol-5-yl) -4,4,4-trifluorobutane-1,3-dione
- 2-Acetamido -6-trifluoromethylpyridine
- 5-(Benzyloxy)-2- bromobenzotrifluoride
- 4-(Aminomethyl)-3-(trifluoromethyl) aniline
- 4-Amino-3-fluorophenol 97%
- 2-Amino-5-[4-fluoro-3-(trifluoromethyl)phenyl]pyridine 98%
- (3-Amino-2-fluorophenyl)methanol.... 97%
- 1-(Azetidin-3-yl)-4,4-difluoropiperidine ditrifluoroacetic acid salt
- 2-Aminomethyl-5-fluorophenylboronic acid hydrochloride
- benzyl N-(4-chloro-3-fluorophenyl)carbamate
- 2-Amino-5-(4-fluoro-3-methoxyphenyl)pyridine F308
- Benzoic acid, 3,4,5-trifluoro-, 1,1-dimethylethyl ester
- 1-(Azetidin-3-ylcarbonyl)-4-methylpiperazine bis(trifluoroacetate)
- 4-Amino-3-(trifluoromethyl)benzoic acid
- 5-Amino-3-[3-(trifluoromethyl)phenyl] isoxazole
- 2-Amino-4-bromo-5-fluorophenol 96%
- 2-Amino-3-(trifluoromethyl)benzoic acid
- 3-Amino-6-fluoropicoline.... 98%
- 4-Amino-2-(trifluoromethyl) benzonitrile
- 4-Amino-2-(trifluoromethyl) phenol
- 3-Amino-n-(2,2,2-trifluoroethyl) benzamide
- 2-Amino-5-(trifluoromethyl) benzamide
- 2-Amino-4-(trifluoromethyl)phenol hydrochloride
- 1-[(Ammoniooxy)methyl]-3-(trifluoromethyl)benzene chloride
- 3-(Benzyloxy)-6-bromo-2- fluorophenol
- 2-Amino-6-(trifluoromethyl) pyridine
- 3-Amino-1,1, 1-trifluoropropan-2-ol
- 2-Amino-3- trifluoromethylbenzonitrile
- 1-Benzoyl-3-(3-fluorophenyl) thiourea
- 3-Azetidineacetic acid trifluoroacetate
- 4-Acetyl-2,2-difluoro-1, 3-benzodioxole
- 1-Benzyloxy-2-chloro-4- fluorobenzene
- Benzoyl-1,1,1- trifluoroacetone
- 4-(4-Aminophenyl)-N-ethyl-2- fluorobenzamide
- 4-(Benzyloxy)-3-fluoroaniline hydrochloride
- 2-(Benzyloxy)- 5-fluorobenzonitrile
- N-Benzyl-2-fluoroaniline hydrochloride
- 4-Amino-5-(2-fluorophenyl)-4h-1,2,4-triazole-3-thiol 95%
- 2-Amino-5-(4-fluorobenzylthio)-1,3,4-thiadiazole 95%
- Benzonitrile,4-(methylsulfonyl) -3-(trifluoromethyl)-
- 2-Amino-4-fluorobenzaldehyde 96%
- 4-(Benzyloxy)- 2-fluoroaniline
- 5-Amino-3-(3-fluorophenyl)-1-methylpyrazole ....95%
- 3-Amino-7-fluoro-1-methylindazole 96%
- 1-(Azetidin-3-yl) -3-fluoroazetidine
- 3-Amino-5-bromo-2-fluoropyridine 98%
- L-Asparagine 7-amido-4-methylcoumarin trifluoroacetate
- 7-[(4aS,7aS)-Octahydro-1H-pyrrolo[3,4-b]pyridin-6-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride
- 5-Fluorocytosine 98%
- 5-Amino-2- trifluoromethylpyridine
- (3R)-3-Amino-1-[3-(trifluoromethyl) -5H,6H,7H,8H-[1,2,4]triazolo[4,3-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one
- 3-Amino-3-(3-fluorophenyl)propanoic acid 97%
- 1-Benzhydryl-3-(trifluoromethyl) azetidin-3-ol
- 2-Amino-4-fluoro-5-methoxybenzoic acid 98%
- 2-(Azetidine-1-carbonyl) -4-fluoroaniline
- 2-Amino-3- trifluoromethylpyridine
- 1-(benzyloxy)-2,3-dichloro- 4-fluorobenzene
- [(3S,4R)-1-Benzyl-4-(3-fluorophenyl)pyrrolidin-3-yl] methanol
- 1-[(Ammoniooxy)methyl]-4-fluorobenzene chloride
- 1,3-Benzodioxole, 4-chloro-2,2-difluoro-
- 2-Amino-3-fluorobutyric acid 95%
- N-(4-Acetylphenyl)-2,2,2-trifluoroacetamide 95%
- 4- Benzyloxybenzotrifluoride
- N-(4-(Benzyloxy)benzylidene) -4-fluoroaniline
- 4-Azidosulfonyl-benzoic acid pentafluorophenyl ester
- 4-Amino-2-fluoropyridine 98%
- N-(2-Benzoyl-4-chlorophenyl) -3,4-difluorobenzamide
- (R)-3-Amino-3-(3-trifluoromethylphenyl)propanoic acid
- 3-Amino-4-methyl-n-[3-(trifluoromethyl) phenyl]benzenesulfonamide
- 3-Amino-2-fluoropyridine 97%
- (S)-1-(Azetidin-3-yl)-3-fluoropyrrolidine dihydrochloride
- 4-(Benzyloxy)-3-chloro-5-fluorophenylboronic acid
- 5-Amino-2-fluorobenzamide 98%
- 2-Amino-3-fluorobenzoic acid 98%
- 2-Amino-3,4-difluorobenzoic acid 98%
- 5-Amino-2-fluoropyridine 97%
- 2-Amino-3-methoxy-4-fluorobenzoic acid 98%
- Benzoic acid, 4-[(1,2,2-trifluoroethenyl)oxy]-
- 4-Amino-2-fluorobenzoic acid 98%
- Benzoyl -1,1,1-trifluoroacetone
- 2-Amino-4-fluorobenzonitrile 98%
- 4-Amino-3-(trifluoromethoxy)benzoic acid
- 2-Amino-4,4,4-trifluoro-n-butyric acid hydrochloride
- 2-Amino-5- trifluoromethylbenzonitrile
- 5-Benzyloxy- 2-fluoroaniline
- 4-(1,3-Benzodioxol-5-yl)-6-(trifluoromethyl) pyrimidine-2-thiol
- Amyltriethyl ammonium
- 2H-1,4-Benzoxazine, 7-fluoro-3,4-dihydro-3-methyl-
- 2-Benzyloxy-4- bromobenzotrifluoride
- 2-Azaspiro[3.3]heptan-6-ol trifluoroacetate
- 2-Amino-4-trifluoromethylphenyl disulfide
- 1-[2-(Benzyloxy)ethoxy]-3-bromo- 5-fluorobenzene
- 1-Benzyl-3,3-difluoropiperidin -4-one
- 1-Benzoyl-3- (2,3-difluorophenyl)thiourea
- 7-[(4aS,7aS) -Octahydro-1H-pyrrolo[3,4-b]pyridin-6-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride
- 2-Amino-5-fluoropyrimidine 98%
- 2-Amino-4-trifluoropyrimidine-5-boronic acid pinacol ester
- 4-(Azetidin-3-ylcarbonyl)morpholine trifluoroacetate
- 4-Amino-2,6-difluoropyridine 97%
- Benzyl ((4-fluoroazepan-4-yl)methyl)carbamate hydrochloride
- 4-Acetamido -3-bromobenzotrifluoride
- Ammonium hexafluorozirconate
- (3Ar,4r,5r,6as) -4-((e)-3,3-difluoro-4-phenoxybut-1-en-1-yl)-5-hydroxyhexahydro-2h-cyclopenta[b]furan-2-one
- 4-Amino-2,5-difluorobenzonitrile 97%
- 4-(6-Aminopyridin-3-yl)-2-fluorobenzoic acid
- 2-Amino-5-trifluoromethyl -1,3,4-thiadiazole
- 2-Amino-3-chloro-5-trifluoromethylpyridine 98%
- 2-Amino-6-(trifluoromethoxy) phenol
- 3-Amino-3-(2-fluorophenyl)propan-1-ol hydrochloride 98%
- ((3Ar,5r,6s,6ar)-2,2-dimethyl-6-(((trifluoromethyl)sulfonyl)oxy)tetrahydrofuro[2,3-d][1,3]dioxol-5-yl)methyl 4-methylbenzoate
- 4-Aminophenylsulfur pentafluoride
- 2-(Benzylaminomethyl) -4-bromo-1-fluorobenzene
- 3-Amino-5-methyl benzotrifluoride
- Benzyl (5-fluoroazepan-4-yl)carbamate hydrochloride
- 3-Amino-5-trifluoromethylphenylboronic acid, pinacol ester
- 5-Amino-2-fluoro-4-hydroxybenzoic acid 95%
- 3-Amino-6-fluoro-1-methylindazole 98%
- 2-Amino-4-(trifluoromethyl)pyridine-5-boronic acid pinacol ester
- (2R)-2-Amino-3-[4-(trifluoromethoxy)phenyl]propanoic acid
- 3-Amino-4- (trifluoromethyl)pyridine
- [4-Amino-2-(trifluoromethyl)phenyl] (4-methyl-1-piperazinyl)methanone
- 1-(benzyloxy)-2,4-difluoro- 5-nitrobenzene
- 5-Amino-2-methyl benzotrifluoride
- 2-Acetamido-4,4,4-trifluorobutanoic acid
- 2-Amino-5-fluorobenzotrifluoride 98%
- 2-Amino-4,5,6- trifluorobenzothiazole
- 4-Amino-2-fluorobenzotrifluoride 97%
- (3As,4r,6ar) -6-((benzyloxy)methyl)-5-fluoro-2,2-dimethyl-4,6a-dihydro-3ah-cyclopenta[d][1,3]dioxol-4-ol
- [2-Amino-1-(2-fluorophenyl)ethyl]dimethylamine 95%
- 2-Amino-6-fluorobenzamide 98%
- (3S,11Ar)-n-(2,4-difluorobenzyl) -6-methoxy-3-methyl-5,7-dioxo-2,3,5,7,11,11a-hexahydrooxazolo[3,2-d]pyrido[1,2-a]pyrazine-8-carboxamide
- 2-Amino-4-(trifluoromethyl)thiazole-5-carboxylic acid
- 2-Amino-3-(trifluoromethoxy)benzoic acid
- (S)-3-Amino-3-(3-trifluoromethylphenyl)propionic acid
- (4-Amino-2-fluorophenyl)acetic acid 98%
- Ammonium trifluoroacetate
- 5-(benzyloxy)-1-chloro-2-fluoro-3- (trifluoromethyl)benzene
- 2-Amino-5- nitrobenzotrifluoride
- 3-Amino-n-(3-trifluoromethyl-phenyl) -benzenesulfonamide
- 2-(Benzyloxy)-1-bromo-5-fluoro -3-nitrobenzene
- 3-Amino-5-chlorobenzotrifluoride 98%
- 4-Amino-n-[4-(trifluoromethyl) phenyl]benzenesulfonamide
- 4-Amino-2,3,5,6- tetrafluoropyridine
- 1-(benzyloxy)-2-chloro-5-fluoro- 4-methylbenzene
- 3-Amino-5-(trifluoromethyl) benzonitrile
- 2-Benzyloxy- 4-fluoroaniline
- 1-(Azetidin-3-yl)-3,3-difluoropyrrolidine dihydrochloride
- 2-(N-Benzylamino) -5-chloro-3-fluoropyridine
- 5-Amino-4-cyano-1-(4-fluorophenyl)-3-methylpyrazole 97%
- 4-Acetamido-3-fluorobenzoic acid
- 5-Amino-2-pyrrolidin -1-yl-n-(3-trifluoromethyl-phenyl)-benzenesulfonamide
- 3-Amino-4-fluorophenol 97%
- 2-Amino-1-(4-(trifluoromethyl)phenyl)ethanone hydrochloride
- 5-Amino-3-(trifluoromethyl) pyridin-2-ol
- (3Ar,4r,5r,6as)-4- ((e)-3,3-difluoro-4-phenoxybut-1-en-1-yl)-5-hydroxyhexahydro-2h-cyclopenta[b]furan-2-one
- 3-Amino-3-(3-trifluoromethylphenyl)propanoic acid
- 2-Amino-4'-fluoroacetophenone hydrochloride 97%
- 5-Amino-2-fluorobenzyl alcohol 96%
- 2-Amino-5-(4-trifluoromethoxyphenyl) pyridine
- 2-Amino-4-[(trifluoromethyl)sulfonyl] phenylamine
- 2-Amino-4- trifluoromethyloxazole
- Ammonium perfluoro(2-methyl-3-oxahexanoate)
- (S)-3-Amino-4-(2-trifluoromethylphenyl)butanoic acid hydrochloride
- 2-Amino-5-(trifluoromethoxy) benzonitrile
- 2-Amino-4-(trifluoromethyl) pyrimidine
- 1-Benzoyl-3-(2,4-difluorophenyl) thiourea
- 2-Amino-6-(trifluoromethoxy) benzo[d]thiazole
- 2-[4-Amino-3-(trifluoromethyl) phenyl]acetonitrile
- (S)-3-Amino-3-(4-trifluoromethylphenyl)propionic acid
- 4-(3-Aminophenyl)-N-ethyl-2- fluorobenzamide
- 2-(Benzyloxy)- 5-fluorobenzamide
- 4-Amino-3-(trifluoromethoxy)phenylboronic acid, pinacol ester
- 4-Amino-3-(trifluoromethoxy) benzonitrile
- 2-Amino-6-(trifluoromethyl) benzonitrile
- 5-Amino-8- trifluoromethoxyquinoline
- 2-Amino-5-(trifluoromethyl)benzoic acid
- 4-Amino-2-(trifluoromethyl)benzoic acid
- 4-Amino-5-chloro-2-ethoxy-N-({4-[(4-fluorophenyl)methyl]morpholin-2-yl}methyl)benzamide 98%
- Benzyl (5,5-difluoropiperidin-3-yl)carbamate hydrochloride
- 1-Benzyl-5-bromo-6- fluorobenzimidazole
- 2-Amino-6-fluorobenzonitrile 98%
- 2-Aminomethy-4-(4-fluorobenzyl) morpholine
- 1-(Benzyloxy)- 4-fluorobenzene
- 3-Amino-2-(2-fluorophenoxy)pyridine 95%
- 2-Amino-3-fluorobenzonitrile 98%
- 2-Aminomethyl-4-fluorophenylboronic acid hydrochloride
- 3-Amino-2-fluorobenzonitrile 97%
- 3-Benzyloxy-4-chloro-2-fluorophenylboronic acid
- 3-[3-Acetyl-5-(4-fluorophenyl)-2-methyl-1h-pyrrol-1-yl]propanoic acid 95%
- 1-Benzyl-3,3-difluoro-4, 4-piperidinediol
- 3-Amino-4-(trifluoromethoxy) benzonitrile
- 3-Amino-5-(trifluoromethyl) pyridin-2(1H)-one
- 4-Amino-2- trifluoromethylpyridine
- (3R)-1-(3-Azetidinyl)-3-fluoro-pyrrolidine dihydrochloride
- N-Benzyl 2-bromo-5-trifluoromethoxybenzenesulfonamide
- Benzeneacetamide, 4-fluoro-n-[4-(5-oxazolyl)phenyl]-
- Benzyl N-[2-(3-fluoropyridin-4-yl)ethyl]carbamate
- 1-({[(Benzyloxy)carbonyl]amino}amino)- 2,2,2-trifluoroethanone
- 2-Amino-6-fluorobenzaldehyde 98%
- N-Benzyl-5-chloro-2- fluorobenzamide
- 4-(Benzyloxy)-1-chloro- 2-fluorobenzene
- 3-Amino-5-fluoropyridin-2-ol 96%
- 4-(Benzyloxy)-2,3-difluorophenylboronic acid
- 1-Benzyl-3,3-difluoropiperidin-4-one oxime
- 2-Amino-3-fluoropyridine 98%
- 4-(1H-Benzimidazol-2-yl)-1-[3-(trifluoromethyl)phenyl] pyrrolidin-2-one
- 3-(benzyloxy)-1-bromo-2,4-difluoro-5 -methylbenzene
- 3- Benzyloxybenzaldehyde
- 2-Amino-5-fluoro-3-iodopyridine 95%
- 2-Amino-5-(3,5-difluorophenyl)pyridine 98%
- 1-(benzyloxy)-3-chloro-5- fluorobenzene
- 1-(benzyloxy)-4-chloro-2 -fluorobenzene
- Aluminum trifluoromethanesulfonate 95%
- 3-Amino-n-(2-fluoro-phenyl)-benzenesulfonamide 95%
- 2-Amino-5-fluoro-6-methylpyridine 98%
- (R)-2-Acetamido-2-(2-fluorophenyl)propanoic acid
- 2-(4-Aminophenyl) -6-fluorobenzothiazole
- 4-Amino-3-fluorobenzoic acid 98%
- 5-Amino-2-fluoro-4-picoline 95%
- Azetidin-3-yl acetate 2,2,2-trifluoroacetate
- 1-(Azetidin-3-yl)-3-fluoropyrrolidine dihydrochloride
- 1-Benzyl-5-bromo-6- fluorobenzotriazole
- 2-Amino-3-fluorophenol hydrochloride....98%
- 4-Amino-3- nitrobenzotrifluoride
- 3-(Benzyloxycarbonylamino)-4-fluorophenylboronic acid, pinacol ester
- 2-Amino-2-(2-fluorophenyl)ethanol 95%
- 2-Amino-4-chlorobenzotrifluoride 97%
- 3-Amino-2-fluorobenzotrifluoride 98%
- Benzyl 1-(4-fluorophenyl)cyclopropylcarbamate
- 4-(Benzyloxy)-3,5-difluorobenzoic acid
- 3-Amino-3-(3-fluorophenyl)propanoic acid...97%
- 5-Amino-6-Chloro-2,2-difluorobenzodioxole 97%
- 6-Benzyl-8,8-difluoro-5,6,7,8-tetrahydropyrido[4,3-d] pyrimidine-2,4-diol
- Benzyl(ethyl)dimethylammonium bis(trifluoromethanesulfonyl)imide
- Benzyl ((4,4-difluoropiperidin-3-yl)methyl)carbamate hydrochloride
- Benzyl 3-fluoro-4-oxopiperidine-1-carboxylate
- Benzyl n-[(3s,4r)-3-fluoropiperidin-4-yl]carbamate hydrochloride
- 3-(Benzyloxy)-2,6-difluorophenylboronic acid
- 2H-1,4-Benzoxazin-3(4h)-one, 6,7-difluoro
- 5-Amino-2,2-difluorobenzodioxole 97%
- 4-Benzyloxy-3,5-difluorophenylboronic acid
- 4-(Allyloxy) -2-fluorobenzonitrile 95%
- 3-(Benzyloxy)-5-fluorobenzoic acid
- 1-Benzyloxy-4-(difluoromethyl) benzene
- Benzyl 3-fluoropropanoate
- N-(1H-1,2,3-Benzotriazol-1-ylmethyl) -4-fluoroaniline
- (2R,3R,4R-3-Benzoyloxy-4-fluoro-4-methyl-5-oxotetrahydrofuran-2-yl)methyl benzoate
- 3-Amino-4-fluorobenzotrifluoride 98%
- Benzyl N-[4-(4-cyano-3-fluorophenyl)phenyl]carbamate
- 5-Acetyl-2, 2-difluoro-1,3-benzodioxole
- 2-(benzyloxy)-1-chloro-3,5- difluorobenzene
- 2-Amino-4-(4-fluorophenyl)thiazole 98%
- 3-(benzyloxy)-1-chloro-2,4-difluoro-5- methylbenzene
- Acetic acid 3-fluoro-4-methyl-phenyl ester
- 2-(Benzyloxy)-1-bromo-3,5- difluorobenzene
- 2-(benzyloxy)-4-chloro-1- (trifluoromethyl)benzene
- 2-Amino-3-nitro-5- fluoropyridine
- 2-Amino-5-fluorothiazole hydrochloride 97%
- (1-Benzyl-4-fluoropiperidin-3-yl) methanol
- Benzyl chlorofluoroacetate
- 2-Amino-5-fluoro-4-methylphenylboronic acid, pinacol ester 97%
- 1-(Azetidin-3-yl)-3-fluoroazetidine oxalate
- 1-Benzoyl-3-[4-(trifluoromethyl) phenyl]thiourea
- Benzyl [2-(3,5-bis(trifluoromethyl)phenyl)-1-methyl-2-oxo-ethyl]carbamate
- : 3-(Benzyloxycarbonylamino)-4-fluorophenylboronic acid, pinacol ester
- 3-(Benzylcarbamoyl)-4-fluorophenylboronic acid
- (1-Benzyl-4,4-difluoropiperidin-3-yl) methanol
- 3-Amino-2-hydroxy-5-(trifluoromethyl)pyridine 98%
- 5-(4-Aminophenyl)-2-fluorobenzoic acid
- 6-Amino-3-(4-fluorophenyl)picolinic acid ....98%
- Benzyl ((3-fluoroazepan-3-yl)methyl)carbamate
- Benzoic acid pentafluorophenyl ester
- 1-(Azetidin-3-ylcarbonyl)azepane trifluoroacetate
- 3-Amino-5-fluorobenzotrifluoride 98%
- 3-Amino-3-(2-fluorophenyl)propanoic acid 98%
- 3-Amino-5-fluoro-1-methylindazole 98%
- 3-Amino-5-bromo-2-fluorobenzoic acid 96%
- Benzyl N-[1-(4-fluorophenyl)cyclobutyl]carbamate
- Benzyl N-[2-(4,4-difluoropiperidin-1-yl)-2-oxoethyl]carbamate
- 2-Amino-4-fluoropyridine 98%
- N-benzyl-3-chloro-5- fluorobenzamide
- 3-Amino-2-methyl benzotrifluoride
- 4-Amino-3-fluorobenzonitrile 98%
- 2-Amino-5-fluorobenzaldehyde 96%
- 1-(2-Aminophenyl) -2,2,2-trifluoroethan-1-one
- Benzyl 2-fluoro-6-nitrobenzoate
- 4-Acetamido-3-fluorophenylboronic acid, pinacol ester
- 2-Acetamido-4-(trifluoromethyl)phenylboronic acid, pinacol ester
- 1-Acetyl -2-bromo-4-fluorobenzene
- 6-Acetamido-2, 3-difluoroanisole
- 2-Amino-4-bromo-6-fluorobenzothiazole 95%
- N-Benzyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- 4-(Azetidin-3-yl)-2,2-dimethylmorpholine ditrifluoroacetate
- Benzyl N-(3-bromo-2-fluorophenyl)carbamate
- 1-(Azetidin-3-yl) -4,4-difluoropiperidine
- 1-(benzyloxy)-3,5-dichloro- 2-fluorobenzene
- Azido-peg4-pfp ester
- 1-(benzyloxy)-3-chloro-5- (trifluoromethoxy)benzene
- 1-(benzyloxy)-3, 5-difluorobenzene
- 3-Amino-4-(trifluoromethyl)phenylboronic acid
- 3-Amino-2-fluoro-6-methylpyridine 98%
- Azetidin-1-yl(azetidin-3-yl)methanone trifluoroacetate
- 4-Amino-2-fluorophenylboronic acid pinacol ester...97%
- 2-Acetamido-5-fluorophenylboronic acid, pinacol ester
- 2-Amino-4-fluorobenzo[d]thiazole 96%
- 4-Amino-2-fluoro-5-methylbenzonitrile 98%
- 4-amino-3-(trifluoromethyl) benzonitrile
- (+/-)-Alpha-methoxy-alpha-trifluoromethylphenylacetic acid 98%
- N6-Benzoyl-2'-fluoro-2'- deoxyadenosine
- 2-Amino-2'-fluoro-5-nitrobenzophenone 98%
- 1-Benzenesulfonyl-5-fluoro-1h-pyrrolo [2,3-b]pyridine
- 5-Amino-3-fluorobenzonitrile 97%
- 2'-Amino-5'-chloro-2,2,2-trifluoroacetophenone 98%
- N4-Benzoyl-2'-fluoro-2'- deoxycytidine
- 2-Amino-4'-fluorobenzophenone 97%
- Benzyl (3-fluoro-4-morpholinophenyl)carbamate
- 2-Acetoxy-2', 4'-difluoroacetophenone
- (4R,12As)-n- (2,4-difluorobenzyl)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-[1,3]oxazino[3,2-d]pyrido[1,2-a]pyrazine-9-carboxamide
- 3-Amino-4-fluorobenzoic acid hydrochloride 98%
- Benzyl N-(5-bromo-2,3-difluorophenyl)carbamate
- 3-(Benzylcarbamoyl)-5-fluorophenylboronic acid
- N-Benzyl-3- fluorobenzamide
- 3-Amino-2-(methylamino)-5-(trifluoromethyl) pyridine
- 1-Benzyl-3,3-difluoro-4- phenylpiperidin-4-ol
- 4-Amino-2-fluoro-5-methylbenzenesulfonamide F310
- 4-(benzyloxy)-2-chloro-1- fluorobenzene
- 2-Amino-4-(trifluoromethoxy) phenol
- 1-(4-Amino-3-fluorophenyl)ethanone 98%
- Benzyl [(1r)-2-(3,5-bis(trifluoromethyl)phenyl)-1-methyl-2-oxo-ethyl]carbamate
- 2-Amino-5-fluoro-4-picoline 95%
- 2-Acetamido -1-nitro-3,5,6-trifluorobenzene
- 2-Benzyloxy- 6-fluorobenzonitrile
- 2-Acetamido-5-fluorobenzoic acid
- 1-(benzyloxy)-3-chloro-2,4- difluorobenzene
- 6-Amino-2-bromo-3-fluorophenol 97%
- 2-Amino-5-fluorophenol 95%
- (4R,12As)-n-(2,4-difluorobenzyl) -7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-[1,3]oxazino[3,2-d]pyrido[1,2-a]pyrazine-9-carboxamide
- Benzyl N-(3,3-difluoro-1-methylcyclobutyl)carbamate
- Benzyl N-[2-(4-fluorophenyl)propan-2-yl]carbamate
- 2-Aminomethyl-4-fluorophenylboronic acid, pinacol ester
- 1-(benzyloxy)-2-chloro-3 -fluorobenzene
- Benzoyl trifluoromethanesulfonate
- 3-(Benzyloxy)-6-bromo-2- fluoroanisole
- (S)-2-(2-((S)-2-Acetamido-4-methylpentanamido)acetamido) -n-(4-methyl-2-oxo-2h-chromen-7-yl)-6-(2,2,2-trifluoroacetamido)hexanamide
- 1-(Azetidin-3-yl)-3-methylazetidin-3-ol di-trifluoroacetate
- 3-Benzyloxy-2-chloro-6-fluorophenylboronic acid
- 2-Amino-5-chlorobenzotrifluoride 98%
- 2-(Benzyloxy)-5-chloro-3- fluoropyridine
- 2-Aminohexafluoropropan-2-ol 95%
- 4-Amino-2-fluorobenzonitrile 98%
- 4-Amino-5-fluoro-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]-1,2-dihydropyrimidin-2-one 98%
- benzyl N-(3-chloro-2-fluorophenyl)carbamate
- 1-Acetamido-3, 5-difluorobenzene
- : {[(2R,3S,4S,5R)-5-(6-Amino-2-fluoro-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid 97%
- 2-Acetamido -3-bromo-5-fluoropyridine
- 2-Benzyloxy-1, 3-difluorobenzene
- 5-Aminomethyl-4-2luorophenylboronic acid, pinacol ester
- 4-(6-Aminopyridin-3-yl)-N-ethyl -2-fluorobenzamide
- 5-Amino-2-fluorophenylacetic acid 98%
- Benzyl N-(4-bromo-2-fluorophenyl)carbamate
- benzyl N-(3-chloro-5-fluorophenyl)carbamate
- N-Acetyl-3-(2,5-difluorophenyl) -l-alanine
- 3-Acetyl-4-fluorobenzoic acid
- 4-Acetyl-3-fluorobenzoic acid
- 4-Acetyl-3-fluoro-benzoic acid benzyl ester
- 4-Acetyl-3-fluoro-benzoic acid methyl ester
- 4-Acetyl-3-fluoro-benzoic acid tert-butyl ester
- 2-Acetyl-5- fluorobenzonitrile
- N-Acetyl 5-fluoro-2-methylaniline
- N-Acetyl 2-fluoro-4-(3-hydroxy-3-methylbut-1-ynyl)aniline
- 3-Acetyl-7-fluorochromen -2-one
- 4-Acetyl-n-(4-fluorobenzyl) benzenesulfonamide
- 4-Acetyl -n-(4-fluorobenzyl)benzenesulfonamide
- 2'-Acetyl-4-fluoro-3- methylbiphenyl
- 5-Acetyl-2-fluorophenylboronic acid, pinacol ester
- 2-Acetyl-3-fluoropyridine 98%
- 3-Acetyl-2-fluoropyridine 97%
- 2-(4-Acetylphenyl)-2,2-difluoroacetic acid 95%
- N-(3-Acetylphenyl)-2-fluorobenzamide 95%
- N-(3-Acetylphenyl)-3-fluorobenzamide 95%
- N-(3-Acetylphenyl)-4-fluorobenzamide 95%
- N-(4-Acetylphenyl)-2-fluorobenzamide 95%
- N-(4-Acetylphenyl)-3-fluorobenzamide 95%
- N-(3-Acetylphenyl)-2,2,3,3,4,4,4-heptafluorobutanamide 95%
- N-(3-Acetylphenyl)-2,2,2-trifluoroacetamide 98%
- N-(4-Acetylphenyl) -2,2,2-trifluoroacetamide 95%
- 1-(4-Acetylphenyl)-2,2,2-trifluoroethanone 95%
- 6-Acetyl-2,2,3,3-tetrafluorobenzo-1,4-dioxene 95%
- S-Acetylthioglycolic acid pentafluorophenyl ester 95%
- 4-Acetyl-2-trifluoromethyl-benzoic acid 95%
- 4-Acetyl-2-trifluoromethyl-benzoic acid methyl ester 95%
- 3-Acetyl-4'-(trifluoromethyl)biphenyl 95%
- N-(1-Adamantylmethyl)-4-fluoroaniline 95%
- 1-(1-Adamantyl)-4,4,4-trifluorobutane-1,3-dione 95%
- L-Alanine-7-amido-4-methylcoumarin trifluoroacetate salt 98%
- 1-Allyl-1h-benzimidazole-2-thiol 95%
- Allyl chlorodifluoroacetate 95%
- Allyl 2-chloro-1,1,2-trifluoroethyl ether 95%
- Allyl 1h,1h-heptafluorobutyl ether 95%
- Allyl 1,1,2,3,3,3-hexafluoropropyl ether 95%
- 1-Allyl-1-methylpyrrolidinium bis(trifluoromethanesulfonyl)imide 98%
- 1-(Allyloxymethyl)-2-fluorobenzene 98%
- 2-Allyloxy-1,1,2,2-tetrafluoroethanesulfonyl fluoride 95%
- Allyl 2,2,3,3,3-pentafluoropropyl ether 97%
- Allyl 1h,1h-perfluorooctyl ether 95%
- 4-Allyl-2,3,5,6-tetrafluorobenzoic acid 90%
- Allyl 1,1,2,2-tetrafluoroethyl ether 95%
- Allyl 2,2,3,3-tetrafluoropropyl ether 95%
- N-Allyl-2,2,2-trifluoroacetamide 95%
- 2-Allyl-4-(trifluoromethoxy)aniline 95%
- 1-Allyl-2-(trifluoromethyl)benzimidazole 98%
- N-Allyl-2-(3-(trifluoromethyl)-4,5,6,7-tetrahydroindazol-1-yl)acetamide 95%
- Alpha-chloro-4-fluorobenzaldoxime 96%
- Alpha-fluorophenylacetic acid 95%
- (Alpha-fluoro)phenylacetic acid methyl ester 95%
- (R)-(+)-Alpha-methoxy-alpha-trifluoromethylphenylacetic acid 97%
- (S)-(-)-Alpha-methoxy-alpha-(trifluoromethyl)phenylacetic acid 98%
- (+)-Alpha-methoxy-alpha-(trifluoromethyl)phenylacetic anhydride 95%
- Alpha-methyl-3-(trifluoromethyl)benzyl alcohol 97%
- Alpha-methyl-m-trifluoromethylbenzyl bromide 98%
- Alpha-(p-toluenesulfonyl)-4-fluorobenzylisonitrile 98%
- (R)-Alpha-(4-trifluoromethyl-benzyl)-proline hydrochloride 95%
- (S)-Alpha-(4-trifluoromethyl-benzyl)-proline hydrochloride 95%
- Alpha-(trifluoromethyl)-gamma-butyrolactone 95%
- Alpha-(trifluoromethyl)styrene 95%
- Aluminium trifluoroacetate 95%
- 4-Aminobenzotrifluoride 98%
- 4-Amino-3',5'-bis(trifluoromethyl)biphenyl 95%
- 5-Amino-2-(4-Boc-piperazino)benzotrifluoride 98%
- 3-Amino-2-bromobenzotrifluoride 97%
- 3-Amino-4-bromobenzotrifluoride 97%
- 3-Amino-2-bromo-6-chloropyridine 97%
- 5-Amino-6-bromo-2,2-difluorobenzodioxole 98%
- 2-Amino-4-bromo-3-fluorobenzoic acid 96%
- 2-Amino-5-bromo-4-fluorobenzoic acid 95%
- 3-Amino-5-bromo-4-fluorobenzoic acid 97%
- 2-Amino-5-bromo-4-fluorophenol 97%
- 2-Amino-6-bromo-4-fluorophenol 95%
- 2-Amino-3-bromo-5-fluoropyridine 98%
- 5-Amino-3-bromo-2-fluoropyridine 97%
- 6-Amino-3-bromo-2-fluoropyridine 98%
- 4-Amino-3-bromo-5-nitrobenzotrifluoride 98%
- 2-Amino-3-Bromo-6-trifluoromethylpyridine 98%
- 3-Amino-2-bromo-6-(trifluoromethyl)pyridine 98%
- 5-Amino-2-chloro-4-fluorobenzoic acid 98%
- 5-Amino-4-chloro-2-fluorobenzoic acid 95%
- 2-Amino-5-chloro-2'-fluorobenzophenone 98%
- 2-amino-5-chloro-4-fluorophenol 95%
- 4-Amino-2-chloro-5-fluoropyrimidine 97%
- 4-Amino-6-chloro-5-fluoropyrimidine 98%
- 2-Amino-5-chloro-3-nitrobenzotrifluoride 95%
- 2-Amino-5-chloro-3-nitrobenzotrifluoride 97%
- 4-Amino-3-chloro-5-nitrobenzotrifluoride 97%
- 3-Amino-2-chloro-6-(trifluoromethyl)pyridine 98%
- 2-Amino-4-chloro-6-(trifluoromethyl)pyrimidine 97%
- : 5-Amino-4-cyano-1-(2,5-difluorophenyl)-3-methylpyrazole 96%
- 5-Amino-4-cyano-1-(2-fluorophenyl)-3-methylpyrazole 97%
- 5-Amino-4-cyano-3-methyl-1-(2,3,5,6-tetrafluorophenyl)pyrazole 98%
- 5-Amino-4,6-dibromo-2,2-difluorobenzodioxole 96%
- 2-Amino-3,5-dibromo-4-fluorobenzoic acid 98%
- 2-Amino-3,5-dibromo-6-trifluoropyridine 95%
- 2-Amino-3,5-dichlorobenzotrifluoride 97%
- 5-Amino-1-(2,6-dichloro-4-trifluoromethylphenyl)-3-cycano pyrazole 98%
- 5-Amino-1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-4-(trifluoromethane)sulfinyl-1H-pyrazole-3-carbonitrile 98%
- 2-Amino-2',4'-difluoroacetophenone hydrochloride 97%
- 4-Amino-2,2-difluoro-1,3-benzodioxole 96%
- 2-Amino-3,5-difluorobenzoic acid 97%
- 2-Amino-4,5-difluorobenzoic acid 98%
- 3-Amino-2,6-difluorobenzoic acid 95%
- 4-Amino-2,5-difluorobenzoic acid 96%
- 5-Amino-2,4-difluorobenzoic acid 97%
- 2-Amino-3,4-difluorobenzonitrile 98%
- 3-Amino-2,6-difluorobenzonitrile 98%
- 4-Amino-2,6-difluorobenzonitrile 96%
- 2-Amino-4,6-difluorobenzothiazole 98%
- 4-Amino-2,5-difluorobenzotrifluoride 98%
- 4-Amino-1-[(2R,4R,5R)-3,3-difluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,2-dihydropyrimidin-2-one 98%
- 4-Amino-2,6-difluorophenol hydrochloride 96%
- (6-Amino-2,3-difluorophenyl)acetic acid 95%
- 5-Amino-2,3-difluorophenylboronic acid 98%
- 3-Amino-4,5-difluorophenylboronic acid, pinacol ester 96%
- 2-Amino-5-(2,3-difluorophenyl)pyridine 98%
- 4-Amino-2-fluoro-5-nitrobenzonitrile 95%
- 2-Amino-3-fluorophenol 97%
- 2-Amino-4-fluorophenol 97%
- N-(2-Amino-4-fluorophenyl)methanesulfonamide .....98%
- N-(2 Amino 4 fluorophenyl)methanesulfonamide
- N-(2-Amino-5-fluorophenyl)methanesulfonamide ....98%
- 2-Amino-3-(trifluoromethoxy) benzoic acid
- 2-Amino-6- trifluoromethylbenzothiazole
- 2-Amino-5-(trifluoromethyl) nicotinaldehyde
- 2-Amino-5- trifluoromethylpyridine
- Amino-3,3,3-trifluoropropanoic acid hydrochloride
- (4R,12As)-7-(benzyloxy) -n-(2,4-difluorobenzyl)-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-[1,3]oxazino[3,2-d]pyrido[1,2-a]pyrazine-9-carboxamide
- 4-[4-Azido-2-(trifluoromethyl)benzoyl] morpholine
- (2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidine 3',5'-dibenzoate
- 3-Benzyloxy-5- bromobenzotrifluoride
- 4-(Benzyloxy)-5-bromo-2-fluorophenylboronic acid
- 1-(Benzyloxy)-2-bromo-4-(trifluoromethyl) benzene
- 1-(benzyloxy)-4-chloro-2,3- difluorobenzene
- 2-(benzyloxy)-1,5-dichloro- 3-fluorobenzene
- 1-Benzyloxy- 3-fluorobenzene
- Carboxes
- 2-Azaspiro[3.3]heptane-6-carboxylic acid trifluoroacetate
- 2-Acetylsulfanyl-2-methylpropionic acid ethyl ester
- 4-Acetoxymethylbenzoic acid
- 3-Acetyl-4-hydroxy-6-methyl-2h- pyran-2-one
- Acetic acid 2-ethylphenyl ester
- Acetic acid 3-buten-1-yl ester
- 8-Aminoquinoline-4-carboxylic acid
- 8-Aminoquinoline-3-carboxylic acid
- 5-Amino-2-phenyl-2h-1,2,3-triazole-4-carboxylic acid
- Ammonium 2,4-dichlorophenoxyacetate
- 5-Aminopyrazolo[1,5-a]pyridine-3-carboxylic acid hydrochloride
- Ammonium sebacate
- Aceclofenac ethyl ester.
- Acepromazine maleate
- 2-Acetamidoacrylic acid
- 5-Acetamidoanthranilic acid
- 2-acetamidoisophthalic acid
- 4-Acetamidobenzoic acid
- 4-Acetamidobicyclo[2.2.2]octane-1-carboxylic acid
- 2'-Acetamidobiphenyl-3-carboxylic acid
- 3'-Acetamidobiphenyl-3-carboxylic acid
- 4'-Acetamidobiphenyl-3-carboxylic acid
- 4-Acetamidobutyric acid
- 2-Acetamido-5-chlorobenzoic acid
- 4-Acetamido-5-chloro-2-methoxybenzoic acid
- 1-Acetamidocyclopentane-1-carboxylic acid
- 2-Acetamido-2-deoxy-1-thio-beta-D-glucopyranose 3,4,6-triacetate
- (1S,2S,3R,4R)-3-((S)-1-Acetamido-2-ethylbutyl)-4-guanidino-2-hydroxycyclopentane-1-carboxylic acid trihydrate
- N-(2-Acetamido)iminodiacetic acidmonosodium salt
- 4-Acetamido-2-methylbenzoic acid
- 3-Acetamido-3-methylbutanoic acid
- S-Acetamidomethyl-3-mercaptopropionic acid
- 3-Acetamido-2-methylphenyl acetate
- (4-Acetamidophenoxy)acetic acid
- 2-(2-Acetamidophenoxy)acetic acid
- 4-Acetamidophenylacetic acid ethyl ester
- 4-(4-Acetamidophenyl)-4-oxobutanoic acid
- 5-Acetamidopyridine-2-carboxylic acid
- 4-Acetamido-1h-pyrrole-2-carboxylic acid
- 3-Acetamido-2,4,6-triiodo-5-(N-methylacetamido)benzoic acid
- Acetic acid acetoxy-(4-chlorosulfonylphenyl)methyl ester
- Acetic acid 4-chloro-3-methylphenyl ester
- Acetic acid, 2-[[4-(4-cyclopropyl-1-naphthalenyl)-4h-1,2,4-triazol-3-yl]thio]-, methyl ester
- Acetic acid-d
- Acetic acid 4-ethylphenyl ester
- Acetic acid n-heptadecyl ester
- Acetic acid 5-hexen-1-yl ester
- Acetic acid, 2-hydrazinyl-2-imino-, ethyl ester
- Acetic acid 7-methoxy-4-oxo-1,4-dihydro-quinazolin-6-yl ester
- ACETIC ACID 8-NONEN-1-YL ESTER
- tert-Butyl 3-carbamimidoylazetidine-1-carboxylate acetate
- Acetic acid trans-2-hepten-1-yl ester
- Acetic acid tridecyl ester
- Acetic acid 2-vinylphenyl ester
- 3-Oxo- N- phenylbutanamide
- Acetoacetic acid n-butyl ester
- Acetoacetic acid hexyl ester
- Acetoacetic acid isoamyl ester
- Acetoacetic acid n-octyl ester
- Acetoacetic acid n-propyl ester
- Acetoacetic acid sec-butyl ester
- Acetoacetyl-l-carnitine chloride
- Acetochloro - alpha- d- mannose
- diethyl 2-(2-oxopropyl)malonate
- Diethyl 2-(2-oxopropyl)succinate
- Acetoxime benzoate
- Acetoxyacetic acid
- Acetonyl acetate
- 2-Acetylphenyl acetate
- 3-acetylphenyl acetate
- Acetoxyacetyl chloride
- 3-Acetoxybenzoic acid
- 4-Acetoxybenzoic acid
- (2-cyanophenyl) acetate
- 6-Acetoxy-n-caproic acid methyl ester
- 3-chloro-2-oxopropyl acetate
- 2-Acetoxy-2-(4-chlorophenyl)acetic acid
- 3-(4-ACETOXYPHENYL)ACRYLIC ACID
- 4-Acetoxy-3-(cyclopropylmethoxy)benzoic acid
- 2-(p-Tolyloxy)ethyl acetate
- 7-Acetoxy-1- heptene
- (R)-2-Acetoxy-3-(hexadecyloxy)propyl (2-(trimethylammonio)ethyl) phosphate
- 2,3-Dihydro-1H-inden-1-yl acetate
- 1-Chloro-2-methyl-1-oxopropan-2-yl acetate
- 4-(Acetyloxy)-3-methoxybenzoic acid
- ((2R,3S)-3-Acetoxy-6-methoxy-3,6-dihydro-2h-pyran-2-yl)methyl acetate
- 3-acetyloxy-2-methylbenzoic acid
- 7-Acetoxy-4- methylcoumarin
- (2-methylnaphthalen-1-yl) Acetate
- (Z)-4-Acetoxy-3-(4-(methylsulfonyl) phenyl)-2-phenylbut-2-enoic acid
- (2S,3S,4S,5R,6R)-6-(Acetoxymethyl)-3-(((trifluoromethyl)sulfonyl)oxy)tetrahydro-2H-pyran-2,4,5-triyl triacetate
- 6-Acetoxy-2-naphthoic acid
- 8-Acetoxy-1- octene
- 3-Acetoxy-2-phenylpropanoic acid
- (+/-)-2-Acetoxypropionic acid
- 2-Acetoxypropionyl chloride
- Succinimidyl acetate
- Acetrizoic acid
- Acetylacrylic acid methyl ester
- 5'-O- Acetyladenosine
- 3-[(Acetylamino)methyl]adamantane-1-carboxylic acid
- 4-(3-Acetylaminophenyl)benzoic acid
- 1-[3-(Acetylamino)phenyl]-5-oxopyrrolidine-3-carboxylic acid
- 1-[4-(Acetylamino)phenyl]-5-oxopyrrolidine-3-carboxylic acid
- 3-(Acetylamino)thiophene-2-carboxylic acid
- 4-(4-Acetylanilino)-4-oxobutanoic acid
- 1-Acetylazepane-4-carboxylic acid
- 1-Acetyl-azetidine-3-carboxylic acid
- 1-Acetyl-azetidine-3-carboxylic acid methyl ester
- 1-Acetylazetidin-3-yl acetate
- 2-Acetylbenzenecarbo nitrile
- 3-Acetylbenzo[f] coumarin
- 2-Acetylbenzoic acid
- 3-Acetylbenzoic acid
- 4-Acetylbenzoic acid
- 3- Acetylbenzonitrile
- 4- Acetylbenzonitrile
- 5-Acetyl-2-benzyloxy-benzoic acid
- 3'-Acetylbiphenyl-3-carboxylic acid
- 4'-Acetylbiphenyl-3-carboxylic acid
- 1'-Acetyl-[1,4'-bipiperidine]-4-carboxylic acid
- 4-Acetylbutyric acid
- 2- Acetylbutyrolactone
- Acetyl-l-carnitine hydrochloride
- N'-Acetyl-2- chloroacetohydrazide
- 4-Acetyl-2-chlorobenzoic acid
- 4-Acetyl-3-chlorobenzoic acid
- 3-Acetyl-3-chloro-dihydro-furan- 2-one
- Acetylcholine bromide
- Acetylcholine perchlorate
- 3- Acetylcoumarin
- 1-Acetyl-cyclopropanecarboxylic acid methyl ester
- 3-O-Acetyl-1,2:5,6-di-o-isopropylidene-alpha-d- glucofuranose
- 3-Acetyl-2,4-dimethyl-5- carbethoxypyrrole
- (1S,3R)-3-Acetyl-2,2-dimethylcyclobutane-1-carboxylic acid
- 5-Acetyl-2,2-dimethyl-1,3-dioxane-4,6- dione
- 4-Acetyl-3,5-dimethyl-1h-pyrrole-2-carboxylic acid
- 5-Acetyl-2,4-dimethyl-1h-pyrrole-3-carboxylic acid
- 4-O-Acetyl-3,6-di-o-(tert-butyldiphenylsilyl)-d- galactal
- 5-Acetyl-2-ethoxybenzoic acid
- 5-Acetyl-2-ethoxy-benzoic acid methyl ester
- 4-(2-Acetylhydrazino)-4-oxobutanoic acid
- 3-Acetyl-4-hydroxy-2H-chromen- 2-one
- 6-O-Acetyl-4-hydroxy-7-methoxy quinazoline
- 3-Acetylindolizine-1-carboxylic acid
- 3-Acetyl-4-isopropoxybenzoic acid
- 5'-Acetyl-2',3'- isopropylideneadenosine
- O-Acetyl mandelic acid
- 5-Acetyl-2-(4-methoxy-benzyloxy)-benzoic acid methyl ester
- 3-Acetyl-7-methoxychromen-2- one
- 4-Acetyl-2-methoxyphenyl acetate
- 5-Acetyl-2-methoxyphenyl acetate
- (5-Acetyl-2-methoxyphenyl)acetic acid
- 3-[3-Acetyl-5-(4-methoxyphenyl)-2-methyl-1h-pyrrol-1-yl]propanoic acid
- N- Acetyl-2-methylalanine
- 2-[Acetyl(methyl)amino]benzoic acid
- 1-Acetyl-2-methylazepane-2-carboxylic acid
- 1-Acetyl-4-methylazepane-4-carboxylic acid
- 4-Acetyl-2-methylbenzoic acid
- 4-Acetyl-2-methyl-benzoic acid benzyl ester
- 3-Acetyl-7-methyl-2h-chromen-2- one
- 3-Acetyl-6-methyl-2-oxo-2h-pyran-4-yl difluoridoborate
- (3-Acetyl-2-methyl-5-phenyl-1h-pyrrol-1-yl)acetic acid
- 3-(3-Acetyl-2-methyl-5-phenyl-1h-pyrrol-1-yl)propanoic acid
- 6-Acetyl-7-methylpyrazolo[1,5-a]pyrimidine-3-carboxylic acid
- 4-Acetyl-1-methyl-1h-pyrrole-2-carboxylic acid
- 1-Acetyloxy-2,5-dioxopyrrolidine-3-sulfonic acid
- (2-Acetyl-phenoxy)-acetic acid
- (3-Acetylphenoxy)acetic acid
- 4-Acetylphenoxyacetic acid
- (3-Acetyl-phenoxy)-acetic acid tert-butyl ester
- (4-Acetyl-phenoxy)-acetic acid tert-butyl ester
- 4-(4-Acetyl-phenoxy)-butyric acid
- 2-(4-Acetylphenoxy)-n,n- diethylacetamide
- 2-(4-Acetylphenoxy)-n,n- dimethylacetamide
- 5-[(2-Acetylphenoxy)methyl]-2-furoic acid
- 5-[(4-Acetylphenoxy)methyl]-2-furoic acid
- 2-(4-Acetylphenoxy)-n- phenylacetamide
- 4-Acetylphenyl acetate
- 2-([(3-Acetylphenyl)amino]carbonyl)cyclohexanecarboxylic acid
- 2-([(4 Acetylphenyl)amino]carbonyl) cyclohexanecarboxylic acid
- 6-([(3-Acetylphenyl)amino]carbonyl)cyclohex-3-ene-1-carboxylic acid
- 6-([(4-Acetylphenyl)amino]carbonyl)cyclohex-3-ene-1-carboxylic acid
- 2-(3-Acetylphenyl)benzoic acid
- 3-(2-Acetylphenyl)benzoic acid
- 4-(2-Acetylphenyl)benzoic acid
- 4-(3-Acetylphenyl)benzoic acid
- 4-(4-Acetylphenyl)benzoic acid
- 3-(3-Acetylphenyl) benzonitrile
- 3-(4-Acetylphenyl) benzonitrile
- 4-(2-Acetylphenyl)-2-chlorobenzoic acid
- 4-(4-Acetylphenyl)-2-chlorobenzoic acid
- 4-Acetylphenyl (3,4-dichlorophenyl) carbamate
- 4-Acetylphenyl dimethylcarbamate
- 2-(4-Acetylphenyl)-1,3-dioxoisoindoline-5-carboxylic acid
- 3-Acetylphenyl ethyl(methyl) carbamate
- 3-(3-Acetylphenyl)-4-fluorobenzoic acid
- 3-(4-Acetylphenyl)-4-fluorobenzoic acid
- 5-(4-Acetylphenyl)-2-furoic acid
- (4-Acetylphenyl) n-methylcarbamate
- 1-Acetyl-4-piperidineacetic acid
- 1-Acetylpiperidine-4-carbonyl chloride
- (S)-1-Acetylpiperidine-3-carboxylic acid
- 1-Acetylpiperidine-3-carboxylic acid
- 1-Acetylpiperidine-4-carboxylic acid
- 6-[(1-Acetylpiperidin-4-yl)amino]pyrimidine-4-carboxylic acid lithium salt
- 5-Acetylpyrazine-2-carboxylic acid
- 3-Acetyl-1h-pyrazole-5-carboxylic acid
- 5-Acetyl-2H-pyrazole-3-carboxylic acid ethyl ester
- 6-Acetyl-2-pyridinecarboxylic acid
- 4-Acetyl-1h-pyrrole-2-carboxylic acid
- 1-Acetyl-2- pyrrolidone
- 6-Acetylquinoline-3-carboxylic acid
- 7-Acetylquinoline-3-carboxylic acid
- Acetylsalicylic anhydride
- Acetylsalicylsalicylic acid
- 4-(Acetylsulfanyl)butanoic acid
- S-Acetylthioacetic acid
- 2-[(Acetylthio)methyl]-3-phenylpropionic acid
- (2S)-3-Acetylthio-2-methylpropionic acid
- 2-(Acetylthio)succinic anhydride
- 3'-O- Acetylthymidine
- 9-Acridinecarboxylic acid
- 2- Propenamide
- 6-Acrylamidohexanoic acid
- Acrylic acid barium salt monomer
- Acrylic acid isoamyl ester
- Acrylic acid tetradecyl ester
- Acrylic anhydride
- 4-(6-Acryloxy-hex-1-yl-oxy)phenyl 4-(hexyloxy) benzoate
- (3-Acryloxypropyl)tris (trimethylsiloxy)silane
- N- Acryloxysuccinimide
- 4- Acryloyloxybenzophenone
- 4-((6-(Acryloyloxy)hexyl)oxy)benzoic acid
- 4'-[(6-Acryloyloxy)hexyloxy]-4- biphenylcarbonitrile
- 1-Adamantaneacetic acid
- 1- Adamantanecarboxamide
- Adamantane-2-carboxylic acid
- 1-Adamantanecarboxylic acid
- Adamantane-1-carboxylic acid methyl ester
- 1,3-Adamantanediacetic acid
- 1,3-Adamantanedicarboxylic acid
- 2-Adamantanone-5-carboxylic acid
- (E)-3-((1r,3r,5R,7S)-Adamantan-2-ylidene)-4- (1-(2,5-dimethylfuran-3-yl)ethylidene)dihydrofuran-2,5-dione
- Adamantan-1-ylmethyl acetate
- [2-(1-Adamantyl)ethoxy]acetic acid
- 5-(1-Adamantyl)-2-furoic acid
- 1-Adamantyl methacrylate
- 3-(1-Adamantyl)-4-methoxybenzoic acid
- 5-(1-Adamantyl)-2-methoxybenzoic acid
- 1-(2-Adamantyl)-4-nitro-1h-pyrazole-3-carboxylic acid
- (1-Adamantyloxy)acetic acid
- [4-(1-Adamantyl)phenoxy]acetic acid
- 4-(1-Adamantyl)phenyl acetate
- 3-(1-Adamantyl)-1-phenyl-1h-pyrazole-4-carboxylic acid
- (2E)-3-[3-(1-Adamantyl)-1-phenyl-1h-pyrazol-4-yl]acrylic acid
- 3-(1-Adamantyl)propanoic acid
- 2-(1-Adamantyl)quinoline-4-carboxylic acid
- (1-Adamantylthio)acetic acid
- Adipic acid dibenzyl ester
- Adipic acid monobenzyl ester
- Adipic acid monoethyl ester
- Adipic anhydride
- Adipic dihydrazide
- Z-Aeeac-oh dcha
- Afatinib dimaleate
- Ahu-377 hemicalcium salt
- Alantola ctone
- Allyl acetoacetate
- Allyl amyl glycolate
- Allyl benzoate
- 3-Allyl-3-hydroxyazetidine-1-carboxylic acid tert-butyl ester
- 3-Allyl-4-methoxybenzoic acid
- Allyl 2-oxocyclohexanecarboxylate
- Allyl 2-oxocyclopentanecarboxylate
- 1-Allyl-5-oxopyrrolidine-3-carboxylic acid
- 3-Allyloxy-azetidine-1-carboxylic acid tert-butyl ester
- 4-(Allyloxy)-1-benzofuran-6-carboxylic acid
- 3-(Allyloxy)benzoic acid
- 4-(Allyloxy)benzoic acid
- 4-(Allyloxy) benzonitrile
- 4-Allyloxycarbonylamino-bicyclo[2.2.2]octane-1-carboxylic acid
- 1-Allyloxycarbonylamino-cyclopentanecarboxylic acid
- 1-Allyloxycarbonylamino-cyclopropanecarboxylic acid
- (2-Allyloxy-ethoxy)-acetic acid benzyl ester
- 3-Allyloxyphenylacetic acid
- 3-Allyloxypropionic acid
- 2-(4-Allyloxy-pyrazol-1-yl)-2-methyl-propionic acid
- [(4-Allyl-5-phenyl-4h-1,2,4-triazol-3-yl)thio]acetic acid
- 1-Allylpiperidine-3-carboxylic acid
- 1-Allylpiperidine-4-carboxylic acid
- Allylsuccinic anhydride
- 2-Allyl-1-(tert-butoxycarbonyl)-2,3-dihydro-1H-benzo[d]imidazole-2-carboxylic acid
- 1-Allyl 4-tert-butyl piperazine-1,4-dicarboxylate
- (Allylthio)acetic acid
- 2-(Allylthio)propanoic acid
- Aloc-o2oc-oh dcha
- Alpha- acetoxyphenylacetonitrile
- (1Alpha,8alpha,9alpha)-bicyclo[6.1.0]non-4-ene-9-carboxylic acid ethyl ester
- (1Alpha,2alpha,4alpha)-1,2,4-cyclohexanetricarboxylic acid
- Alpha,alpha'- dilaurin
- Alpha,alpha-dimethyl-gamma- butyrolactone
- Alpha- angelicalactone
- Alpha-d-cellobiose octaacetate
- Alpha-cyanocinnamic acid
- Alpha-cyanocinnamic acid ethyl ester
- alpha-Cyclopentylmandelic acid
- Alpha-cyclopentylphenylacetic acid
- Alpha-(Fmoc-amino)-omega-(succinimidyl propionate) tetra(ethylene glycol)
- Alpha-d-galacturonic acid hydrate
- 2-O-Alpha-d-glucopyranosyl-l-ascorbic acid
- Alpha-hydroxy-gamma- butyrolactone
- Alpha-ketobutyric acid sodium salt
- Alpha-ketoglutaric acid sodium salt
- H-Alpha-me-leu- oh
- (R)-2-AMINO-2,4-DIMETHYLPENTANOIC ACID
- Alpha-methoxyphenylacetic acid sodium salt
- Alpha-methylcinnamic acid
- Alpha-methylene-gamma- butyrolactone
- Alpha-methylene-gamma- valerolactone
- Alpha-methyl-gamma- butyrolactone
- Alpha-phenylcinnamic acid
- Alpha-(phenylthio)phenylacetic acid
- L-Alpha- phosphatidylinositol
- 2H-Pyran- 2-one
- D-Alpha-tocopheryl acetate
- 4-Amidinobenzoic acid hydrochloride
- 2-Aminoadamantane-2-carboxylic acid
- 3-Amino-1-adamantanecarboxylic acid
- 3-Amino-1-adamantanecarboxylic acid hydrochloride
- 6-Amino-3-aza-bicyclo[3.1.1]heptane-3-carboxylic acid
- 6-Amino-2-azaspiro[3.3]heptane-6-carboxylic acid
- 6-Amino-2-azaspiro[3.3]heptane-6-carboxylic acid dihydrochloride
- 6-Amino-2-aza-spiro[3.3]heptane-2-carboxylic acid tert-butyl ester
- 6-Amino-2-aza-spiro[3.3]heptane-2,6-dicarboxylic acid 2-tert-butyl ester
- 2-Amino-7-azaspiro[3.5]nonane-7-carboxylic acid tert-butyl ester
- 2-Amino-6-aza-spiro[3.4]octane-6-carboxylic acid tert-butyl ester
- 3-Amino-azepane-1-carboxylic acid tert-butyl ester
- 3-Aminoazetidine-3-carboxylic acid hydrochloride
- 4-Aminobenzene-1,3-dicarboxylic acid
- 4-Amino-1-benzofuran-2-carboxylic acid
- 5-Amino-1-benzofuran-2-carboxylic acid
- 5-Amino-1h-benzoimidazole-2-carboxylic acid methyl ester
- 2-Aminobenzophenone-2'-carboxylic acid
- 5-Amino-1-benzothiophene-2-carboxylic acid
- 3-(4-Aminobenzylamino)pyrrolidine-1-carboxylic acid tert-butyl ester hydrochloride
- 3-Amino-5-(4-benzyloxyphenyl)thiophene-2-carboxylic acid methyl ester
- 3-Amino-1-benzylpyrrolidine-3-carboxylic acid
- 5-Amino-1-benzyl-1,2,3,6-tetrahydro-pyridine-4-carboxylic acid ethyl ester
- 5-Amino-1-benzyl-1h-1,2,3-triazole-4-carboxylic acid
- 3-Aminobicyclo[2.2.1]hept-5-ene-2-carboxylic acid hydrochloride
- 4-Aminobicyclo[2.2.2]octane-1-carboxylic acid
- 4-Amino-bicyclo[2.2.2]octane-1-carboxylic acid cyclopropylamide, hydrochloride
- 4-Amino-bicyclo[2.2.2]octane-1-carboxylic acid cyclopropylmethyl-amide, hydrochloride
- 4-Amino-bicyclo[2.2.2]octane-1-carboxylic acid methylamide, hydrochloride
- 3-Aminobicyclo[1.1.1]pentane-1-carboxylic acid hydrochloride
- 1'-Amino-1,1'-bi(cyclopropyl)-1-carboxylic acid hydrochloride
- 2'-Aminobiphenyl-3-carboxylic acid
- 3'-Aminobiphenyl-3-carboxylic acid
- 4'-Aminobiphenyl-3-carboxylic acid
- 2'-Amino-[1,1'-biphenyl]-4-carboxylic acid hydrochloride
- 3'-Amino-[1,1'-biphenyl]-3-carboxylic acid hydrochloride
- 4'-Amino-[1,1'-biphenyl]-4-carboxylic acid hydrochloride
- 3'-Amino-biphenyl-3-carboxylic acid methyl ester hydrochloride
- (1S,3R,4R)-4-Amino-3-(boc-amino)-cyclohexanecarboxylic acid ethyl ester
- 4-Amino-1-Boc-4-piperidinedicarboxylic acid ethyl ester
- (3R,4S)-4-Amino-1-boc-3-pyrrolidinecarboxylic acid ethyl ester hydrochloride
- 2-Amino-4- bromobenzamide
- 3-Amino-6-bromo-benzofuran-2-carboxylic acid methyl ester
- 3-Amino-5-bromobenzoic acid
- 4-Amino-5-bromo-2-methoxybenzenecarboxylic acid
- 4-Amino-8-bromo-6-methylquinoline-3-carboxylic acid ethyl ester
- 5-Amino-1-(4-bromophenyl)-1H-pyrazole-4-carboxylic acid
- 2-Amino-3-bromopyrazine-5-carboxylic acid
- 3-Amino-4-bromo-1h-pyrazole-5-carboxylic acid
- 4-Amino-5-bromopyridine-2-carboxylic acid
- 3-Amino-6-bromopyridine-2-carboxylic acid ethyl ester
- 3-Amino-5-bromopyridine-2-carboxylic acid hydrochloride
- 3-Amino-5-bromo-pyridine-2-carboxylic acid isopropyl ester
- 4-Amino-6-bromoquinoline-3-carboxylic acid
- 4-Amino-8-bromoquinoline-3-carboxylic acid ethyl ester
- 3-Amino-5-bromo-thiophene-2-carboxylic acid methyl ester hydrochloride
- 2-Amino-3-carbamoyl-4,7-dihydro-5h-thieno[2,3-c]pyridine-6-carboxylic acid benzyl ester
- 2-Amino-3-carbamoyl-4,7-dihydro-5h-thieno[2,3-c]pyridine-6-carboxylic acid tert-butyl ester
- N-[3-(Aminocarbonothioyl) phenyl]cyclopropanecarboxamide
- 4-[(Aminocarbonyl)amino]benzoic acid
- (1S,2R,3R,4S)-3-(Aminocarbonyl)bicyclo[2.2.1]hept-5-ene-2-carboxylic acid
- 3-Aminocarbonyl-1-Boc- pyrrolidine
- 2-(Aminocarbonyl)cyclopropanecarboxylic acid
- 2-[4-(Aminocarbonyl)phenoxy]quinoline-4-carboxylic acid
- 3-(3-Aminocarbonylphenyl)benzoic acid
- 4-(3-Aminocarbonylphenyl)benzoic acid
- 1-[4-(Aminocarbonyl)phenyl]-5-oxopyrrolidine-3-carboxylic acid
- 5-(Aminocarbonyl)thiophene-2-carboxylic acid
- 4-Amino-4-carboxytetrahydrothiopyran hydrochloride
- 3-Amino-4-chloro-benzofuran-2-carboxylic acid methyl ester
- 4-Amino-2- chlorobenzonitrile
- 4-Amino-3-([(2-chlorobenzyl) amino]carbonyl ) isothiazole-5-carboxylic acid
- 6-Amino-5-chloro-2-cyclopropylpyrimidine-4-carboxylic acid
- 4-Amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid
- 5-Amino-1-(2-chloro-5-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-1-(4-chloro-2-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- 6-Amino-5-(4-chloro-3-methylphenyl)pyridine-3-carboxylic acid
- 1-[2-Amino-1-(2-chloro-phenyl)-ethyl]-pyrrolidine-3-carboxylic acid
- 5-Amino-1-(4-chlorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-4-chloro-1h-pyrazole-3-carboxylic acid
- 2-Amino-6-chloropyridine-4-carboxylic acid
- 3-Amino-2-chloroisonicotinic acid
- 3-Amino-5-chloropyridine-2-carboxylic acid
- 3-Amino-6-chloropyridine-2-carboxylic acid
- 5-amino-2-chloropyridine-3-carboxylic acid
- 3-Amino-5-chloropyridine-2-carboxylic acid hydrochloride
- 3-Amino-6-chloropyridine-2-carboxylic acid hydrochloride
- 2-Amino-5-chloropyridin-3- ol
- 6-Amino-5-chloro-4-pyrimidinecarboxylic acid
- 2-Amino-5-chloropyrimidine-4-carboxylic acid hydrochloride
- 3-Amino-6-chloro-thieno[3,2-b]pyridine-2-carboxylic acid methyl ester
- 4-Amino-5-chlorothiophene-2-carboxylic acid methyl ester
- 2-Amino-5- cyanobenzamide
- 3-Aminocyclobutanecarboxylic acid
- 1-Amino-cyclobutane-carboxylic acid ethyl ester hydrochloride
- 1-Aminocyclobutanecarboxylic acid hydrochloride
- 3-Aminocyclobutanecarboxylic acid hydrochloride
- 1-Amino-cyclobutane-carboxylic acid methyl ester hydrochloride
- 3-Aminocyclobutanecarboxylic acid tert-butyl ester
- 7-Amino-2-cyclobutyl[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 1-Amino-1-cycloheptanecarboxylic acid
- (1R,2R)-2-Aminocyclohexanecarboxylic acid
- (1S,2S)-2-Aminocyclohexanecarboxylic acid
- (1S,3S)-3-Aminocyclohexanecarboxylic acid
- 3-Aminocyclohexanecarboxylic acid
- (1R,3S)-3-Aminocyclohexanecarboxylic acid hydrochloride
- (1S,3R)-3-Aminocyclohexane-1-carboxylic acid hydrochloride
- (1S,3S)-3-Aminocyclohexane-1-carboxylic acid hydrochloride
- 2-Aminocyclohexane-1-carboxylic acid hydrochloride
- (1R,2S)-(+)-2-Aminocyclohex-3-enecarboxylic acid hydrochloride
- (1S,2S)-2-Aminocyclopentanecarboxylic acid
- (1R,2S)-(-)-2-Amino-1-cyclopentanecarboxylic acid hydrochloride
- (1R,3R)-3-Aminocyclopentanecarboxylic acid hydrochloride
- (1S,3S)-3-Aminocyclopentane-1-carboxylic acid hydrochloride
- (1S,4R)-4-Aminocyclopent-2-enecarboxylic acid hydrochloride
- 5-Amino-3,6-dichloropyrazine-2-carboxylic acid
- 3-Amino-2,6-dichloropyridine-4-carboxylic acid
- 1-Amino-3,3-difluorocyclobutanecarboxylic acid
- 1-Amino-4,4-difluorocyclohexane-1-carboxylic acid
- (1R)-1-Amino-2,2-difluorocyclopropane-1-carboxylic acid
- 1-Amino-2,2-difluorocyclopropane-1-carboxylic acid
- (2S,4R)-4-Amino-2-difluoromethyl-pyrrolidine-1-carboxylic acid tert-butyl ester
- 5-Amino-1-(2,4-difluorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-1-(3,5-difluorophenyl)-1h-pyrazole-4-carboxylic acid
- 2-Amino-4,7-dihydro-5-[2-[4-(carboxy)phenyl]ethyl]-4-oxo-3h-pyrrolo[2,3-d]pyrimidine hydrate
- 6-Amino-1,4-dihydro-4-oxo-2-quinolinecarboxylic acid methyl ester
- 2-Amino-7,8-dihydro-5h-pyrido[4,3-d]pyrimidine-6-carboxylic acid benzyl ester
- 2-Amino-5,8-dihydro-6h-pyrido[3,4-d]pyrimidine-7-carboxylic acid tert-butyl ester
- 2-Amino-7,8-dihydro-6h-pyrido[3,2-d]pyrimidine-5-carboxylic acid tert-butyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyran-3-carboxylic acid tert-butyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyridine-3,6-dicarboxylic acid 6-benzyl ester 3-ethyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyridine-3,6-dicarboxylic acid 6-benzyl ester 3-methyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyridine-3,6-dicarboxylic acid di-tert-butyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]thiopyran-3-carboxylic acid ethyl ester
- 2-Amino-5,8-dimethoxy-1,2,3,4-tetrahydronaphthalene-2-carboxylic acid
- 3-Amino-4,6-dimethylthieno[2,3-b]pyridine-2-carboxylic acid
- 2-Amino-4,5-dimethylthiophene-3-carboxylic acid Synonyms : 2-amino-4,5-dimethylthiophene-3-carboxylic acid
- 2-Amino-4,5-dimethyl-thiophene-3-carboxylic acid methyl ester
- 1-(2-Aminoethyl)cyclopropanecarboxylic acid
- 1-(2-Aminoethyl)cyclopropane-1-carboxylic acid hydrochloride
- 4-(2-Aminoethyl)-5-(2-furyl)-1,2-dihydro-3h- pyrazol-3-one
- 2-Amino-4-ethyl-5-methylthiophene-3-carboxylic acid methyl ester
- 2-Amino-5-ethylthiophene-3-carboxylic acid methyl ester
- 4-Amino-2-ethylthiopyrimidine-5-carboxylic acid
- 4-Amino-2-(ethylthio)-5-pyrimidinecarboxylic acid ethyl ester
- 1-(2-Aminoethyl)-1h-1,2,3-triazole-4-carboxylic acid hydrochloride
- 2-Amino-5- fluorobenzamide
- 3-Amino-4-fluoro-benzofuran-2-carboxylic acid methyl ester
- 3-Amino-6-fluoro-benzofuran-2-carboxylic acid methyl ester
- 3-Amino-2-fluorobenzoic acid
- 4-Amino-6-fluorocinnoline-3-carboxylic acid
- 1-Amino-3-fluoro-cyclobutanecarboxylic acid
- (2R,4R)-4-Amino-6-fluoro-2-methylchroman-4-carboxylic acid
- 3-Amino-5-(3-fluoro-2-methylphenyl)benzoic acid
- 1-[2-Amino-1-(4-fluoro-phenyl)-ethyl]-pyrrolidine-3-carboxylic acid
- 5-Amino-1-(2-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-1-(4-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- (1R,3S,4S)-3-Amino-4-hydroxy-cyclohexanecarboxylic acid ethyl ester hydrochloride
- (1S,3R,4R)-3-Amino-4-hydroxy-cyclohexanecarboxylic acid ethyl ester hydrochloride
- 2-Amino-4-hydroxy-7,8-dihydro-5h-pyrido[4,3-d]pyrimidine-6-carboxylic acid tert-butyl ester
- 5-Amino-1-(2-hydroxyethyl)pyrazole-4-carboxylic acid
- 1-Amino-3-(hydroxymethyl)cyclobutane-1-carboxylic acid
- 4-Amino-2-hydroxypyrimidine-5-carboxylic acid hydrobromide
- 3-Amino-3-imino-propanoic acid, ethyl ester hydrochloride
- 1-Aminoindan-1-carboxylic acid
- 2-Amino-2-indancarboxylic acid
- 3-Amino-1H-indazole-5-carboxylic acid
- 3-Amino-1h-indazole-4-carboxylic acid
- 3-Amino-1h-indazole-6-carboxylic acid
- 5-Amino-1H-indazole-3-carboxylic acid
- 6-Amino-1h-indazole-7-carboxylic acid
- 4-Amino-5-iodo-2-methoxybenzenecarboxylic acid
- 3-Amino-6-iodopyrazine-2-carboxylic acid
- 5-Amino-6-iodo-2-pyrazinecarboxylic acid
- N-(6-Amino-1-isobutyl-2,4-dioxo-1,2,3,4-tetrahydro-pyrimidin-5-yl) -4-chloro-n-(2-methoxy-ethyl)-butyramide
- 3-Aminoisoquinoline-6-carboxylic acid
- 4-Aminoisoquinoline-6-carboxylic acid
- 4-Aminoisoquinoline-8-carboxylic acid
- 7-Aminoisoquinoline-4-carboxylic acid
- 8-Aminoisoquinoline-5-carboxylic acid
- 3-Aminoisoquinoline-8-carboxylic acid hydrochloride
- 4-Amino-2-mercaptopyrimidine-5-carboxylic acid
- 3-Amino-4- methanesulfonylbenzonitrile
- 2-Amino-5-(4-methoxy-phenylcarbamoyl)-4-methyl-thiophene-3-carboxylic acid ethyl ester
- 1-[2-Amino-1-(2-methoxy-phenyl)-ethyl]-pyrrolidine-3-carboxylic acid
- 4-Amino-1-(4-methoxyphenyl)-1h-pyrazole-3-carboxylic acid
- 5-Amino-1-(4-methoxyphenyl)-1h-pyrazole-4-carboxylic acid
- 6-Amino-5-methoxy-pyridine-2-carboxylic acid methyl ester
- 4-Amino-6-methoxyquinoline-3-carboxylic acid ethyl ester
- 2-Amino-6-methoxy-1,2,3,4-tetrahydro-naphthalene-2-carboxylic acid
- 2-Amino-8-methoxy-1,2,3,4-tetrahydro-naphthalene-2-carboxylic acid
- 2-Aminomethyl-7-azaspiro[3.5]nonane-7-carboxylic acid tert-butyl ester
- 2-Amino-3- methylbenzamide
- 3-(Aminomethyl)cyclobutanecarboxylic acid hydrochloride
- 1-Aminomethyl-cyclohexanecarboxylic acid methyl ester hydrochloride
- 1-Aminomethyl-3,4-dihydro-1h-isoquinoline-2-carboxylic acid tert-butyl ester oxalate
- 3-Aminomethyl-3-fluoropiperidine-1-carboxylic acid tert-butyl ester
- 5-(Aminomethyl)furan-2-carboxylic acid hydrochloride
- 5-Amino-1-methyl-1h-indazole-3-carboxylic acid
- (R)-3-Aminomethyl-morpholine-4-carboxylic acid tert-butyl ester
- 3-Aminomethyl-morpholine-4-carboxylic acid tert-butyl ester
- 1-Aminomethyl-2-oxa-5-aza-bicyclo[2.2.1]heptane-5-carboxylic acid tert-butyl ester
- 7-Aminomethyl-6-oxa-2-aza-spiro[3.4]octane-2-carboxylic acid tert-butyl ester
- 5-Amino-1-(4-methylphenyl)-1H-pyrazole-4-carboxylic acid
- 4-Amino-1-methyl-4-piperidinecarboxylic acid
- 4-Amino-1-methylpiperidine-4-carboxylic acid hydrochloride
- 4-Amino-1-methyl-4-piperidinecarboxylic acid methyl ester
- 3-Amino-5-methylpyrazine-2-carboxylic acid
- 3-Amino-1-methyl-1h-pyrazole-4-carboxylic acid
- 3-Amino-1-methyl-1h-pyrazole-5-carboxylic acid
- 4-Amino-1-methyl-1h-pyrazole-3-carboxylic acid
- 4-Amino-3-methyl-1h-pyrazole-5-carboxylic acid
- 5-Amino-1-methyl-1h-pyrazole-3-carboxylic acid
- 4-Amino-1-methyl-1h-pyrazole-5-carboxylic acid hydrochloride
- 4-Amino-3-methyl-pyrazole-1-carboxylic acid tert-butyl ester
- 5-(Aminomethyl)pyrazolo[1,5-a]pyridine-3-carboxylic acid hydrochloride
- 7-Amino-2-methylpyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 7-Amino-3-methylpyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 2-Amino-4-methylpyridine-3-carboxylic acid
- 2-Amino-6-methyl-3-pyridinecarboxylic acid
- 3-Amino-2-methylpyridine-5-carboxylic acid
- 3-Amino-4-methylpyridine-2-carboxylic acid
- 3-Amino-5-methyl-pyridine-2-carboxylic acid amide
- 5-Amino-6-methyl-3-pyridinecarboxylic acid ethyl ester
- 5-Amino-2-methylpyridine-4-carboxylic acid hydrochloride
- 2-Amino-4-methyl-pyrimidine-5-carboxylic acid
- 4-Amino-6-methyl-pyrimidine-2-carboxylic acid methyl ester
- 4-Amino-1-methyl-1h-pyrrole-2-carboxylic acid hydrochloride
- 2-Aminomethyl-pyrrolidine-1-carboxylic acid benzyl ester
- 2-Amino-7-methyl-7h-pyrrolo[2,3-d]pyrimidin-6-carboxylic acid methyl ester
- 7-Amino-2-(methylsulfanyl)[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 3-(Aminomethyl)-1-[(tert-butoxy)carbonyl]azetidine-3-carboxylic acid hydrochloride
- 4-Aminomethyltetrahydropyran-4-carboxylic acid
- 4-(Aminomethyl)tetrahydro-2h-pyran-4-carboxylic acid methyl ester hydrochloride
- 2-Amino-5-methylthiophene-3-carboxylic acid
- 3-Amino-4-methylthiophene-2-carboxylic acid
- 4-Amino-5-methylthiophene-3-carboxylic acid
- 4-Amino-2-(methylthio)pyrimidine-5-carboxylic acid
- 7-Amino-2-methyl[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 5-Amino-1-methyl-3-(trifluoromethyl)-1h-pyrazole-4-carboxylic acid
- 2-Amino-3- nitrobenzonitrile
- 3-Amino-5-(3-nitro-phenyl)-thiophene-2-carboxylic acid ethyl ester
- 3-Amino-7-oxabicyclo[2.2.1]hept-5-ene-2-carboxylic acid hydrochloride
- 1-Amino-4-oxocyclohexanecarboxylic acid ethylene ketal
- 3-Amino-5-oxo-4,5-dihydro-pyrazine-2-carboxylic acid methyl ester
- 6-Amino-4-oxo-1,4-dihydroquinoline-2-carboxylic acid
- 2-Amino-4-oxo-1,5,7,8-tetrahydro-4h-pyrido[4,3-d]pyrimidine-6-carboxylic acid tert-butyl ester
- 4-(4-Aminophenethyl)piperazine-1-carboxylic acid tert-butyl ester
- 3-Amino-5-phenylbenzoic acid
- 2-(3-Aminophenyl)-6-bromoquinoline-4-carboxylic acid
- 2-(3-Aminophenyl)-6-chloroquinoline-4-carboxylic acid
- 2-(4-Aminophenyl)-6-chloroquinoline-4-carboxylic acid
- 1-(3-Amino-phenyl)-cyclopropanecarboxylic acid amide
- 2-(3-Aminophenyl)-6,8-dimethylquinoline-4-carboxylic acid
- 1-(3-Aminophenyl)-5-oxo-4,5-dihydro-1h-pyrazole-3-carboxylic acid
- 1-(4-Aminophenyl)-2-oxopyridine-3-carboxylic acid
- 4-Amino-1-phenyl-1h-pyrazole-3-carboxylic acid
- 5-Amino-1-phenyl-1h-pyrazole-4-carboxylic acid
- 2-Amino-4-phenylpyrimidine-5-carboxylic acid
- 2-(3-Aminophenyl)quinoline-4-carboxylic acid
- 2-(4-Amino-phenyl)-quinoline-4-carboxylic acid
- 3-Amino-5-phenylthiophene-2-carboxylic acid
- 4-Aminopiperidine-4-carboxylic acid
- 1-Aminopropan-2-one hydrochloride
- 4-(3-Amino-propionyl)-piperazine-1-carboxylic acid tert-butyl ester
- 1-(3-Aminopropyl)-5-tert-butyl-2-methylpyrrole-3-carboxylic acid
- 3-Amino-1H-pyrazole-5-carboxylic acid
- 5-Amino-1h-pyrazole-4-carboxylic acid
- 5-Amino-1h-pyrazole-3-carboxylic acid hydrochloride
- 5-Amino-pyrazole-1,4-dicarboxylic acid 1-tert-butyl ester 4-ethyl ester
- 2-Aminopyrazolo[1,5-a]pyrimidine-3-carboxylic acid
- 7-Aminopyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 3-(5-Amino-1h-pyrazol-3-yl)-azetidine-1-carboxylic acid tert-butyl ester
- 5-Amino-pyridazine-4-carboxylic acid
- 6-Aminopyridazine-3-carboxylic acid
- 6-Aminopyridine-2-carboxylic acid ethyl ester
- 4-Aminopyridine-2-carboxylic acid monohydrate
- 4-Aminopyridine-2,6-dicarboxylic acid
- 4-(6-Amino-pyridin-3-yloxy)-pyridine-2-carboxylic acid methylamide
- 5-Amino-pyrimidine-4-carboxylic acid
- 3-Amino-pyrrolidine-1,3-dicarboxylic acid 1-tert-butyl ester
- 5-Amino-1h-pyrrolo[2,3-b]pyridine-2-carboxylic acid
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-5-carboxylic acid
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-6-carboxylic acid
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-5-carboxylic acid methyl ester
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-6-carboxylic acid methyl ester
- 4-Amino-7h-pyrrolo[2,3-d]pyrimidine-5-carboxylic acid
- 2-Aminoquinoline-3-carboxylic acid
- 2-Aminoquinoline-6-carboxylic acid
- 3-Aminoquinoline-5-carboxylic acid
- 3-Aminoquinoline-6-carboxylic acid
- 3-Aminoquinoline-7-carboxylic acid
- 3-Aminoquinoline-8-carboxylic acid
- 4-Aminoquinoline-6-carboxylic acid
- 5-Aminoquinoline-3-carboxylic acid
- 7-Aminoquinoline-5-carboxylic acid
- 3-Amino-1-(tert-butoxycarbonyl)-3-azetidinecarboxylic acid
- 5-Amino-2,3,4,5-tetrahydro-benzo[b]azepine-1-carboxylic acid tert-butyl ester
- 2-Amino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylic acid
- 2-Amino-4,5,6,7-tetrahydro-benzo[b]thiophene-3-carboxylic acid isopropyl ester
- 2-Amino-4,5,6,7-tetrahydro-benzo[b]thiophene-3-carboxylic acid methyl ester
- 2-Amino-4,5,6,7-tetrahydro-benzo[b]thiophene-3-carboxylic acid tert-butyl ester
- 3-Aminotetrahydrofuran-3-carboxylic acid
- 3-Aminotetrahydro-3-furancarboxylic acid butyl ester
- 1-Amino-1,2,3,4-tetrahydro-1-naphthalenecarboxylic acid hydrochloride
- 2-Amino-1,2,3,4-tetrahydro-naphthalene-2-carboxylic acid hydrochloride
- 3-Amino-5,6,7,8-tetrahydrothieno[2,3-b]quinoline-2-carboxylic acid
- 3-Amino-tetrahydro-thiophene-3-carboxylic acid
- 2-Amino-6h-[1,3,4]thiadiazine-5-carboxylic acid ethyl ester hydrochloride
- 2-Amino-6h-1,3,4-thiadiazine-5-carboxylic acid hydrochloride
- 4-Aminothieno[3,2-d]pyrimidine-7-carboxylic acid
- 5-Aminothieno[2,3-d]pyrimidine-6-carboxylic acid
- 4-Aminothiophene-2-carboxylic acid
- 3-Aminothiophene-2-carboxylic acid potassium salt
- 3-Amino-thiophene-2-carboxylic acid sodium salt
- 3-Amino-3- thioxopropanamide
- 6-Amino-[1,2,4]triazine-5-carboxylic acid
- 5-Amino-4h-1,2,4-triazole-3-carboxylic acid
- 3-Amino[1,2,4]triazolo[4,3-a]pyridine-6-carboxylic acid
- 7-Amino[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 6-(5-Amino-1h-1,2,4-triazol-3-yl)cyclohex-3-ene-1-carboxylic acid
- Amino-tri-(carboxyethoxymethyl)- methane
- 4-Amino-3,5,6-trichloropyridine-2-carboxylic acid
- 5-Amino-1-[4-(trifluoromethoxy)phenyl]-1h-pyrazole-4-carboxylic acid
- 2-Amino-3,3,3-trifluoropropanoic acid hydrochloride
- 2-[(3-Amino-2,4,6-triiodophenyl)methyl]butanoic acid
- (1R,2R,3S,5R)-2-Amino-2,6,6-trimethyl-bicyclo[3.1.1]heptane-3-carboxylic acid methyl ester hydrochloride
- Ammonium adipate
- Ammonium 2-(1,3-benzodioxol-5-yloxy)-2-methylpropanoate
- Ammonium carbamate
- Ammonium citrate dibasic
- Ammonium dodecanedioate
- Ammonium ([2-(2-furyl)cyclohexyl]oxy)acetate
- Ammonium hydrogen maleate
- Ammonium succinate
- Amphotericin b
- Angelic acid
- Angelic acid isoamyl ester
- Angelic acid isobutyl ester
- Angelic anhydride
- 1,5-Anhydro-3-o-benzoyl-4,6-o-benzylidene-2-deoxy- 2-(n4-benzoylcytidin-1-yl)-d-altro-hexitol
- 2-Anilinobenzoic acid methyl ester
- 1[(Anilinocarbonyl)amino]cyclopropanecarboxylic acid
- 4-[5-(Anilinocarbonyl)-1,3,4-thiadiazol-2-yl]butanoic acid
- 3-[5-(Anilinocarbonyl)-1,3,4-thiadiazol-2-yl]propanoic acid
- 2-Anilinonicotinic acid
- Ansamitocin p 3'
- 1-Anthracenecarboxylic acid
- 2-Anthracenecarboxylic acid
- 9-Anthracenecarboxylic acid
- (1R,2R)-2-(Anthracene-2,3-dicarboximido) cyclohexanecarboxylic acid
- (1S,2S)-2-(Anthracene-2,3-dicarboximido) cyclohexanecarboxylic acid
- 9-Anthracenylmethyl acrylate
- 9-Anthracenylmethyl methacrylate
- Anthraquinone-2-carboxylic acid
- Anthraquinone-2,3-dicarboxylic acid
- Anti-3-oxotricyclo[2.2.1.0(2,6)]heptane-7-carboxylic acid
- D-Araboascorbic acid
- Arachidic acid
- Arachidic acid ethyl ester
- Arachidic acid methyl ester
- Arachidic anhydride
- (3Ar*,6ar*)-benzylhexahydropyrrolo [3,2-b]pyrrole-1(2h)-carboxylate
- (3S,3Ar,5r,6ar,7r)-6-iodo-2-oxooctahydro-3,5-methanocyclopenta[b]pyrrole-7-carboxylic acid
- (4Ar,8ar)-rel-1-methyl-decahydro-1,5-naphthyridine, oxalic acid
- (3aR,6aR)-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-1(2H)-carboxylate
- (3aR,6aR)-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-5(1H)-carboxylate
- (4Ar,9ar)-tert-butyl octahydro-[1,4]oxazino[3,2-c]azepine-4(4ah)-carboxylate
- (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-6,10,10b-Trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-3-vinyldodecahydro-1H-benzo[f]chromen-5-yl acetate
- (3S,3Ar,5s,6as,7s)-2-oxooctahydro-3,5-methanocyclopenta[b]pyrrole-7-carboxylic acid
- (3Ar,5s,6as)-tert-butyl 5-hydroxyhexahydrocyclopenta [c]pyrrole-2(1H)-carboxylate
- (3Ar,6as)-tert-butyl 5-oxohexahydrocyclopenta [c]pyrrole-2(1h)-carboxylate
- (4Ar,7as)-tert-butyl tetrahydro-2h-[1,4]dioxino[2,3-c]pyrrole-6(3h)-carboxylate
- Ar-13324 hydrochloride
- (1S,7S,8S,8aR)-8-(2-((2R,4R)-4-Hydroxy-6-oxotetrahydro-2H-pyran-2-yl)ethyl)-7-methyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (S)-2-methylbutanoate
- (3S,11Ar)-6-methoxy-3-methyl-5,7-dioxo-2,3,5,7,11,11a-hexahydrooxazolo[3,2-d]pyrido[1,2-a]pyrazine-8-carboxylic acid
- (9Ar)-octahydro-1h-pyrido[1,2-a]piperazine oxalic acid
- (6S,8Ar)-3-oxooctahydroindolizine-6-carboxylic acid
- (3aS,9aR,10aR,10bS,E)-6,9a- Dimethyl-3-methylene-3a,4,5,8,9,9a,10a,10b-octahydrooxireno[2',3':9,10]cyclodeca[1,2-b]furan-2(3H)-one
- (3As,4r,5s,6ar)-(+)-hexahydro-5-hydroxy- 4-(hydroxymethyl)-2h-cyclopenta[b]furan-2-one
- (3As,4s,6ar)-4-methoxytetrahydrofuro [3,4-b]furan-2(3h)-one
- (1R,4As,10ar)-methyl 7-isopropyl-1,4a-dimethyl-1,2,3,4,4a,9,10,10a-octahydrophenanthrene-1-carboxylate
- (2R,3aS,6S,6aS,7R,10R,11aR,11bS)-2-(Furan-3-yl)- 11b-methyloctahydro-4H-3a,6:7,10-dimethanofuro[2,3-c]oxepino[4,5-e]oxepine-4,8(6H)-dione
- (3As,5r,6s,6as)-6-hydroxy-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxole-5-carboxylic acid Synonyms : (3aS,5R,6S,6aS)-6-hydroxy-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxole-5-carboxylic acid
- (4As,8as)-tert-butyl hexahydro-1h-pyrido[3,4-b][1,4]oxazine-6(7h)-carboxylate
- (3As*,6as*)-tert-butylhexahydropyrrolo [3,4-c]pyrrole-2(1H)-carboxylate
- (3aS,6aS)-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-5(1H)-carboxylate
- L-Ascorbic acid
- L-Ascorbic acid 2-phosphate sesquimagnesium salt hydrate
- L-Ascorbyl 2,6-dibutyrate
- L-Ascorbyl 2,6-dipalmitate
- (4R,12As)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-pyrido[1',2':4,5]pyrazino[2,1-b][1,3]oxazine-9-carboxylic acid
- (9As)-octahydro-1h-pyrido[1,2-a]piperazine, oxalic acid
- Atractyloside potassium salt
- Atracurium oxalate
- Atropic acid
- 7- Geranyloxycoumarin
- Aurintricarboxylic acid
- AVERMECTIN B1A
- 7-Azabicyclo[2.2.1]heptane-2-carboxylic acid ethyl ester hydrochloride
- (1R*,2S*,4S*)-7-Aza-bicyclo[2.2.1]heptane-2-carboxylic acid hydrobromide
- 2-Azabicyclo[2.2.1]heptane-6-carboxylic acid hydrochloride
- 2-Azabicyclo[3.1.1]heptane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.1]heptane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[4.1.0]heptane-6-carboxylic acid hydrochloride
- 3-Azabicyclo[4.1.0]heptane-3-carboxylic acid, 6-(hydroxymethyl)-, 1,1-dimethylethyl ester
- 2-Aza-bicyclo[2.2.1]heptane-2,6-dicarboxylic acid 2-tert-butyl ester
- 2R-7-Aza-bicyclo[2.2.1]heptane-2,7-dicarboxylic acid 7-tert-butyl ester
- 2-Azabicyclo[2.2.1]heptane-2-propanoic acid
- 2-Azabicyclo[2.2.1]hept-5- en-3-one
- 3-Azabicyclo[3.1.0]hexane-1-carboxylic acid
- (1R,5S,6S)-3-Azabicyclo[3.1.0]hexane-6-carboxylic acid hydrochloride
- 2-Azabicyclo[2.1.1]hexane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.0]hexane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.0]hexane-6-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.0]hexane-1-carboxylic acid, methyl ester, hydrochloride (1:1)
- 2-Azabicyclo[2.1.1]hexane-2,5-dicarboxylic acid 2-tertbutyl ester
- 8-Azabicyclo[3.2.1]octane-8-carboxylic acid, 3-(hydroxymethyl)-, 1,1-dimethylethyl ester, (3-endo)-
- 2-Azabicyclo[2.2.2]octane-2-carboxylic acid, 5-oxo-1,1-dimethylethyl ester
- 8-Azabicyclo[3.2.1]octane-3,8-dicarboxylic acid, 8-(1,1-dimethylethyl) ester, (3-endo)-
- 2-Azabicyclo[2.2.2]octane-2,6-dicarboxylic acid 2-tertbutyl ester
- 2-Azabicyclo[2.2.2]oct-7-ene-6-carboxylic acid
- 1-Azabicyclo[2.2.2]oct-2-ene-3-carboxylic acid methyl ester hydrochloride
- Azadibenzocyclooctyne acid
- 2-Azaspiro[4.5]decane-7-carboxylic acid
- 5-Azaspiro[2.4]heptane-1-carboxylic acid
- 2-Azaspiro[3.3]heptane-2-carboxylic acid tert-butyl ester
- 1-Azaspiro[3.3]heptane hemioxalate
- 2-Azaspiro[3.3]heptane hemioxalate
- 4-Azaspiro[2.4]heptane hemioxalate
- 2-(5-Azaspiro[2.4]heptan-7-yl)acetic acid
- 5-Azaspiro[2.3]hexane hemioxalate
- 2-Azaspiro[4.4]nonane-2-carboxylic acid, 7-hydroxy-, 1,1-dimethylethyl ester
- 2-Aza-spiro[4.4]nonane hemioxalate
- 2-Aza-spiro[4.4]non-7-ene-2-carboxylic acid tert-butyl ester
- 2-Azaspiro[3.4]octane hemioxalate
- 5-Azaspiro[3.4]octane hemioxalate
- 6-Azaspiro[3.4]octane hemioxalate
- 5-Azaspiro[3.4]octane oxalate
- 3-Azaspiro[5.5]undecane-3,9-dicarboxylic acid 3-(tert-butyl) ester
- Azaerythromycin A
- Azelaic acid monomethyl ester
- 3-(1-AZEPANYL)BUTANOIC ACID
- (([5-(Azepan-1-ylcarbonyl)-1,2,4-oxadiazol-3-yl]methyl)thio)acetic acid
- 3-(([5-(Azepan-1-ylcarbonyl)-1,2,4-oxadiazol-3-yl]methyl)thio)propanoic acid
- 2-Azepan-1-ylisonicotinic acid
- (2E)-3-[3-(Azepan-1-ylmethyl)-4-methoxyphenyl]acrylic acid
- 3-Azepan-1-yl-2-methylpropanoic acid
- Azepan-1-yl(oxo)acetic acid
- 3-Azepan-1-ylpropanoic acid
- 5-(Azepan-1-ylsulfonyl)-2-methylbenzoic acid
- Azetidine-3-carboxylic acid
- 1-Azetidinecarboxylic acid, 3-fluoro-3-(hydroxymethyl)-, phenylmethyl ester
- 1-Azetidinecarboxylic acid, 3-[4-(phenylmethyl)-1-piperazinyl], 1,1-dimethylethyl ester
- 1,3-Azetidinedicarboxylic acid, 3-amino-, 1-(1,1-dimethylethyl) 3-methyl ester
- 2-Azetidinepropanoic acid, 2-(methoxycarbonyl)-1-(phenylmethyl)-, methyl ester
- Azetidine-1,3,3-tricarboxylic acid 1-tert-butyl ester 3-ethyl ester
- 2- Azetidinone
- Azetidin-3-yl acetate
- Azetidin-3-yl-acetic acid
- 2-(Azetidin-3-yl)acetic acid hydrochloride
- 1-(Azetidin-3-yl)azetidin-3-ol oxalate
- 2-(Azetidin-3-yl)-2-methylpropanoic acid
- 3-(Azetidin-1-yl)propanoic acid
- 4-(Azetidin-3-yl)tetrahydro-2h-pyran-2-one hydrochloride
- 1-Azido-1-deoxy-beta-d-galactopyranoside tetraacetate
- O-(2-Azido-2-deoxy-3, 4,6-tri-o-acetyl-beta-d-galactopyranosyl)-trichloroacetimidate
- 2-[2-(2-Azidoethoxy)ethoxy]ethyl 2,3,4,6-tetra-o-acetyl-d-galactopyranoside
- Azido-peg12- acid
- 2-(Azido-peg3-amido)-1,3-bis(carboxylethoxy) propane
- 4-Azidosulfonyl-benzoic acid 2,5-dioxo-pyrrolidin-1-yl ester
- 11-Azido-3,6,9-trioxaundecanoic acid
- Azilsartan medoxomil
- 2-(((3-(Aziridin-1-yl)propanoyl)oxy)methyl)-2-ethylpropane-1,3-diyl bis(3-(aziridin-1-yl)propanoate)
- Azobenzene-4,4'-dicarboxylic acid
- Azobenzene-4,4'-dicarboxylic acid dimethyl ester
- 3-Azocan-1-ylpropanoic acid hydrochloride
- 3,3'-Azodibenzoic Acid
- Azoxybenzene-4,4'-dicarboxylic acid diethyl ester
- Balofloxacin dihydrate
- Bapta tetraethyl ester
- Docosanoic acid
- Behenic Anhydride
- Behenyl acrylate
- Bendamustine hydrochloride hydrate
- Benzal diacetate
- Benzamidine hydrochloride
- 2-Benzamidoethyl benzoate
- 2-(3-Benzamidophenyl)acetic acid
- (Z)-N'-Hydroxybenzimidamide hydrobromide
- Benzeneacetic acid, alpha,alpha-diphenyl-, rhodium(2+) salt (2:1)
- 1,4-Benzenediacrylic acid
- 1,2-Benzenedicarboxylic acid,4-chloro-,1,2-dimethyl ester
- Benzene-1,2,3,4,5,6-hexacarboxylic acid
- Benzenepentacarboxylic acid
- Benzenepropanoic acid, alpha-(hydroxymethyl)-, ethyl ester
- Benzenepropanoic acid, 4-methoxy-3-(3-methoxypropoxy)-a-(1-methylethyl)-, (ar)-
- 5-Benzenesulfonyl-5h-pyrrolo[3,2-d]pyrimidine-6-carboxylic acid
- 1,2,3-Benzenetricarboxylic acid
- 1,2,4-Benzenetricarboxylic acid
- 1,2,3-Benzenetricarboxylic acid hydrate
- 1,2,4-Benzenetricarboxylic acid tripropyl ester
- 1-Benzhydrylazetidin-3-yl 2-cyanoacetate
- 1H-Benzimidazole-1-carboxylic acid, 2,3-dihydro-2-oxo-, 1,1-dimethylethyl ester
- 1H-Benzimidazole-7-carboxylic acid, 1-[[2'-(2,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl)[1,1'-biphenyl]-4-yl]methyl]-2-ethoxy-, methyl ester
- Benzimidazole-5,6-dicarboxylic acid
- 3-(1H-Benzimidazol-1-yl)butanoic acid
- 3-(1H-Benzimidazol-2-yl)-2H-chromen -2-one
- 3-(1H-Benzimidazol-2-yl)-3-hydroxypropanoic acid
- Imidazoles
- 2-Amino-5, 6-dichlorobenzimidazole
- N-(6-Aminohexyl)-5-((3as,4s,6ar)-2-oxohexahydro-1h-thieno[3,4-d]imidazol-4-yl) pentanamide
- N-(4-Aminobutyl) biotinamide
- D4476 CK1 Inhibitor
- 2-Amino-6-chloroimidazo[1,2-b] pyridazine
- 5-Amino-1h-imidazol- 4-carbonitrile
- 2- Aminobenzimidazole
- 8- Aminoguanosine
- 1-Amino-1h-imidazole- 5-carboxamide
- 1-(4-Aminobenzyl)-n-[3-(trifluoromethyl)phenyl]- 1h-imidazole-4-carboxamide
- 2-Amino-9-(4-hydroxy-3-(hydroxymethyl)butyl)-1,9-dihydro-6H-purin-6- one
- 3-Amino-5-ethyl-5-methylimidazolidine-2, 4-dione
- 5-Amino-1,4-dimethylimidazole hydrochloride
- 4-Amino- 5-imidazolecarboxamide
- 1-Acetyl- 2-imidazolidinone
- 1-Allyl-2-(trifluoromethyl) benzimidazole
- 1-Aminobenzimidazole-2-sulfonic acid
- 4- Acetylimidazole
- 1-Acetyl- 2-thiohydantoin
- Allylchloro[1,3-bis(2,6-di-i-propylphenyl) -4,5-dihydroimidazol-2-ylidene]palladium(ii)
- 2-[(2-Allylphenoxy)methyl] -1h-benzimidazole
- 1-(4-Aminobenzyl)-n-(2,4-dimethylphenyl)- 1h-imidazole-4-carboxamide
- 1-(5-Acetyl-4-methylthiazol-2-yl) imidazolidin-2-one
- 4-Amino-1h-imidazole- 5-carbonitrile
- 3-Acetyl-6-chloroimidazo[1,2-b] pyridazine
- 5- Aminobenzimidazolone
- 5-Amino-4-bromo- benzimidazole
- 2- Acetylimidazole
- 1-(4-Acetylphenyl) imidazole
- 2-Amino-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl]-6, 9-dihydro-1H-purin-6-one
- 2-Amino-9-[4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-1, 9-dihydro-6h-purin-6-one
- 2-Amino-9-((1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl)-1,9-dihydro-6H-purin-6-one hydrate
- 1-(4-Aminobenzyl)-n-(5-chloro-2-methylphenyl)- 1h-imidazole-4-carboxamide
- 5-Aminoimidazole-4-carboxamide 1-beta-D-ribofuranoside
- 2-Amino-1,9-dihydro-9-[(1s,3r,4s)-4-(benzyloxy)-3-(benzyloxymethyl)-2-methylenecyclopentyl]- 6h-purin-6-one
- 1-(4-Aminobenzyl)-n-phenyl-1h-imidazole- 4-carboxamide
- Acetylene-PEG4-biotin conjugate
- 1-(4-Aminobenzyl)-n-(3-methoxyphenyl)- 1h-imidazole-4-carboxamide
- 9-[(2-Acetoxyethoxy)methyl] -acetylguanine
- Ac-his-oh h2o
- 2-Amino-1h-benzo[d]imidazole- 5-carbonitrile
- 2-Amino-5, 6-dimethylbenzimidazole
- 1-(4-Aminobenzyl)-n-(3,4-dimethoxyphenyl)-1h-imidazole- 4-carboxamide
- 2-Amino-7-bromo-1h,4h-imidazo[2,1-f][1,2,4] triazin-4-one
- 1-Allyl-3-methylimidazolium bromide
- 1-(4-Aminobenzyl)-n-(2-methoxy-5-methylphenyl)- 1h-imidazole-4-carboxamide
- 1-(2-Aminoethyl)- 2-imidazolidone
- 1- Acetylimidazole
- 1-Allyl-3-methylimidazolium chloride
- 3-Acetoxy- 5-bromoindole
- 4-Amino-7-bromoimidazo[2,1-f][1,2,4] triazine
- 2-[(4-Allyl-2-methoxyphenoxy)methyl] -1h-benzimidazole
- (1-[3-(Acetylamino)phenyl]-2,5-dioxoimidazolidin-4-yl)acetic acid
- 3-Amino-n-cyclopropyl-4-(1h-imidazol-1-yl) benzamide
- 1-(1-Adamantyl)- 1h-benzimidazole-2-thiol
- Alpha-(2,3-dimethylphenyl)-1-(trityl)-1h-imidazole- 4-methanol
- 1-Allyl-3-methylimidazolium dicyanamide
- 1- Allylhydantoin
- Alpha-[5-(1,2-dithiolan-3-yl)pentamido]-omega-biotinyl undeca(ethylene glycol)
- Ac-beta-ala-his-ser-his- oh
- 1-(5-Acetyl-4-methylthiazol- 2-yl)imidazolidin-2-one
- 1-(4-Aminobenzyl)-n-(3-chlorophenyl)-1h-imidazole- 4-carboxamide
- N(Alpha), n-(im)-di-boc-l-histidine methyl ester
- 1- Allylimidazole
- Acalabrutinib .
- 1-(4-Aminobenzyl)-n-(3,5-dimethylphenyl)- 1h-imidazole-4-carboxamide
- 1-(4-Aminobenzyl)-n-(4-ethylphenyl)- 1h-imidazole-4-carboxamide
- 1-(4-Aminobenzyl)-n-(4-methoxyphenyl)- 1h-imidazole-4-carboxamide
- N-Acetyl-l- carnosine
- 2-Amino-9-(4-hydroxy-3-(hydroxymethyl)butyl) -1,9-dihydro-6H-purin-6-one
- N-[2-[2-(2-Aminoethoxy)ethoxy]ethyl] biotinamide
- 6-Amino-3-bromo-imidazo[1,2-a] pyridine
- 2-Amino-7-benzyl-1h-purin-6(7h)- one
- N-(2-Aminoethyl) biotinamide
- 3-(4-Aminobenzyl)-5,5-dimethylimidazolidine-2, 4-dione
- (2R)-2-Acetamido-3-(1h-imidazol-4-yl)propanoic acid
- 2-amino-9-{[(1,3-dihydroxypropan-2-yl)oxy]methyl}-6, 9-dihydro-1H-purin-6-one
- 1-(4-Aminobenzyl)-n-(4-fluoro-2-methylphenyl)- 1h-imidazole-4-carboxamide
- N-Acetyl-dl-histidine monohydrate
- 5- Aminobenzimidazole
- Z-Ala-his -ome
- Allyl 1h-imidazole-1-carboxylate
- 3-Amino-5,5-diphenylimidazolidine-2, 4-dione
- Ac-his- nhme
- 3-Amino-6-chloroimidazo[1,2-b] pyridazine
- 2-Amino-1-Boc -imidazole
- 2-Amino-6-ethyl-7-hydroxy-3-(4-methyl-1h-imidazol-2-yl)- 4h-chromen-4-one
- N-Alpha-methyl-l-histidine hydrochloride
- Alpha-(2,4-dichlorophenyl) -1h-imidazole-1-ethanol
- 2-[(4-Allyl-2-methoxyphenoxy)methyl]- 1h-benzimidazole
- Acyclovir-side chain-2-3h
- 2-Amino-9-[(2-hydroxyethoxy)methyl]- 6,9-dihydro-1H-purin-6-one
- 5-Amino-1-((3ar,4r,6r,6ar)-6-(hydroxymethyl)-2,2-dimethyltetrahydrofuro[3,4-d][1,3]dioxol-4-yl)- 1h-imidazole-4-carboxamide
- 6-Amino-3-chloroimidazo[1,2-a] pyridine
- 3-Acetyl-2-methylimidazo [1,2-a]pyridine
- 3-Amino-1,3-diaza-spiro[4.5]decane-2, 4-dione
- 8- Aminoguanine
- 1-Allyl-3- methylimidazolium
- Alpha-methyl-dl-histidine dihydrochloride
- 2-Acetamido- 6-hydroxypurine
- 2-Amino-4-bromo- 1-methylimidazole
- 1-(4-Aminobenzyl)-n-(2,4-difluorophenyl)- 1h-imidazole-4-carboxamide
- (R)-(-)-Alpha-methylhistamine dihydrochloride
- Ac-His(1-Trt)- OH
- 3'-Amino-2', 3'-dideoxyguanosine
- 2-Amino-9-((2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-1h-purin-6(9h)-one dihydrate
- 1-Allyl-1h-benzimidazole- 2-thiol
- Alpha-methyl-imidazo[1,2-a]pyridine- 3-methanol
- Deuterated
- 1-Bromo-3-(methoxy-d3) -5-nitrobenzene
- 1-(Benzyloxy)-3-bromo-5-(methoxy-D3) benzene
- Methyl 2-(methoxy-d3)benzoate
- Acetonitrile-d3 98%
- 5-Amino-1-(methyl-d3) pyridin-2-one
- 5-Amino-1-(methyl-d3)pyridin-2-one hydrochloride
- 1-(Benzyloxy)-3-chloro-5-(methoxy-d3) benzene
- 4-(Benzyloxy)-1-chloro-2-(methoxy-d3) -benzene
- 4-(Benzyloxy)-2-chloro-1-(methoxy-D3) benzene
- 1-(Benzyloxy)-3-(methoxy-d3)-5-(trifluoromethoxy) benzene
- 4-(Benzyloxy) -1-(methoxy-d3)-2-(trifluoromethyl)benzene
- 4- (Benzyloxy)-1-(methoxy-d3)-2-(trifluoromethyl)benzene
- (S)-1-Boc-2-(n-methyl-d3-aminomethyl)- pyrrolidine
- 3-Bromo-4-(methoxy-d3) aniline
- 3-Bromo-5-(methoxy-d3) aniline
- 4-Bromo-3-(methoxy-d3) aniline
- 3-Bromo-4-(methoxy-d3)benzoic acid
- 5-Bromo-2-(methoxy-d3)benzoic acid
- 3-Bromo-5-(methoxy-d3) phenol
- 5-Bromo-1-(methyl-d3) indole
- 1-(2-Bromophenyl)-4-(methyl-d3) piperazine
- 1-(3-Bromophenyl)-4-(trideuteriomethyl) piperazine
- 1-(4-Bromophenyl)-4-(methyl-d3) piperazine
- 1-Chloro-2,4-difluoro-5-(methoxy-d3) benzene
- 3-Chloro-5-(methoxy-d3) aniline
- 4-Chloro-3-(methoxy-d3) aniline
- 2-Chloro-4-(methoxy-d3) phenol
- 3-Chloro-4-(methoxy-d3) phenol
- 3-Chloro-5-(trideuteriomethoxy) phenol
- 4-Chloro-3-(methoxy-d3) phenol
- 5-Chloro-1-(methyl-d3) -4-nitro-1h-pyrazole
- 4-Cyano-N-(methyl-d3) benzenesulfonamide
- Dimethylsulfoxide- d6
- 2-Fluoro-5-(methoxy-d3) benzonitrile
- 3-Fluoro-5-(methoxy-d3) phenol
- [2-fluoro-5-(methoxy-d3) phenyl]methanamine
- 2-(Methoxy-d3)benzoic acid
- 3-(Methoxy-d3)benzoic acid
- 4-(Methoxy-d3) benzoic acid
- 2-(Methoxy-d3) naphthalene-1-carbaldehyde
- 3-(Methoxy-D3)5-(trifluoromethoxy) phenol
- Methyl 5-bromo-2-(methoxy-d3)benzoate
- Methyl 4-(methoxy-d3)benzoate
- 1-(Methyl-d3) -4-nitro-1H-pyrazole
- 1-(Methyl-d3) -2-oxopyridine-4-carbonitrile
- 1-(Methyl-d3)-6-oxopyridine-3-carboxylic acid
- 4-[Methyl-d3-sulfamoyl]benzoic acid
- Methyl-d3 p-toluenesulfonate
- Methyl-d3 triflate
- Methyl-d3 (2,2,2-trifluoroethyl)amine hydrochloride
- Pyrimidines
- 4-Amino-5-(2-methylphenyl)-4h-1,2, 4-triazole-3-thiol
- 5-Aminopyrimidine-2- carboxylic acid
- 2-Amino-4-hydroxy-6- phenylpyrimidine
- 2-Acetamido-5- bromopyrimidine
- 3-Acetamido-6- chloropyridazine
- 3-Acetamido-4,5-dibromo- 1H-pyrazole
- 2- Acetamidopyrimidine
- 2-Acetamidopyrimidine-5-boronic acid, pinacol ester
- 3- Acetylaminopyrazole
- 2-Acetyl-3,5(6)-dimethylpyrazine, mixture of isomers
- 5-Acetyl-1,3-dimethyl-1,2,3,4-tetrahydropyrimidine-2, 4-dione
- 2-Acetyl-3- methylpyrazine
- 5-Acetyl-4-methylpyrimidin-2(1h)- one
- 2-[(Acetyloxy)methyl]-4-(2-amino-9H-purin-9-yl)butyl acetate
- 1-(4-Acetylphenyl)-4-bromo-3,5- dimethylpyrazole
- 2- ACETYLPYRAZINE
- 1-(1H-Pyrazol-4-yl) ethanone
- 1-Acetyl-1H-pyrazole-4-boronic acid, pinacol ester
- 2- Acetylpyrimidine
- 5- Acetylpyrimidine
- 1-(1H-Pyrrol-2-yl)ethanone 2-acetyl pyrrole
- 1-quinoxalin-2- ylethanone
- 1-Acetyl-4-tert- butoxycarbonylpiperazine
- 5-Acetyl-1H-pyrimidine- 2,4-dione
- Adenine phosphate
- Adenine hemisulfate
- Alfuzosina. ..
- Adenosine ..
- Alfuzosin ..
- 5-Allyl-4,6- dichloropyrimidine
- 5-Allyl-2,6-dimethyl- 4-pyrimidinol
- 5-Allyl-2-mercapto-6- methylpyrimidin-4(3h)-one
- 4-Allyl-1-mercapto-4h-[1,2,4]triazolo[4,3-a] quinazolin-5-one
- 2-Allyl-6-(methylthio)-1h-pyrazolo [3,4-d]pyrimidin-3(2h)-one
- 4-(Allyloxy)-2-chloro-6- methylpyrimidine
- 5-Allylsulfanyl-[1,3,4]thiadiazol- 2-ylamine
- 2-Amidinopyrimidine hydrochloride
- 4-Amino-5-aminomethyl-2- methylpyrimidine
- 3-Amino-6-(aminomethyl)-1,2,4-triazin-5(4h)-one dihydrochloride
- 6-Amino-5-benzylamino-1-butyl- 1h-pyrimidine-2,4-dione
- 2-Amino-5- chlorobenzo[d]thiazole
- 4-Amino-2-chloro-5- fluoropyrimidine
- 2-Amino-4-chloro-6- hydroxypyrimidine
- 3-Amino-6-chloroimidazo [1,2-b]pyridazine
- 4-Amino-2-chloro-5- iodopyrimidine
- 2-Amino-4-chloro-6- isopropylpyrimidine
- 2-Amino-5-chloro-3- methoxypyrazine
- 2-Amino-4-chloro-6- methoxypyrimidine
- 4-Amino-6-chloro-5- methoxypyrimidine
- 2-Amino-3-chloro-5- methylpyrazine
- 2-Amino-4-chloro-6- methylpyrimidine
- 4-Amino-2-chloro-5- methylpyrimidine
- 4-Amino-6-chloro-2- methylpyrimidine
- 5-Amino-2-chloro-4- methylpyrimidine
- 4-Amino-6-chloro-2- (methylthio)pyrimidine
- 5-Amino-3-(4-chlorophenyl) isoxazole
- 2-Amino-6- chloropurine
- Cladribine ..
- 4-Amino-6-chloro-5 -fluoropyrimidine
- 3-Amino-5- hydroxypyrazole
- 2-Amino-3- hydroxypyrazine
- 4-Amino-6-hydroxy-2- methylpyrimidine
- 5-Amino-1-(2-hydroxyethyl) pyrazole
- 7-Amino-furo[2,3-b]pyrazine-6-carboxylic acid ethyl ester
- 2-Amino-5- fluoropyrimidine
- 4-Amino-5-(2-fluorophenyl) -4h-1,2,4-triazole-3-thiol
- 2-Amino-5-(4-fluorobenzylthio)- 1,3,4-thiadiazole
- 2-Amino-3- ethynylpyrazine
- 4-(2-Aminoethyl)-5-methyl-1,2- dihydro-3h-pyrazol-3-one
- 2-Amino-3-(ethylamino) pyrazine
- 2-Amino-5- ethoxypyrazine
- 4-Amino-5-(2-ethoxyphenyl)-4h-1, 2,4-triazole-3-thiol
- 6-Amino-1,3- dimethyluracil
- 5-Amino-1,3-dimethylpyrimidine-2, 4(1H,3H)-dione
- 4-Amino-1,5-dimethylimidazole hydrochloride
- 2-Amino-3-(dimethylamino) pyrazine
- 2-Amino-4,6-dimethoxy-1 ,3,5-triazine
- 4-Amino-N-(2,6-dimethoxypyrimidin-4-yl) benzene-1-sulfonamide
- 2-Amino-5- hydroxypyrimidine
- 4-Amino-6- hydroxypyrimidine
- 3-Amino-1H-indazole-7-boronic acid pinacol ester
- 2-Amino-4-iodo-6- methoxypyrimidine
- 2-Amino-5-iodo-4- methoxypyrimidine
- 4-Amino-6-iodo-2- methylpyrimidine
- 2-Amino-3- iodopyrazine
- 2-Amino-5- iodopyrazine
- 3-Amino-4-iodo- 1h-pyrazole
- 4-Amino-3-iodo-1H-pyrazolo [3,4-d]pyrimidine
- 2-Amino-5- iodopyrimidine
- 4-Amino-5- iodopyrimidine
- 4-Amino-6-iodopyri midine
- 5-Amino-1-isopropyl-1h- indazole
- 4-Amino-1-isopropylpyrazole hydrochloride
- 4-Aminoisoxazole hydrochloride
- 3-Aminoisoxazolo[4,5-b] pyrazine
- 4-Amino-5-(2-methoxy-5-methyl-phenyl)- 4h-[1,2,4]triazole-3-thiol
- 2-Amino-4-methoxy-6- methylpyrimidine
- 2-Amino-4-methoxy-6-methyl-1,3,5- triazine
- 3-Amino-5-(2-methoxyphenyl)- 1H-pyrazole
- 4-Amino-5-(2-methoxy-phenyl)-4h- [1,2,4]triazole-3-thiol
- 2-([4-Amino-5-(2-methoxyphenyl)-4h-1,2,4-triazol-3-yl]sulfanyl)acetic acid
- 2-Amino-3- methoxypyrazine
- 2-Amino-5- methoxypyrazine
- 2-Amino-6- methoxypyrazine
- 3-Amino-6- methoxypyridazine
- 2-Amino-4- methoxypyrimidine
- 2-Amino-5- methoxypyrimidine
- 4-Amino-2- methoxypyrimidine
- 4-Amino-6- methoxypyrimidine
- 4-Amino-N-(5-methoxypyrimidin-2-yl) benzene-1-sulfonamide
- 2-Amino-8- methoxyquinazoline
- 6-Aminomethyl-1H -benzotriazole
- 6-((4-(Aminomethyl)benzyl)oxy)- 7H-purin-2-amine
- 2-Aminomethyl-3- chloropyrazine
- 4-Amino-5-(2-methyl-3-furyl)-4h- 1,2,4-triazole-3-thiol
- 5-Amino-3-methyl-isoxazole-4-carboxylic acid ethyl ester
- 2-(Aminomethyl)- 5-methylpyrazine
- 5-(Aminomethyl)-2-methylpyrimidin-4-amine dihydrochloride
- 6-Amino-1-methyl-5- nitrosouracil
- 5-Amino-3-methyl-1- phenylpyrazole
- 3-Amino-2-methyl-5-phenyl-3h-thieno [2,3-d]pyrimidin-4-one
- 4-Amino-6-(4-methyl-1-piperazinyl) pyrimidine
- 4-Amino-2-methyl-5-propylpyrazole-3-carboxamide hydrochloride
- 2- Aminomethylpyrazine
- 2-Amino-3- methylpyrazine
- 2-Amino-6- methylpyrazine
- 3- (Aminomethyl)pyrazole
- 3-Amino-5- methylpyrazole
- 4-Amino-1-methyl-1H-pyrazole- 3-carbonitrile
- 5-Amino-1-methyl-1H-pyrazole- 4-carbonitrile
- 5-Amino-1-methyl-1H-pyrazole-4- carboxylic acid
- 3-Amino-4-methyl- pyridazine
- 3-Amino-6- methylpyridazine
- 2-Amino-4- methylpyrimidine
- 4-(Aminomethyl) pyrimidine
- 4-Amino-5- methylpyrimidine
- 5-Amino-4- methylpyrimidine
- 2-Amino-4-methylpyrimidine-5- carbonitrile
- 2-Aminomethylpyrimidine hydrochloride
- (4-Amino-2-methyl-5-pyrimidinyl) methanol
- 2-[4-(6-Amino-2-methylpyrimidin-4-yl)piperazin-1-yl] ethanol
- 4-Amino-2-(methylsulfanyl)pyrimidine-5- carbonitrile
- 4-Amino-N-(5-methyl-1,3,4-thiadiazol-2-yl)benzene-1- sulfonamide
- 5-Amino-2-(methylthio)pyrimidine-4-carboxylic acid
- 4-Amino-2-(methylthio)-6- pyrimidinol
- 4-Amino-6- morpholinopyrimidine
- 2-Amino-3-morpholin-4- ylpyrazine
- 2-Amino-4-morpholin-4-yl- pyrimidine
- 3-Amino-5- nitroindazole
- 5-Amino-1-(4-nitrophenyl)-1h-pyrazole-4- carbonitrile
- 4-Amino-5-(2-nitro-phenyl)-4h-[1,2,4] triazole-3-thiol
- 2-Amino-5- nitropyrimidine
- 2-((2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy)ethyl L-valinate hydrochloride
- (S)-2-(4-(2-(2-Amino-4-oxo-4,7-dihydro-1h-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl)benzamido) pentanedioic acid
- 4-(2-(2-Amino-4-oxo-4,7-dihydro-1h-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl)benzoic acid
- 2-(4-Aminophenyl) oxazole
- 3-(4-Aminophenyl) pyrazole
- 3-Amino-4-phenyl-1H- pyrazole
- 3-Amino-5- phenylpyrazole
- 5-Amino-1-phenylpyrazole-4- carbonitrile
- 2-(5-Amino-3-phenyl-1h-pyrazol-1-yl) ethanol
- 5- Aminophthalazine
- 2-Amino-4-piperidino-6- methylpyrimidine
- 4-Amino-6- piperidinopyrimidine
- 6-Amino-1-propyl-3H- pyrimidine-2,4-dione
- 1-((4-Amino-2-propylpyrimidin-5-yl)methyl)-2-methylpyridin-1-ium chloride hydrochloride
- 2-Aminopurine riboside
- Adenosine 5'-monophosphate
- {[2-(6-Amino-9H-purin-9-yl)ethoxy]methyl}phosphonic acid
- 5-Aminopyrazine-2- carbonitrile
- 3-Aminopyrazine-2-carboxylic acid
- 6-Aminopyrazine-2-carboxylic acid
- 3-Amino-2(1H)- pyrazinone
- 4-Amino-1H- pyrazole
- 3-Amino-1H-pyrazole-4- carbonitrile
- 3-Aminopyrazole-4- carboxylic acid
- 4-Aminopyrazole hydrochloride
- 3-Aminopyrazolo[1,5-a] pyrimidine
- 4-Aminopyrazolo[3,4-d] pyrimidine
- 4-(3-Amino-1H-pyrazol-4-yl) benzonitrile
- 2-(3-Amino-1H-pyrazol-1-yl) ethanol
- 2-(4-Amino-1h-pyrazol-1-yl) ethanol
- 2-(4-Amino-1H-pyrazol-1-yl)ethanol hydrochloride
- 4- Aminopyridazine
- 3-Aminopyridazine hydrochloride
- 4- Aminopyrimidine
- 5- Aminopyrimidine
- 2-Amino-4- pyrimidinecarbonitrile
- 2-Aminopyrimidine-5- carbonitrile
- 2-Amino-5- pyrimidinecarboxaldehyde
- 2-Aminopyrimidine-4-carboxylic acid
- 2-Aminopyrimidine-5-carboxylic acid
- 2-Aminopyrimidine-5- carboxylic acid
- 4-Aminopyrimidine-5-carboxylic acid
- 6-Amino-pyrimidine-4-carboxylic acid
- 2-Amino-pyrimidine-5-carboxylic acid ethyl ester
- 2-Amino-5- pyrimidinesulfonamide
- 2-Aminopyrimidin- 4-ol
- 5-Aminopyrimidin-4- ol
- 2-(2-Aminopyrimidin-5-yl) benzaldehyde
- 4-Amino-N-(pyrimidin-2-yl)benzene-1- sulfonamide
- 3-(2-Aminopyrimidin-5-yl)benzoic acid
- 4-(2-Aminopyrimidin-5-yl)benzoic acid
- 1-(2-Aminopyrimidin-4-yl) ethanone
- 1-Amino-1H-pyrrole-2- carboxamide
- 4-Amino-7H-pyrrolo[2,3-d] pyrimidine
- 4-Amino-7h-pyrrolo[2,3-d]pyrimidine hydrogen sulfate
- 6-Aminopyrrolo[1,2-f][1,2,4] triazin-4(3h)-one
- 2- Aminoquinazoline
- 6- Aminoquinazoline
- 4-Aminoquinazoline-8- carbonitrile
- 4-Amino-2-sulfanylpyrimidine-5- carbonitrile
- 3-Amino-5-tert- butylpyrazole
- 5-Amino-1-tert-butyl-1h-pyrazole-4- carbonitrile
- (7-Amino-3-((tetrahydrofuran-2-yl)methyl)-3h-[1,2,3]triazolo[4,5-d]pyrimidin-5-yl) methanol
- 2-Amino-5,6,7,8-tetrahydropyrido-[4,3-d]-pyrimidine dihydrochloride
- 4-[(5-Amino-1,3,4-thiadiazol-2-yl)methoxy] benzenesulfonic acid
- 2-(5-Amino-[1,3,4]thiadiazol-2-ylsulfanyl)-n-(1,5-dimethyl-3-oxo- 2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)-acetamide
- 2-(5-Amino-[1,3,4]thiadiazol-2-ylsulfanyl)-n-(1,5-dimethyl- 3-oxo-2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)-propionamide
- 2-Amino-5-(2-thienyl)-1,3,4- thiadiazole
- 6-Amino-2-thiouracil monohydrate
- 3-Amino-5-p- Tolylpyrazole
- 3-Amino-1,2,4- triazole
- 4-Amino-1,2,4- triazole
- 3-(5-Amino-4h-[1,2,4]triazol-3-yl)- propan-1-ol
- N-Benzyl-2-chloroquinazolin-4- amine
- N-Benzyl-2-chloroquinazolin- 4-amine
- 7-Benzyl-4-chloro-7h-pyrrolo[2,3-d] pyrimidine
- 4-Benzyl-2-chloro- pyrimidine
- 6-Benzyl-4-chloro-6,7-dihydro-5h-pyrrolo [3,4-d]pyrimidine
- 1-Benzyl-4-bromopyrazole-5-boronic acid, pinacol ester
- 1-Benzyl-4-bromo-1H- pyrazole
- Quinolines
- 2-Methylquinoline-6- carbonitrile
- (2-Acetyl-1,2-dihydroisoquinolin-1-yl)acetic acid
- 3-Amino-7-bromoisoquinoline-4- carbonitrile
- 4-(Benzylsulfanyl) benzaldehyde
- 5- Acetamidoisoquinoline
- 1-Acetyl-8-allyl-1,2,3,4- tetrahydroquinoline
- 5-Acetyl-8-(benzyloxy)quinolin- 2(1h)-one
- 3-Acetyl-6-bromo-4- phenylquinolin-2(1h)-one
- 3-Acetyl-6-bromo-4-phenylquinolin- 2(1h)-one
- (2-Acetyl-6,7-dimethoxy-1,2,3,4-tetrahydro-isoquinoline-1-yl)-acetic acid
- 5-Acetyl-8-hydroxy-1h-quinolin- 2-one
- 3- Acetylquinoline
- 6- Acetylquinoline
- 3-Acetyl-1h-quinolin- 2-one
- 2-Acetyl-1,2,3,4-tetrahydro-3-isoquinolinecarboxylic acid
- 1-Acetyl-1,2,3,4-tetrahydroquinolin-7-amine hydrochloride
- 1-Acetyl-1,2,3,4-tetrahydro-6- quinolinecarbonitrile
- 1-Acetyl-1,2,3,4-tetrahydroquinoline-6- sulfonyl chloride
- 6-Acetyl-1,2,3,4- tetrahydroquinolin-2-one
- 1-(1-Acetyl-2,2,4,6-tetramethyl-1,2,3,4- tetrahydroquinolin-7-yl)ethanone
- 1-(1-Acetyl-2,2,4,8-tetramethyl-1,2,3,4- tetrahydroquinolin-7-yl)ethanone
- 3-Allyl-1,5- naphthyridine
- 4-Allyl-5-(2-phenylquinolin-4-yl)-4h-1,2,4- triazole-3-thiol
- 4-Amino-2,8-bis(trifluoromethyl)- quinoline
- 4-Amino-3- bromoisoquinoline
- 8-Amino-5- bromoisoquinoline
- 4-Amino-3-bromo-8- methoxyquinoline
- 2-Amino-6-bromo-3- methylquinoline
- 4-Amino-6-bromo-2- methylquinoline
- 4-Amino-6-bromo-7- methylquinoline
- 6-Bromo-4-chloro-7- methoxyquinoline
- 4-Amino-8-bromo-2- methylquinoline
- 4-Amino-3-bromo-6- nitroquinoline
- 2-Amino-6-bromopyrido[2,3-d]pyrimidin-4 (3H)-one
- 1-Chloroisoquinoline-6- carbonitrile
- 1-Chloroisoquinoline-7- carbonitrile
- 6-Chloroisoquinoline-1- carbonitrile
- 4-chloroisoquinoline hydrochloride
- 1-Chloroisoquinoline-5-sulfonyl chloride
- 1-Chloroisoquinolin- 7-ol
- 1-Chloroisoquinolin-7- ol
- 3-Chloroisoquinolin-4- ol
- 3-Chloroisoquinolin-5- ol
- 1-Chloroisoquinolin -6-ol
- 3-Chloroisoquinolin- 4-ol
- 3-Chloroisoquinolin- 5-ol
- 1-Chloro-4- methoxyisoquinoline
- 3-Chloroisoquinolin- 6-ol
- 1-Chloroisoquinolin- 6-ol
- Methyl 1-chloroisoquinoline-7-carboxylate
- Methyl 3-chloroisoquinoline-6-carboxylate
- Methyl 3-chloroisoquinoline-7-carboxylate
- 2-Methylquinoline-4- carbohydrazide
- 2-Methyl-4- quinolinecarbonitrile
- 4-Methylquinoline-2- carbonitrile
- 2-Methylquinoline-4- carboxamide
- Methyl quinoline-2-carboxylate
- 1,2,3,4-Tetrahydro-4- methylquinoline
- 1,2,3,4-Tetrahydro-1-methyl-8- quinolinol
- 1,2,3,4-Tetrahydro-3-oxo-2-quinoxalineacetic acid
- (8S)-5,6,7,8-Tetrahydro-8- quinolinamine
- (R)-5,6,7,8-Tetrahydroquinolin-8- amine
- 1,2,3,4-Tetrahydroquinolin-4- amine
- 5,6,7,8-Tetrahydroquinolin-3- amine
- (S)-5,6,7,8-Tetrahydroquinolin-8-amine dihydrochloride
- 1,2,3,4-Tetrahydroquinolin-7-amine hydrochloride
- IMPURITY CHEMICALS
- Abacavir EP Impurity E (Dihydro Abacavir)
- Artemether Related Compound B (-Artemether)
- 3-Piperazinyl-1,2-benzisothiazole. HCl
- Almotriptan Related Compound C (N-Desmethyl Almotriptan)
- Acetazolamide EP Impurity D
- Ascorbic Acid EP Impurity E (Oxalic Acid)
- 4-[2-(1,3-Dihydro-1,3-dioxo-2H-isoindol-2-yl)ethoxy]-3-oxobutanoic Acid Methyl Ester
- Anastrozole Monoacid
- 1-(3,4-Dimethoxyphenyl)-2- methylpropan-1-one
- (2RS)-2-(3,4-Dimethoxyphenyl)-3- methylbutanenitrile
- Abacavir EP Impurity B/ Abacavir Related Compound D (O-Pyrimidine Derivative Abacavir)
- Abacavir EP Impurity C/ Abacavir Related Compound A (Descyclopropyl Abacavir)
- Abacavir EP Impurity C/ Abacavir Related Compound A (Descyclopropyl Abacavir)
- Abacavir EP Impurity F/ O-t-Butyl Derivative Abacavir (Abacavir t-Butyl Ether)
- Abacavir Related Compound B
- Abacavir Related compound C (Abacavir 6- Chloro Analog)
- 2-cyano-3- morpholinoacrylamide
- Abacavir Cyclopropyl Diamino Purine Impurity
- (1S,4R)-4-Amino-2-cyclopenten-1-carboxylic Acid
- N-(2-Amino-4,6-dichloro-5-pyrimidinyl) formamide
- (1S,4R)-(4-Aminocyclopent-2-enyl)methanol hydrochloride
- rac 2-Azabicyclo[2.2.1]hept-5-en-3-one
- Abiraterone Methyl Ether
- Acarbose EP Impurity A
- Acarbose EP Impurity C
- Acarbose EP Impurity E
- Acarbose EP Impurity F
- Acarbose EP Impurity G
- Acebutolol EP Impurity A (Acebutolol Epoxypropoxy Impurity)
- Acebutolol EP Impurity B/ Acebutolol Related Compound B (Diacetolol)
- Acebutolol EP Impurity C
- Acebutolol EP Impurity D
- Acebutolol EP Impurity E (Acebutolol Desacetyl Impurity)
- Acebutolol EP Impurity J
- Aceclofenac EP Impurity A (Diclofenac)
- Aceclofenac EP Impurity B (Diclofenac Methyl Ester)
- Aceclofenac EP Impurity C (Diclofenac Ethyl Ester)
- Aceclofenac EP Impurity D (Aceclofenac Methyl Ester)
- Aceclofenac EP Impurity E (Aceclofenac Ethyl Ester)
- Aceclofenac EP Impurity F (Aceclofenac Benzyl Ester)
- Aceclofenac EP Impurity G (Acetic Aceclofenac)
- Aceclofenac Impurity EP H (Diacetic Aceclofenac)
- Aceclofenac EP Impurity I (Diclofenac EP Impurity A/ Diclofenac USP Related Compound A/ Diclofenac Amide)
- Acemetacin EP Impurity A (Indomethacin EP Impurity A/ Indomethacin Related Compound B/ Bezafibrate EP Impurity B)
- Acemetacin EP Impurity B (Indomethacin)
- Acesulfame Potassium Impurity A/ Acetoacetamide
- Acetazolamide EP Impurity A
- Acetazolamide EP Impurity B
- Acetazolamide EP Impurity C
- Acetazolamide EP Impurity E
- Acetazolamide EP Impurity G
- Acetylsalicylic Acid EP Impurity A (Butyl Parahydroxybenzoate EP Impurity A/ p-Salicylic Acid/ 4-Hydroxybenzoic acid)
- Acetylsalicylic Acid EP Impurity B (4-Hydroxyisophthalic Acid)
- Acetylsalicylic Acid EP Impurity C (Salicylic Acid/ Lamivudine EP Impurity C/ Mesalazine EP Impurity H/ Sulfasalazine EP Impurity H)
- Acetylsalicylic Acid EP Impurity D (Acetylsalicylsalicylic acid)
- Acetylsalicylic Acid EP Impurity E (Salsalate/ Salicylsalicylic acid)
- Acetylsalicylic Acid EP Impurity F (Acetylsalicylic Anhydride)
- Salicylaldehyde Azine
- Acyclovir EP Impurity A (O-Acetyl Aciclovir/ Valaciclovir EP Impurity I/ Valaciclovir Related Compund I)
- Acyclovir EP Impurity B (Guanine/ Valaciclovir EP Impurity A/ Valaciclovir Related Compound A)
- Acyclovir EP Impurity C (Aciclovir N7-Isomer)
- Acyclovir EP Impurity F (N2-Acetyl Aciclovir)
- Acyclovir EP Impurity G (Aciclovir N,O-Diacetate)
- Acyclovir Impurity H
- Acyclovir EP Impurity J
- Acyclovir EP Impurity L
- Acyclovir EP Impurity M (N,O-Diacetate Isoacyclovir)
- Acyclovir EP Impurity P
- Acyclovir L-Alaninate (Valaciclovir EP Impurity H/ Valaciclovir Related Compund H/ Didesmethyl Valacyclovir)
- Acyclovir L-Isoleucinate (Valaciclovir EP Impurity J)
- Acyclovir N-Methylene Dimer (Valaciclovir EP Impurity L)
- Acyclovir D-Valinate (Valaciclovir EP Impurity R/ D-Valacyclovir)
- 2-amino-8-hydroxy-9- ((2-hydroxyethoxy)methyl)-1,9-dihydro-6H-purin-6-one
- Adapalene EP Impurity B (Adapalene 3-Hydroxy Impurity)
- Adapalene EP Impurity C/ Adapalene Related Compound C (o-Adamantylanisole)
- Adrenaline EP Impurity B
- Adrenaline Impurity C/ Epinephrine Impurity C (Adrenalone)
- Adrenaline EP Impurity D
- Adrenaline Impurity E/ Epinephrine Impurity E
- Adrenaline EP Impurity F
- Epinephrine Sulfonic Acid
- Agomelatine Dimer Acetamide
- Agomelatine Desmethyl Impurity
- 7-Methoxy-1- naphthaleneacetonitrile
- Albendazole EP Impurity A (Albendazole Amine/ Desmethoxycarbonyl Albendazole)
- Albendazole EP Impurity C (Albendazole Sulfone)
- Albendazole EP Impurity D (Desmethoxycarbonyl Albendazole Sulfone)
- Albendazole EP Impurity E (Carbendazim)
- Albendazole EP Impurity F
- Albendazole EP Impurity I (Oxibendazole)
- Albendazole EP Impurity J
- Albendazole EP Impurity K
- Albendazole EP Impurity L
- O-Desmethyl-O-ethyl Albendazole
- Alfuzosin EP Impurity A/ Alfuzosin Related Compund A (Alfuzosin Tetradehydro Impurity)
- Alfuzosin EP Impurity B (Prazosin EP Impurity A/ Terazosin EP Impurity A/ Doxazosin EP Impurity F/ Doxazosin Related Compund C)
- Alfuzosin EP Impurity C
- Alfuzosin EP Impurity D / Alfuzosin Related Compound D (Alfuzosin Aminopropyl Impurity)
- Alfuzosin EP Impurity E
- Alfuzosin EP Impurity F
- Alfuzosin EP Impurity G (Dimer)
- (2-Chloro-6,7-dimethoxyquinazoline-4-yl)- (3,4-dimethoxyphenyl)-amine
- Alimemazine EP Impurity C (Promethazine EP Impurity A/ Phenothiazine)
- Allopurinol EP impurity A/ Allopurinol Related Compound A
- Allopurinol EP impurity B/ Allopurinol Related Compound B
- Allopurinol EP impurity C/ Allopurinol Related Compound C
- Allopurinol EP impurity D/ Allopurinol Related Compound D
- Allopurinol EP Impurity E/ Allopurinol Related Compound E
- Almotriptan Related Compound A (N-Hydroxymethyl Almotriptan)
- Almotriptan Related Compound B (Almotriptan Didesmethyl Impurity)
- Almotriptan Related Compound D (Almotriptan N-Oxide)
- 6-Chloro-3- methyluracil
- Alogliptin 6-Chloro Impurity
- 2-Cyanobenzyl bromide
- Alogliptin S-Isomer
- 4,6-Dichloro-2- (methylthio)pyrimidine
- 4,6-Dichloro-2-(methylsulfonyl) pyrimidine
- 6-Chloro-2-(methylthio) pyrimidin-4-ol
- Altizide EP Impurity A (Hydrochlorothiazide EP Impurity B/ Hydrochlorothiazide Related Compound A/ Benzothiadiazine Related Compound A/ Salamide )
- Amantadine EP Impurity B (N-Acetyl Amantadine)
- Ambrisentan Hydroxy Acid Impurity
- Ambroxol EP Impurity A (Bromhexine EP Impurity A)
- Ambroxol EP Impurity B
- Ambroxol EP Impurity C (Dehydro Ambroxol)
- Ambroxol EP Impurity D (cis-Ambroxol HCl/ Acebrophylline Impurity D)
- Ambroxol EP Impurity E (Bromhexine EP Impurity B)
- 2-Amino-3-benzoyl--oxo-benzeneacetic Acid
- 7-Benzoylindoline- 2,3-dione
- 6-Aminocaproic Acid Dimer
- Amiodarone EP Impurity B/ Amiodarone Related Compound B (Desethyl Amiodarone)
- Amiodarone EP Impurity D/ Amiodarone Related Compound D
- Amiodarone EP Impurity E/ Amiodarone Related Compound E
- Amiodarone EP Impurity F/ Amiodarone Related Compound F
- Amiodarone EP Impurity H/ Amiodarone Related Compound H
- Amiodarone Methoxy Impurity
- 2-butyl benzofuran
- (2S)-2-Aminomethyl-1- ethylpyrrolidine
- 2,4- dimethylaniline
- Amlodipine Impurity-A (EP) /Amlodipine Related Compound D (Phthaloyl Amlodipine)
- Amlodipine Impurity-B (EP) (Methyl amino phthaloyl Amlodipinee)
- Amlodipine EP Impurity C (Bis(aminoethoxy) Amlodipine)
- Amlodipine Impurity-D (EP) /Amlodipine Related Compound A (Aromatic Amlodipine/ Dehydro Amlodipine)
- Amlodipine Impurity-E (EP) /Amlodipine Related Compound E (Amlodipine Diethyl Ester)
- Amlodipine Impurity-G (EP) /Amlodipine Related Compound C
- Amlodipine Impurity-H (EP) ( 2-Carboxybenzoyl Amlodipine)
- Amlodipine 4-Chloro Analogue (Para chloro Amlodipine)
- Amlodipine Dimethoxy intermediate Impurity
- Amlodipine Aspartic Acid Impurity
- Amlodipine Lactose Adduct (Amlodipine N-Lactoside)
- Amlodipine Deschloro Impurity
- Methyl benzenesulfonate
- ethyl benzenesulfonate
- Isopropyl Benzenesulfonate
- 4-(2-Phthalimidoethoxy)acetoacetic Acid Ethyl Ester
- Ethyl 3-aminocrotonate
- 5-ethyl 7-methyl 6-(2-chlorophenyl)-8-methyl-3,4,6,7-tetrahydro-2H-benzo[b][1,4]oxazine-5,7-dicarboxylate
- 4-(2-Chlorophenyl)-2,6-bis[[2-(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)ethoxy]methyl]-1,4-dihydro-3,5-pyridinedicarboxylic Acid Ethyl Methyl Ester
- N-(2-Hydroxyethyl) phthalimide
- Amodiaquine Dichloro Impurity (Hydroxychloroquine EP Impurity G/ ChloroquineRelated Compound A)
- N-(4-Hydroxyphenyl) acetamide
- Amoxicillin EP Impurity B/ Amoxicillin Related Compound B (L-Amoxicillin)
- Amoxicillin EP Impurity C/ Amoxicillin Related Compound C (Amoxicillin Diketopiperazine)
- Amoxicillin EP Impurity G/ Amoxicillin Related Compound G
- Amoxicillin EP Impurity H/ Amoxicillin Related Compound H
- Amoxicillin EP Impurity I/ Amoxicillin Related Compound I (Cefadroxil EP Impurity A/ Cefoperazone Impurity 3)
- Amoxicillin EP Impurity J/ Amoxicillin Related Compound J (Amoxicillin Dimer)
- Amoxicillin EP Impurity K
- Amoxicillin EP Impurity L (6-APA Amoxicillin Amide)
- Amoxicillin Trimer
- N-Pivaloyl Amoxicillin
- Ampicillin EP Impurity A (Amoxicillin EP Impurity A
- 2-Methyl-pyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 2-(tert- Butoxycarbonyloxyimino)phenylacetonitrile
- (S)-1-(2-Chloroacetyl) pyrrolidine-2-carbonitrile
- Anagrelide Related Compound A
- Anagrelide Related Compound B
- Anagrelide Related Compound C (Anagrelide open ring ethyl ester)
- Anagrelide open ring methyl ester
- 2,3-Dichloro-6-nitrobenzyl Alcohol
- Anastrozole EP Impurity A
- Anastrozole EP Impurity F (Lisinopril EP Impurity B, Tizanidine EP Impurity I, Sultamicillin EP Impurity B)
- Anastrozole EP Impurity H/ Anastrozole Related Compound A
- 2,6-di-tert-butyl-4-(2- hydroxyethyl)phenol
- 1-(4-amino phenyl)-3-morpholino-5,6-dihydropyridin-2 (1H)-one
- Ethyl 2-chloro-2-(2 -(4-methoxyphenyl)hydrazono)acetate chloro ester
- 1-(4-iodophenyl)-3-morpholino-5,6- dihydropyridin-2(1H)-one
- Apremilast EP Impurity B (Apremilast 3-Acetamido phthalic acid Impurity)
- Apremilast EP Impurity H (Apremilast Des Acetamidophthalic acid Impurity)
- Apremilast 2-Acetamido Benzoic Acid Imurity
- [1(S)-Phenylethoxy]-Aprepitant/ Aprepitant S,R,S-Isomer
- Aripiprazole EP Impurity A/ Aripiprazole Dihydroquinolinone Impurity
- Aripiprazole EP Impurity B/ Aripiprazole Related Compound C
- Aripiprazole EP Impurity C (Aripiprazole 3-Deschloro Impurity)
- Aripiprazole EP Impurity D (Aripiprazole 2-Deschloro Impurity)
- Aripiprazole EP Impurity E/ Aripiprazole Related Compound G (Dehydro Aripiprazole)
- Aripiprazole EP Impurity F/ Aripiprazole Related Compound F (Aripiprazole N-Oxide)
- Aripiprazole EP Impurity G (Aripiprazole Dimer)
- Aripiprazole Related Compound H
- Aripiprazole Hydroxybutylpiperazine Impurity
- Aripiprazole Related Compound B (Aripiprazole Hydroxybutoxyquinoline Impurity)
- Aripiprazole Diquinoline Butanediol Impurity
- Aripiprazole N,N-Dioxide
- Aripiprazole Impurity-1
- Aripiprazole Impurity 2
- Aripiprazole Bromo Impurity/ 7-(4-bromobutoxy)quinolin-2(1H)-one
- Aripiprazole Quinolinone Impurity
- Aripiprazole Chlorophenyl piperazine Impurity
- Aripiprazole 3,4-Dichloro Impurity
- Aripiprazole Bromobutoxyquinoline Impurity
- Aripiprazole 3,4-Dichlorophenyl Piperazine
- Aripiprazole Butanoic Acid Impurity
- Aripiprazole Desethylene Impurity
- Aripiprazole Iodobutoxyquinoline Impurity
- Aripiprazole Methoxybutoxyquinoline Impurity
- Aripiprazole N-Bromobutyl Impurity
- Aripiprazole N-Isomer
- Aripiprazole Quinolinediol Impurity
- Chlorobutoxy Dihydroquinolinone
- Di-[2-(2,3-dichlorophenyl) aminoethyl]amine
- 1,4- Dibromobutane
- Thiobenzhyd rol
- Artemether Ph. Int. Impurity A (Tricabonly compound)
- Artemether Related Compound A (Artemisinin Dihydro Impurity)
- Artesunate IP Impurity B (Artemisinin)
- Ascorbic Acid EP Impurity C
- Ascorbic Acid EP Impurity D
- Asenapine Tetradehydro Impurity
- Asenapine N-Desmethyl Impurity
- Asenapine N-Oxide
- rel-(3aR,12bR)-11-Chloro-2,3,3a,12b-tetrahydro-2-methyl-1H-dibenz [2,3:6,7]oxepino[4,5-c]pyrrol-1-one
- N-Desmethyl Asenapine
- Aspartame EP Impurity A/ Aspartame Related Compound A
- Aspartame EP Impurity B/ Aspartame Related Compound B (Aspartame Acid)
- Aspartame EP Impurity C (Nateglinide EP Impurity D)
- (2S,3S)-3-Boc-amino-1,2-epoxy-4- phenylbutane
- (2R,3S)-3-(tert-Boc)amino-1,2-epoxy-4- phenylbutane
- Atenolol EP Impurity A
- Atenolol EP Impurity B/ Atenolol Related Compound A
- Atenolol EP Impurity C
- 7-Hydroxy-3,4-dihydro quinolin-2(1H)-one
- 1-(2,3-Dichlorophenyl) piperazine hydrochloride
- 7-[4-[4-(2-Chlorophenyl)-1-piperazinyl]butoxy] -3,4-dihydro-2(1H)-quinolinone
- 7-[4-[4-(3-Chlorophenyl)-1-piperazinyl]butoxy]- 3,4-dihydro-2(1H)-quinolinone
- 7-(4-(4-(2,3-dichlorophenyl) piperazin-1-yl)butoxy)-3,4-quinolin-2(1H)-one
- 4-(2,3-Dichlorophenyl)-1-[4-(2-oxo-1,2,3,4-tetrahydro quinolin-7-yloxy)butyl] piperazine-1-oxide
- 1,1'-(Ethane-1,1-diyl)bis(2,3-dichloro-4{4-[3,4-dihydro quin olin-2(1H)-one-7-yloxy butyl] piperazin-1-yl}benzene)(U/D)
- 7-(4-Hydroxy-butoxy)-3,4- dihydroquinolin-2(1H)-one
- 7-(4-Bromobutoxy)-3,4- dihydroquinolin-2-one
- 7,7'-[Butane-1,4,diylbis(oxy) ]bis[3,4-dihydroquinolin-2 (1H) -one
- 1-(3,4-Dichloro phenyl) piperazine.HCl
- 4-Hydroxy benzoic acid
- 4-Hydroxy Isopthalic acid
- 2-((2-Acetoxybenzoyl)oxy) benzoic acid
- 2-((2-Hydroxybenzoyl)oxy) benzoic acid
- 2-Acetoxybenzoic anhydride
- N-[3-[(4-Amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl]furan-2-carboxamide hydrochloride
- 4-Amino-2-chloro-6,7- dimethoxyquinazoline
- N-(3-(4-Amino-6,7-dimethoxyquinazolin-2-ylamino)propyl)-N-methyltetrahydrofuran- 2-carboxamide
- N-(4-Amino-6,7-dimethoxyquinazolin-2-yl)-N-methylpropane-1,3-diamine hydrochloride
- N-[3-[(4-Amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl]formamide hydrochloride
- N-Methyl-3-phenoxy-3-phenylpropan-1-amine hydrochloride
- (R)-(+)-N-Methyl-3-phenyl- 3-hydroxypropylaMine
- (3S)-N-Methyl-3-(2-methylphenoxy)-3-phenylpropan-1-amine hydrochloride
- (3R)-N-Methyl-3-(3-methylphenoxy)-3-phenylpropan-1-amine hydrochloride
- (3R)-N-Methyl-3-(4-methylphenoxy)-3-phenylpropan-1-amine hydrochloride
- L-(+)-Mandelic Acid
- 3,5-Bis(1-cyano-1-methylethyl)benzyl Bromide
- 4-Methylbenzenesulfonic acid monohydrate
- 2-[4-[(2RS)-2,3-dihydroxypropoxy] phenyl]acetamide
- 2-[4-[[(2RS)-oxiran-2-yl]methoxy]phenyl] acetamide
- 2-[4-[(2RS)-2-Hydroxy-3-[(1-methylethyl)amino]propoxy]phenyl]acetic acid
- 2-(4-(3-(Ethylamino)-2-hydroxypropoxy) phenyl)acetamide
- 4-(6-bis(2-hydroxyethyl)amino-3-methyl benzimidazoyl(2)butyric acid
- 4-(1-Methyl-5-morpholino-1H -benzimidazol-2-yl)butanoic acid
- 4-(5-(Bis(2-hydroxyethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acid ethyl ester
- 4-(5-((2-Chloro4-(5-((2-Chloroethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acidethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acid
- 5-[(2-Chloroethyl)(2-hydroxyethyl)amino]-1-methyl-1H-benzimidazole-2-butanoic acid HCl
- Ethyl 5-[Bis(2-chloroethyl)amino]-1-methyl-1H-benzimidazole-2-butanoate
- 4-[5-[Bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid isopropyl ester
- N-[(1S)-2-Amino-2-oxo-1-(phenylmethyl)ethyl]-2- pyrazinecarboxamide
- (S)-3-Phenyl-2-[(pyrazine-2-carbonyl) amino]propionic Acid
- (S)-N-(1-(isopentylaMino)-1-oxo-3-phenylpropan-2-yl) pyrazine-2-carboxaMide
- (2S)-N-[(1R)-1-hydroxy-3-methylbutyl]-3-phenyl-2-(pyrazin-2-ylformamido) propanamide
- (2S)-N-[(1S)-1-hydroxy-3-methylbutyl]-3-phenyl-2-(pyrazin-2-ylformamido)propanamide;(S)-Hydroxy Des(boric Acid) Bortezomib
- (RS)-1-(4-Hydroxymethylphenoxy)-3- isopropylaminopropan-2-ol
- (2RS)-3-[4-((2-Isopropoxyethoxy)methyl) phenoxy]-1,2-propanediol
- 4-[((2RS)-2-Hydroxy-3-(isopropylamino)-propyl)oxy]benzaldehyde hydrochloride
- 4-[(2-Isopropoxyethoxy)methyl] phenol
- (2RS)-1-(Isopropylamino)-3- (4-methylphenoxy)propan-2-ol
- 4- Hydroxybenzaldehyde
- 4-[[3-(1-Methylethyl)-2-oxo-5-oxazolidinyl]methoxy] benzaldehyde
- 4-(2,3-Epoxypropoxy) carbazole
- 2-(2-Methoxyphenoxy)ethylamine hydrochloride
- 1-(p-Chloro--phenylbenzyl) piperazine
- 4-[(4-Chlorophenyl)phenylmethyl]-1-piperazineacetic Acid
- 2-[2-[4-[(2-Chlorophenyl)phenylmethyl]-1-piperazinyl]ethoxy]acetic Acid Dihydrochloride
- 1,4-bis((4-chlorophenyl)(phenyl)methyl) piperazine
- RS)-2-[2-[2-[4-[(4-Chlorophenyl)phenylmethyl]piperazin-1-yl]ethoxy] ethoxy]acetic acid
- 2-[2-[4-(Diphenylmethyl)-1-piperazinyl]ethoxy]acetic Acid Hydrochloride
- 4-[(R)-(4-Chlorophenyl)phenylmethyl]-1-piperazine ethanol Hydrochloride
- [2-[4-(4-Chlorophenyl)phenylmethyl]-1-oxido-1-piperazinyl]ethoxy]acetic Acid
- (RS)-2-[2-[4-[(4-Chlorophenyl)phenylmethyl]piperazin-1-yl]ethoxy]acetic acid ethyl ester dihydrochloride
- (RS)-2-[2-[4-[(4-Chlorophenyl)phenylmethyl]piperazin-1-yl]ethoxy]acetic acid methyl ester dihydrochloride
- 3-(2-Chloro-10H-phenothiazin-10-yl)-N,N-dimethylpropan-1-amine S-oxide
- 2-Chloro-10-[3-[[3-(dimethylamino)propyl]methylamino] propyl]phenothiazine
- N,N-Dimethyl-10H-phenothiazine-10-propanamine hydrochloride
- 2-Chloro-N-methyl-10H-phenothiazine-10-propanamine hydrochloride
- 2-Chloro-10H- phenothiazine
- (R)-1-(Naphthalen-1-yl)ethanamine HCl
- (R)-N-Benzyl-1-(naphthalen-1-yl)ethanamine HCl
- (R)-N-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)-N-(3-(3-(trifluoromethyl)phenyl) propyl)propan-1-amine
- R)-N-(1-(Naphthalen-1-yl)ethyl)-3-m-tolylpropan-1- amine hydrochloride
- (R)-N-(1-(Naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl) phenyl)propan-1-amine N-oxide
- 3-(3-Trifluoromethylphenyl)propyl Methanesulfonate
- 3-[3-(Trifluoromethyl) phenyl]propan-1-ol
- 7-chloro-1-cyclopropyl-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
- 1-Cyclopropyl-6-fluoro-7-(piperazin-1-yl) quinolin-4(1H)-one
- 1-cyclopropyl-6-fluoro-7-(4-formylpiperazin-1-yl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
- 11,11'-(Piperazine-1,4-diyl)bis(8-chloro- 5H-dibenzo[b,e][1,4]diazepine)
- 8-Chloro-11-(1-piperazinyl)-5H-dibenzo[b,e][1,4]diazepine Hydrochloride
- 2-Chloro-N-(2-chloroethyl)ethanamine Hydrochloride
- 3-(2-Chloroethyl)octahydro-2-hydroxy-1,3,6,2-oxadiazaphosphonine 2-Oxide
- 3-Aminopropyl dihydrogen phosphate
- 3-[[2-[(2-Chloroethyl)amino]ethyl]amino]propyl Monophosphate Dihydrochloride
- 3-Aminopropan- 1-ol
- 1-(2,6-Dichlorophenyl)-1,3- dihydro-2H-indol-2-one
- 2-[(2,6-Dichlorophenyl)amino] benzaldehyde
- [2-[(2,6-Dichlorophenyl)amino]phenyl] methanol
- 1,3-Dihydro-2H-indol-2- one
- N-(4-Chlorophenyl)-2-(2,6-dichlorophenyl) acetamide
- Ethyl [2-[(2,6-dichlorophenyl)amino]phenyl]acetate
- Methyl [2-[(2,6-dichlorophenyl)amino]phenyl]acetate
- O-(2,6-Dichlorophenylamino)-phenylglyoxalic acid
- N-(2,6-Dichlorophenyl)-N-phenyl-N- chloro
- 5-Chloroacetyl-6-chloro-1,3-dihydro- 2H-indole-2-one
- 6-Methyl-2-(4-methylphenyl)-imidazo[1,2-a]pyridine-3- carboxaldehyde
- 4-Methyl-N-(5-methyl-2-pyridinyl) benzamide
- 5,5-[[2-(3,4-Dimethoxyphenyl)ethyl]imino]bis[2-(3,4-dimethoxyphenyl)-2-(1-methylethyl) pentane nitrile] hydrochloride
- 2RS)-2-(3,4-Dimethoxyphenyl)-5-[[2-(3,4-dimethoxyphenyl)ethyl]amino]-2-(1-methylethyl)pentanenitrile monohydrochloride
- (2RS)-2-(3,4-Dimethoxyphenyl)-2-[2-[[2-(3,4-dimethoxyphenyl)ethyl] (methyl)amino]ethyl]-3-methylbutanenitrile monohydrochloride
- 3,4- Dimethoxybenzaldehyde
- (2RS)-2-(3,4-Dimethoxyphenyl)-5-(methyl amino)-2-(1-methylethyl) pentanenitrile monohydrochloride
- (3,4-Dimethoxyphenyl) methanol
- 3-Chloro-N-[2-(3,4-dimethoxyphenyl)ethyl]-N-methylpropan- 1-amine
- 2-(3,4-Dimethoxyphenyl)-N,N-dimethylethanamine Hydrochloride
- 2-(3,4-Dimethoxyphenyl)-N-methyl ethanamine monohydrochloride
- N,N-Bis[2-(3,4-dimethoxyphenyl)ethyl]-N,N-dimethylpropane-1,3-diamine dihydrochloride
- 1-(2-Ethoxycarbonylbenzofuran-5-yl) piperazine
- Indoles & Oxindoles
- 1-Acetyl-5-bromo-1h-indole-2, 3-dione
- 1-Acetyl- 5-nitroindoline
- 4-Acetyl- 7-azaindole
- 1-Acetyl-5-bromo-4-chloro-1h-indol-3-yl acetate
- 1-Acetyl-5-amino-2,3-dihydro- 1H-indole
- 1- Acetylindoline
- (3-Acetyl-1h-indol-1-yl)acetic acid
- N-Acetyl-DL- Tryptophan
- 1-Acetyl- 3-bromoindole
- 1-Acetyl-4-chloro- 1H-indazole
- 1- Acetylisatin
- Chemical Name: ([(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]amino)(phenyl)acetic acid
- 2-(3-Acetyl-1h-indol-1-yl)propanoic acid
- 3-[(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]propanoic acid
- 3-(3-Acetyl-1h-indol-1-yl)propanoic acid
- N-Acetyl-3- hydroxyindole
- 2-Acetamido-3-(7-chloro-1h-indol-3-yl)propanoic acid
- 5- Acetyloxindole
- N-Acetylindole- 3-carboxaldehyde
- 5-Acetyl-1h-indole-2-carboxylic acid
- 1-Acetyl-5-fluoro-4-iodo- 7-azaindole
- (R)-N-Acetyl- 6-chloro-trp-ome
- N-Acetyl-5-bromo- 3-hydroxyindole
- (R)-N-Acetyl-5-fluoro-trp- ome
- (R)-N-Acetyl-6-fluoro-trp- ome
- 1-Acetyl-5-bromoindolin- 3-one
- 7- Acetoxyindole
- 1-Acetyl- 5-bromoindole
- 3- Acetoxyindole
- 1-Acetyl-2,3-dihydro-1H-indole-5-sulfonyl chloride
- 5- Acetylindole
- N-Acetyl- 3-hydroxyindole
- N1-(1-Acetyl-2,3-dihydro-1h-indol-5-yl)-4-chloro- 3-nitrobenzamide
- 1- Acetylindole
- (1-Acetyl-2,3-dihydro-1h-indol-5-yl)boronic acid
- 2-(4-Acetylphenyl)-1h-isoindole-1, 3(2h)-dione
- (3R)-2-Acetyl-1-phenyl-2,3,4,9-tetrahydro-1h-beta-carboline-3-carboxylic acid
- 1-Acetyl-5-bromo-7- nitroindoline
- 1-Acetyl-6-fluoro-1h-indol-3-yl acetate
- 1-Acetyl-5-bromo-4-chloro- pseudoindoxyl
- Acetic acid, 2-[(1,3-dihydro-1,3-dioxo-2h-isoindol-2-yl)oxy]-, 2,5-dioxo-1-pyrrolidinyl ester
- (R)-N-Acetyl- 5-fluoro-trp-ome
- N-Acetyl- 5-methyl-dl-tryptophan
- 4- Acetoxyindole
- (3-Acetyl-2-methyl-indol-1-yl)-acetic acid
- 7-Acetylamino-4-fluoro-1h- indole
- 1-Acetyl-4-bromo- 1H-indazole
- 1-Acetyl-5-nitro- 1H-indazole
- N-Acetylindoline-2-carboxylic acid
- 3-Acetylamino-1h-indole-2-carboxylic acid
- 1-Acetylindoline-5-carboxylic acid
- (3-Acetyl-7-ethyl-1h-indol-1-yl)acetic acid
- 1-Acetyl-7-amino-2,3-dihydro- 1h-indole
- (2R)-1-Acetyl-2,3-dihydro-1h-indole-2-carboxylic acid
- N-Acetyl-D- Tryptophan
- 1-Acetyl- 3-indolinone
- 3- Acetylindole
- 1-Acetyl- 3-acetoxyindole
- N-Acetyl-5-methoxy- l-tryptophan
- 4-([(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]amino)butanoic acid
- 2-([(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]amino)-3-phenylpropanoic acid
- 1-Acetyl-1h-indole- 6-carbonitrile
- 2-(4-Acetylphenyl)-1h-isoindole-1,3(2h)- dione
- 1-Acetylindolin- 2-one
- (R)-N-Acetyl- 6-fluoro-trp-ome
- (3R)-2-Acetyl-2,3,4,9-tetrahydro-1h-beta-carboline-3-carboxylic acid
- 1-Acetyl-6-chloro- 1H-indazole
- 3-Acetyl-4-chloro- 7-azaindole
- 2- Acetylindole
- (R)-N-Acetyl-5-chloro-trp- ome
- 1-Acetyl-2-methylindolin-5-amine hydrochloride
- 1-Acetyl-3-bromo- 4-chloroindole
- 1-Acetyl-5-iodo-1h-indol-3-yl acetate
- 3-Acetyl-7(1H) -azaindole
- (S)-1-Acetyl-2,3-dihydro-1h-indole-2-carboxylic acid
- 3-Acetyl-1h-indole- 6-carbonitrile
- 5-Acetyl-3-methyl-1h-indole-2-carboxylic acid
- 1-Acetyl-2-methylindoline-5-sulphonyl chloride
- 1-Acetylindole-2-carboxylic acid
- 1-Acetyl-3, 3-dimethylindoline
- 1-Acetyl-2-methyl- 5-nitroindoline
- 1-Acetyl- 6-aminoindoline
- 2-[2-(Acetylamino)phenyl]-n-[2-(1h-indol-3-yl)ethyl]- 2-oxoacetamide
- 2-Hydroxy benzoic acid
- Nitro Compounds
- Sulfonamides
- 2- Cyanobenzenesulfonamide
- 2-Amino-6-chloro-n-(2,4-dimethylphenyl)benzene sulfonamide
- 1-(2-Cyanophenyl)-N- pentylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- cyclohexylmethanesulfonamide
- 4,5-Dimethyl-4H-1,2,4-triazole-3- thiol
- 4-Acetylbenzenesulfonyl chloride
- Alpha -(p-toluenesulfonyl)-4- fluorobenzylisonitrile
- 3- Cyanobenzenesulfonamide
- 2-(1,3-Dioxoisoindol-2-yl)-N,N- diethylethanesulfonamide
- 4-Amino-N-cyclopropylbenzene sulfonamide
- 2-(Dimethylsulfamoyl)pyrazole-3-boronic acid
- 2-Allyloxy-1,1,2,2-tetrafluoroethane sulfonyl fluoride
- N-Ethyl-4- nitrobenzenesulfonamide
- Ethyl 5-(ethylsulfonyl)pyridine-2-carboxylate
- Difluoromethanesulfonyl chloride
- 2,3-Dioxo-1H-indole-5-sulfonyl chloride
- 1-(Ethanesulfonyl)-4-nitro-2 -(trifluoromethyl)benzene
- Alpha - toluenesulfonylchloride
- 4-Cyanobenzene-1- sulfonamide
- N,N-Diethyl 5-bromo-2-methoxybenzenesulfonamide
- N-Cyclohexyl 3-boronobenzenesulfonamide
- 3,5-Difluorobenzenesulfonyl chloride
- 3-(5-Diethylsulfamoyl-1-ethyl-1h-benzoimidazol-2-yl)-propionic acid
- 1,2-Dimethyl-1h-imidazole-5-sulphonyl chloride
- N-(2-Acetylphenyl) benzene sulfonamide
- 2,5-Dimethoxybenzenesulfonyl chloride
- N,N-Dimethyl 3-bromo-5-methylbenzenesulfonamide
- N,4-Dimethyl-n-(oxiran-2-ylmethyl) benzenesulfonamide.
- N-Allyl-4-aminobenzene sulfonamide
- 3-(1,1-Dioxido-2-isothiazolidinyl)phenylboronic acid
- (Dimethylsulfamoyl) methylamine
- 2,6-Dimethylmorpholine-4-sulfonyl chloride
- N-Cyclopropyl 3-nitrobenzenesulfonamide
- N-(4-Acetylphenyl) methane sulfonamide
- N-(3-Acetylphenyl) benzene sulfonamide
- 4-Amino -n- utylbenzene sulfonamide
- 4-cyano-N- methylbenzenesulfonamide
- Dansyl fluoride
- 3-(2,3-Dihydro-indole-1-sulfonyl)-benzoic acid
- 4-(Dimethylamino)- 3-nitrobenzenesulfonamide
- N-(3-Acetylphenyl) methane sulfonamide
- 3-(3,4-Dihydro-2h-quinoline-1-sulfonyl)-4-methoxy-benzoic acid
- N,N-Dimethyl 4-fluorobenzenesulfonamide
- N-Cyclohexyl-2- nitrobenzenesulfonamide
- Cyclopropylmethane sulfonamide
- N-(1,1-Dioxidotetrahydrothien-3-yl)-n-propylamine hydrochloride
- 3,4- Difluorobenzenesulfonamide
- 4-(Difluoro-methanesulfonyl)-benzoic acid
- 3,4-Dimethyl-(4H)1,4-benzothiazine-1,1- dioxide
- 2,4-Dioxo-1,2,3,4-tetrahydropyrimidine-5-sulfonyl chloride
- 1-(Ethanesulfonyl)-2-methoxy-4- nitrobenzene
- (3-Cyanophenyl)methanesulfonyl chloride
- 4-Acetamidobenzene sulfonamide
- 4-Acetamido-3-chlorobenzenesulfonyl chloride
- 2-Acetamido-5-ethylbenzenesulfonyl chloride
- 5-Acetonyl-2-methoxybenzene sulfonamide
- 4-(Acetylamino)benzenesulfonyl fluoride
- 5-(Acetylamino)-2-methoxybenzenesulfonyl chloride
- 2-(Acetylamino)-5-methylbenzenesulfonyl chloride
- 4-Acetylamino-2-methyl-benzenesulfonyl chloride
- 4-Acetylbenzene sulfonamide
- 3-Acetyl- 5-chlorothiophene- 2-sulfonamide
- 1-Acetyl-2,3-dihydro -1H-indole -5-sulfonyl chloride
- 4-Acetyl-n- (4-fluorobenzyl) benzenesulfonamide
- 4-Acetyl-n -(2-furylmethyl)benzene sulfonamide
- 4-Acetyl-n-(2-furylmethyl) benzenesulfonamide
- N-(5-Acetyl-2-hydroxy-phenyl)- methanesulfonamide
- 3-(4-Acetyl-piperazine-1-sulfonyl)-benzoic acid
- 4-(4-Acetylpiperazinosulfonyl) Bromobenzene
- Acetyl sulfamethoxazole
- 1-Adamantaneethyl sulfonamide
- 4-(Adamantan-1-yl)phenyl trifluoromethane sulfonate
- N-1-Adamantyl-4-aminobenzene sulfonamide
- N-[2-(1-Adamantyloxy)propyl]methane sulfonamide
- alfa-(4-Toluenesulfonyl)- 3-methoxybenzylisocyanid
- alfa-(4-Toluenesulfonyl)-4- methoxybenzylisocyanide
- 4-[(Allylamino) sulfonyl]benzoic acid
- N-Allyl-4-isothiocyanato benzene sulfonamide
- N-Allylmethane sulfonamide
- N-(Allyloxy)-2-nitrobenzene sulfonamide
- 1-Allyl-N-tert-butyl cyclopropane-1-sulfonamide
- N-Allyl -p -toluenesulfonamide
- Alpha-sulfophenylacetic acid
- Alpha -toluenesulfonamide
- Alpha- toluenesulfonamide
- Alpha- (p-toluenesulfonyl)- 4- fluorobenzylisonitrile
- N-(4-(2-Aminoacetyl)-5-methoxy-2-phenoxyphenyl)methanesulfonamide hydrochloride
- 2-Aminobenzene sulfonamide
- 3-Aminobenzene sulfonamide
- 4-aminobenzene -1- sulfonamide
- 2-Aminobenzene sulfonic acid (1-methylethylidene) di-4,1-phenylene ester
- (4-Amino-benzenesulfonylamino) -acetic acid
- 2-(3-Amino-benzenesulfonylamino)-benzoic acid
- 3-Aminobenzene-1-sulfonyl fluoride
- 2- [4-(2-Aminobenzenesulfonyl) piperazin-1-yl] ethanol
- 1-Amino benzimidazole-2 -sulfonic acid
- 2-Amino- 1,3-benzothiazole- 6-sulfonamide
- 4-Amino-n- benzylbenzene sulfonamide
- 4-Amino-n- benzyl-n- methylbenzenesulfonamide
- 2-Amino-5-bromobenzene sulfonamide
- N -(2-Amino-3-bromophenyl) methanesulfonamide
- 6-Amino-5-bromopyridine-3-sulfonic acid
- 2-Amino-N-butyl benzene sulfonamide
- 4-Amino-n-butylbenzene sulfonamide
- 2-Amino -N- butylbenzene sulfonamide hydrochloride
- N-(4-Aminobutyl)-5-chloro-2- naphthalenesulfonamide hydrochloride
- 3-Amino-N-butyl- 4-methylbenzene sulfonamide
- 4-Amino-n-butyl-n-methylbenzene sulfonamide
- N-(4-Aminobutyl)-2-naphthalene sulfonamide hydrochloride
- 4-Amino-6-chloro-1,3-benzenedi sulfonamide
- 4-Amino-6-chloro-benzene-1,3-disulfonyl dichloride
- 2-Amino-5-chlorobenzene sulfonamide
- 3-Amino-4-chloro-n-(2-chloro-phenyl)-benzene sulfonamide
- 5-Amino-2-chloro-n-(4-chloro-phenyl)-benzene sulfonamide
- 3-Amino-4-chloro-n,n-diethyl-benzene sulfonamide
- 5-Amino-2-chloro-n,n-diethyl-benzene sulfonamide
- 5-Amino-2-chloro-n,n-dimethyl-benzene sulfonamide
- 4-Amino-2-chloro-5-hydroxybenzen sulfonamide
- 3-Amino-n-(2-chloro-phenyl)-benzene sulfonamide
- 3-Amino-n-(4-chloro-phenyl)-benzene sulfonamide
- 4-Amino-n- (3-chlorophenyl) benzene sulfonamide
- 4-Amino-n-(4-chloro-phenyl)-benzene sulfonamide
- 3-Amino-n-(2-chloro-phenyl)-4-hydroxy-benzene sulfonamide
- 3-Amino-n-(3-chloro-phenyl)-4-hydroxy-benzene sulfonamide
- N-(2-Amino-5-chlorophenyl)methane sulfonamide
- 3-Amino-n-(3-chloro-phenyl)-4-methyl-benzene sulfonamide
- 3-Amino-n-(4-chloro-phenyl)-4-methyl-benzene sulfonamide
- 5-Amino-n-(2-chloro-phenyl)-2-morpholin-4-yl-benzene sulfonamide
- benzene sulfonamide
- Sulfachloropyridazine sodium
- 3-Amino-N-cyclohexyl-4-methylbenzene sulfonamide
- 4-Amino-N-cyclohexyl-N-methyl-benzene sulfonamide
- 3-Amino-N-cyclopropyl-4-methylbenzene sulfonamide
- 4-Amino-n-(cyclopropylmethyl)benzene sulfonamide
- 5-Amino-N-cyclopropyl-2-methylbenzene sulfonamide
- 2-Chloro-5-(trifluoromethyl)benzenesulfonyl chloride
- 4-Chloro-3-(trifluoromethyl)benzenesulfonyl chloride
- 4-Chloro-2-(trifluoromethyl)benzenesulphonyl chloride
- 3-Chloro-n,n,4-trimethylbenzene-1- sulfonamide
- 2-Cyanobenzenesulfonyl chloride
- 3-Cyanobenzenesulfonyl chloride
- 4-Cyanobenzenesulfonyl chloride
- N'-[(4-Cyanobenzene)sulfonyl]-N,N- dimethylmethanimidamide
- 2-cyanobenzenesulfonyl fluoride
- 3-cyanobenzenesulfonyl fluoride
- 4-cyanobenzenesulfonyl fluoride
- N-(2-(4'-Cyano-[1,1'-biphenyl]-4-yl)propyl)propane-2- sulfonamide
- 3-Cyano-N- cyclopropylbenzenesulfonamide
- 3-Cyano-N,N- dimethylbenzenesulfonamide
- 4-Cyano-N,N- dimethylbenzenesulfonamide
- 2- cyanoethanesulfonamide
- 3-cyano-N-ethylbenzene-1- sulfonamide
- 2-Cyano-5-fluorobenzene-1-sulfonyl chloride
- 3-Cyano-4-fluorobenzenesulfonyl chloride
- N-(2-Cyano-4-fluoro-phenyl)- methanesulfonamide
- N-(4-cyano-2-fluorophenyl) methanesulfonamide
- 3-Cyano-4-fluoro-n-(thiazol-2-yl) benzenesulfonamide
- N-(4-Cyano-2-iodo-5-(trifluoromethyl)phenyl) methanesulfonamide
- 3-Cyano-N- isopropylbenzenesulfonamide
- 2-Cyano-5-methoxybenzene-1-sulfonyl chloride
- 3-Cyano-N- methylbenzenesulfonamide
- 2-Cyano-5-methylbenzenesulfonyl chloride
- 3-Cyano-n- phenylbenzenesulfonamide
- 1-(4-Cyanophenyl)-N- cyclohexylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- cyclopropylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- cyclopropylmethanesulfonamide
- 1-(2-Cyanophenyl)-N,N- diethylmethanesulfonamide
- 1-(4-Cyanophenyl)-N,N- diethylmethanesulfonamide
- 1-( 2-Cyanophenyl)-N,N-dimethylmethanesulfonamide
- 1-( 4-Cyanophenyl)-N,N-dimethylmethanesulfonamide
- 1-(2-Cyanophenyl )-N-ethylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- ethylmethanesulfonamide
- N-(2-Cyanophenyl)-N- ethylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- isopropylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- isopropylmethanesulfonamide
- N-(2-Cyanophenyl)-N- isopropylmethanesulfonamide
- 1-(2-Cyanophenyl) methanesulfonamide
- 1-(4-Cyanophenyl) methanesulfonamide
- (2-Cyanophenyl)methanesulfonyl chloride
- (4-Cyanophenyl)methanesulfonyl chloride
- 1-(2-Cyanophenyl)-N-(2-methoxyethyl) methanesulfonamide
- 1-(4-Cyanophenyl)-N-(2-methoxyethyl) methanesulfonamide
- 1-(2-Cyanophenyl)-N-methoxy-N- methylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- methylmethanesulfonamide
- 1-(4-Cyanophenyl)-n- methylmethanesulfonamide
- 1-(2-Cyanophenyl)-N-(2-methylpropyl) methanesulfonamide
- 1-(4-Cyanophenyl)-N-(2- methylpropyl)methanesulfonamide
- 1-(2-Cyanophenyl)-N- phenylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- phenylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- propylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- propylmethanesulfonamide
- ([(4-Cyanophenyl)sulfonyl]amino) acetic acid
- 4- Cyanophenylthiourea
- N-Cyano-N-phenyl-p- toluenesulfonamide
- N-(5-(5-Cyanothiophen-2-yl)-2-methylphenyl)-4- methylbenzenesulfonamide
- Cyclamic acid
- N,N'-(1S,2S)-1,2-Cyclohexanediylbis[2-hydroxy-7,7-dimethyl-(1r,1'r,2s,2's,4s,4's)-bicyclo[2.2.1]heptane-1 -methanesulfonamide]
- Cyclohexane sulfonamide
- Cyclohexanone tosylhydrazone
- N-Cyclohexyl 2-aminobenzenesulfonamide
- N-Cyclohexyl 3-aminobenzenesulfonamide
- N-Cyclohexyl 4-aminobenzenesulfonamide
- 3-(Cyclohexylamino)-2-hydroxy-1-propanesulfonic acid
- 3-(Cyclohexylamino)propane-1-sulfonic acid
- 4-Cyclohexylbenzenesulfonyl chloride
- N-Cyclohexyl 4- boronobenzenesulfonamide
- N-Cyclohexyl 3- bromobenzenesulfonamide
- N-Cyclohexyl 4- bromobenzenesulfonamide
- N-Cyclohexyl 3-bromo-4- methylbenzenesulfonamide
- N-Cyclohexyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N-Cyclohexyl-n-(1,1-dioxidotetrahydrothien-3-yl)amine hydrochloride
- N-Cyclohexyl 4-fluorobenzenesulfonamide
- N-Cyclohexyl-3-hydroxybenzene-1- sulfonamide
- N-Cyclohexyl-4-hydroxybenzene-1- sulfonamide
- N-cyclohexyl-4-hydroxy-N-[(4-methoxyphenyl)methyl]benzene-1- sulfonamide
- N-Cyclohexyl-4- isothiocyanatobenzenesulfonamide
- 1-Cyclohexyl-3-({4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-1,2,3,4-tetrahydroisoquinolin-2-yl) ethyl]benzene}sulfonyl)urea
- N-Cyclohexyl-4-methyl-3- nitrobenzenesulfonamide
- N-Cyclohexyl 3-nitrobenzenesulfonamide
- N-Cyclohexyl-4- nitrobenzenesulfonamide
- N-Cyclohexyl-2-oxo-3- oxazolidinesulfonamide
- 3-(Cyclohexylsulfamoyl)benzoic acid
- 4-(N-Cyclohexylsulfamoyl)-2-trifluoromethylphenylboronic acid
- N- Cyclohexyltaurine
- N- Cyclohexyl-p-toluenesulfonamide
- 4- (Cyclopentanesulfonyl)aniline
- 2-(Cyclopentanesulfonyl)aniline hydrochloride
- Cyclopentanesulfonyl chloride
- 4-(Cyclopentylsulfanyl) benzaldehyde
- 3-(Cyclopentylsulfonyl) aniline
- 1-Cyclopentyl-1-tosylmethyl isocyanide
- 1,1-Cyclopropane dimethanol cyclic sulfite
- Cyclopropane sulfonamide
- Cyclopropanesulfonyl chloride
- N-Cyclopropyl 2-aminobenzenesulfonamide
- N-Cyclopropyl 3-aminobenzenesulfonamide
- 4-[(Cyclopropylamino)sulfonyl]benzoic acid
- N-Cyclopropyl 3-boronobenzenesulfonamide
- N-Cyclopropyl 4-boronobenzenesulfonamide
- N-Cyclopropyl 4-bromobenzenesulfonamide
- N-Cyclopropyl 4-bromo-3-methylbenzenesulfonamide
- N-Cyclopropyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N-Cyclopropyl 2-bromo-4-trifluoromethoxybenzenesulfonamide
- N-Cyclopropyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N-cyclopropyl-2-(1,3-dioxoisoindol-2-yl) ethanesulfonamide
- N-Cyclopropyl 4-fluorobenzenesulfonamide
- N-Cyclopropyl-3-hydroxybenzene-1- sulfonamide
- N-Cyclopropyl-4-hydroxybenzene-1- sulfonamide
- N-cyclopropyl-3-hydroxy-N-[(4-methoxyphenyl)methyl]benzene-1- sulfonamide
- N-cyclopropyl-4-hydroxy-N-[(4-methoxyphenyl)methyl]benzene-1 -sulfonamide
- 3-(Cyclopropylmethanesulfonyl) aniline
- 1-(Cyclopropylmethyl)cyclopropane-1- sulfonamide
- N-Cyclopropyl-2-methyl-5- nitrobenzenesulfonamide
- N-Cyclopropyl-4-methyl-3- nitrobenzenesulfonamide
- 3-(Cyclopropylmethyl) sulfonylazetidine
- N-Cyclopropyl-2- nitrobenzenesulfonamide
- N-Cyclopropyl-4- nitrobenzenesulfonamide
- N-Cyclopropyl-1-(3-nitrophenyl) methanesulfonamide
- 2-(4-Cyclopropylpiperazine-1-sulfonyl) aniline
- Dabrafenib mesylate
- N,N-Diallyl-4 -methylbenzenesulfonamide
- 3,4- Diaminobenzenesulfonamide
- 3,4-Diaminobenzenesulfonamide dihydrochloride
- 3,4-Diaminobenzenesulfonic acid
- 4,4'-Diaminobiphenyl-2,2'-disulfonic acid hydrate
- 2,5-Diamino-n-(2-chlorophenyl) benzenesulfonamide
- 3,4-Diamino-n-(2-chloro-phenyl) -benzenesulfonamide
- 3,4-Diamino-n-(4-chloro-phenyl)- benzenesulfonamide
- 3,4-Diamino-n,n-diethyl- benzenesulfonamide
- 3,4-Diamino-N,N -dimethylbenzenesulfonamide
- 3,4-Diamino-n-(1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)- benzenesulfonamide
- (2R,4R)-1-[(2S)-5-[(Diaminomethylidene)amino]-2-[(3R)-3-methyl-1,2,3,4-tetrahydroquinoline-8-sulfonamido]pentanoyl]-4-methylpiperidine-2-carboxylic acid
- 3-[({2-[(Diaminomethylidene)amino]-1,3-thiazol-4-yl}methyl)sulfanyl]-N- sulfamoylpropanimidamid
- 3,4-Diamino-n-(3-trifluoromethyl-phenyl)- benzenesulfonamide
- Dibenzene sulfonimide
- Dibenzo[b,d]furan-2-sulfonyl chloride
- N,N-Dibenzyl-1 -phenylmethanesulfonamide
- 2,4-Dibromobenzenesulfonyl chloride
- 2,5-Dibromobenzenesulfonyl chloride
- 1,2-Dibromo-4- methanesulfonylbenzene
- 1,3-Dibromo-5- methanesulfonylbenzene
- 3,4- Dibromosulfolane
- 4,5-Dibromothiophene-2-sulfonyl chloride
- 2,4 -Dichlorobenzenesulfonamide
- 3,5- Dichlorobenzenesulfonamide
- 2-(3,4-Dichloro-benzenesulfonylamino)-benzoic acid
- 2,3-Dichlorobenzenesulfonyl chloride
- 2,4-Dichlorobenzenesulfonyl chloride
- 3,5-Dichlorobenzenesulfonyl chloride
- 3',4'-Dichloro[1,1'-biphenyl]-4-sulfonyl chloride
- 1,4-Dichloro-2- [(2-chloroethane)sulfonyl]benzene
- 2,4-Dichloro-7, 8-dihydro-5H-S,S-di-oxoisothiopyrano[4,3-d]pyrimidine
- 2,2-Dichloro-N-[(1R,2S)-3-fluoro-1-hydroxy-1 -(4-methanesulfonylphenyl)propan-2-yl]acetamide
- 2,6-Dichloro-3- methanesulfonylpyridine
- 2,4-Dichloro-6-methylbenzenesulphonyl chloride
- 1,2-Dichloro-4- methylsulfonylbenzene
- 1,4-Dichloro-2-[(2-chloroethane)sulfonyl] benzene
- 2,4-Dichloro-7,8-dihydro-5H-S,S-di-oxoisothiopyrano[4,3-d] pyrimidine
- 2,2-Dichloro-N-[(1R,2S)-3-fluoro-1-hydroxy-1-(4-methanesulfonylphenyl)propan-2-yl] acetamide
- N,N-Diethyl 2-aminobenzenesulfonamide
- N,N-Diethyl 3-aminobenzenesulfonamide
- N,N-Diethyl 4-aminobenzenesulfonamide
- 5-[(Diethylamino)sulfonyl]-2-morpholin-4-ylbenzoic acid
- 1-(4-[(Diethylamino)sulfonyl]phenyl)-5-oxopyrrolidine-3-carboxylic acid
- N,N-Diethyl-4,4'-bipiperidine-1- sulfonamide
- N,N-Diethyl 2-boronobenzenesulfonamide
- N,N-Diethyl 3-boronobenzenesulfonamide
- N,N-Diethyl 4-boronobenzenesulfonamide
- N,N-Diethyl 5-borono-2-methoxybenzenesulfonamide
- N,N-Diethyl 3-borono-4-methylbenzenesulfonamide
- N,N-Diethyl 3-bromobenzenesulfonamide
- N,N-Diethyl 4-bromobenzenesulfonamide
- N,N-Diethyl 3-bromo-4-methylbenzenesulfonamide
- N,N-Diethyl 3-bromo-5-methylbenzenesulfonamide
- N,N-Diethyl 4-bromo-3-methylbenzenesulfonamide
- N,N-Diethyl 2-Bromo-4-trifluoromethoxybenzenesulfonamide
- N,N-Diethyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N,N-Diethyl 4-bromo-3-trifluoromethylbenzenesulfonamide
- N,N-Diethyl-4- fluorobenzenesulfonamide
- N,N-Diethyl-3-hydrazinocarbonyl- benzenesulfonamide
- N,N-Diethyl-2-hydroxybenzene-1- sulfonamide
- N,N-Diethyl-3-hydroxybenzene-1- sulfonamide
- N,N-Diethyl-4-hydroxybenzene-1- sulfonamide
- N,N-Diethyl-4 -isothiocyanatobenzenesulfonamide
- N,N-Diethyl-4-methyl-3 -nitrobenzenesulfonamide
- N,N-Diethyl-7-methyl-3-oxo-3,4-dihydro-2h-1,4- benzoxazine-6-sulfonamide
- N,N-Diethyl-2- nitrobenzenesulfonamide
- N,N-Diethyl 3-nitrobenzenesulfonamide
- N,N-Diethyl 4-nitrobenzenesulfonamide
- N,N-Diethyl-3-nitro-4-piperazin-1 -ylbenzenesulfonamide
- N,N-Diethyl(sodiosulfanyl) carbothioamide
- 3-Diethylsulfamoylbenzoic acid
- 4-Diethylsulfamoyl-benzoic acid
- [4-(diethylsulfamoyl)-3-methylphenyl]boronic acid
- 4-(N,N-Diethylsulfamoyl)-2-methylphenylboronic acid
- 3-[4-(Diethylsulfamoyl)phenyl]propanoic acid
- N,N-Diethyl-p- toluenesulfonamide
- Diethyl (tosyloxy)methylphosphonate
- 2,4 -Difluorobenzenesulfonamide
- 2,5- Difluorobenzenesulfonamide
- 2,6- Difluorobenzenesulfonamide
- 3,5- Difluorobenzenesulfonamide
- 2,5-Difluorobenzenesulfonyl chloride
- 2,6-Difluorobenzenesulfonyl chloride
- 3,4-Difluorobenzenesulfonyl chloride
- 2,2-Difluoroethyl trifluoromethanesulfonate
- N-(2,4-Difluoro-3-formylphenyl) propane-1-sulfonamide
- Difluoromethanesulfonamide .
- 3-(Difluoromethoxy)benzenesulfonyl chloride
- 1,3-Difluoro-5 -methylsulfonylbenzene
- 2,4-Difluoro-5-nitrobenzenesulfonyl chloride
- 3-(2,4-Difluorophenylsulfonylamino)-2-methoxypyridine-5-boronic acid pinacol ester
- 2,3-Dihydro-benzo[1,4]dioxine-6-sulfonic acid amide
- 6-(2,3-Dihydro-benzo[1,4]dioxine-6-sulfonylamino)-hexanoic acid
- 2,3-Dihydro-1-benzofuran-5- sulfonamide
- 2,3-Dihydro-1-benzofuran-5-sulfonyl chloride
- 3,4-Dihydro-2h-1,4-benzothiazine 1,1-dioxide
- 2,3-Dihydro-1,1-dioxo-1,2- benzisothiazole
- (4R,6S)-5,6-Dihydro-4-hydroxy-6-methylthieno[2,3-b]thiopyran-7,7- dioxide
- 5-(2,3-Dihydro-indole-1-sulfonyl)-2-methoxy-benzoic acid
- 3,4-Dihydroisoquinoline-2(1h)-sulfonyl chloride
- 2-((R)-4-((3R,5S,7S,8R,9S,10S,13R,14S,17R)-3,7-Dihydroxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)pentanamido)ethane-1-sulfonic acid
- N,N-Diisopropyl 4-(4-Bromopyrazol-1-yl)benzenesulfonamide
- N,N-Diisopropyl 4-fluorobenzenesulfonamide
- N,N-Diisopropyl-4- isothiocyanatobenzenesulfonamide
- 2,4- Dimethanesulfonylbenzaldehyde
- 2,5-dimethanesulfonylbenzoic acid
- 3,4- Dimethanesulfonylbenzonitrile
- 1,2 -Dimethanesulfonyl-4-nitrobenzene
- 1,2 -Dimethanesulfonylpyrrole
- 1,4-Dimethanesulfonyl- 2-(trifluoromethyl)benzene
- 2,5- Dimethoxybenzenesulfonamide
- 2,4-Dimethoxybenzenesulfonyl chloride
- N-(2,2-Dimethoxyethyl)-4 -methylbenzenesulfonamide
- 2-(2,5- Dimethoxyphenylthio)benzaldehyde
- 2-({[(4,6-Dimethoxypyrimidin-2-yl)carbamoyl]amino}sulfonyl)-N,N-dimethylpyridine-3- carboxamide
- N-((4,6-Dimethoxypyrimidin-2-yl)carbamoyl)-3-(ethylsulfonyl)pyridine-2- sulfonamide
- 2,3-Dimethoxy-5-sulfamoylbenzoic acid
- 4-(Dimethylamino) benzenesulfonamide
- N,N-Dimethyl 4-aminobenzenesulfonamide
- 2-(Dimethylamino)-N,N-dimethyl-5- nitrobenzenesulfonamide
- 5-Dimethylamino-1- naphthalenesulfonamide
- 4-(Dimethylamino)-3- nitrobenzenesulfonamide
- 4-[(Dimethylamino)sulfonyl]benzoic acid
- ((3-[(Dimethylamino)sulfonyl]benzoyl)amino)acetic acid
- 3-(5-[(Dimethylamino)sulfonyl]-1-ethyl-1h-benzimidazol-2-yl)propanoic acid
- 5-[(Dimethylamino)sulfonyl]-2-[(2-hydroxyethyl)amino]benzoic acid
- 2,5-Dimethyl- benzenesulfonamide
- N,4- Dimethylbenzenesulfonamide
- N,N- Dimethylbenzenesulfonamide
- 2,5-Dimethylbenzenesulfonyl chloride
- 3,5-Dimethylbenzenesulfonyl chloride
- N,N-Dimethyl benzoimidazole-1-sulfonamide
- N,N-Dimethyl-4,4'-bipiperidine-1- sulfonamide
- N,N-Dimethyl 2-boronobenzenesulfonamide
- N,N-Dimethyl 3-boronobenzenesulfonamide
- N,N-Dimethyl 4-boronobenzenesulfonamide
- N,N-Dimethyl 3-borono-4-methylbenzenesulfonamide
- N,N-Dimethyl 2-bromobenzenesulfonamide
- N,N-Dimethyl 3-bromobenzenesulfonamide
- N,N-Dimethyl 4-bromobenzenesulfonamide
- N,N-Dimethyl 5-bromo-2-methoxybenzenesulfonamide
- N,N-Dimethyl 3-bromo-4-methylbenzenesulfonamide
- N,N-Dimethyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N,N-Dimethyl 5-bromopyridine-3-sulfonamide
- N,N-Dimethyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N,N-Dimethyl 4,5-diiodo-1H-imidazole-1-sulfonamide
- 3-(3,5-Dimethyl-1,1-dioxo-1,2-dihydro-1lambda6-[1,2,6]thiadiazin-4-yl)-propionic acid
- Dimethyl 2,2'-dithiobisbenzoate
- N,N-Dimethyl imidazole-1-sulfonamide
- N,N-Dimethyl 4-iodo-1H-imidazole-1-sulfonamide
- 3,5-Dimethylisoxazole-4- sulfonamide
- 3,5-Dimethylisoxazole-4-sulfonyl chloride
- 4,6-Dimethyl-2- methylsulfonylpyrimidine
- 4-(2,6-Dimethyl-morpholine-4-sulfonyl)-benzoic acid
- 2,5-Dimethyl-1-[3-(morpholine-4-sulfonyl)-phenyl]-1h-pyrrole-3- carbaldehyde
- N,N-Dimethylnicotinamide- 2-sulfonamide
- N,2-Dimethyl-5- nitrobenzenesulfonamide
- N,4-Dimethyl-3 -nitrobenzenesulfonamide
- N,N-Dimethyl 3-nitrobenzenesulfonamide
- N,N-Dimethyl-4- nitrobenzenesulfonamide
- N,N-Dimethyl-1-(3-nitrophenyl) methanesulfonamide
- N-(1,5-Dimethyl-3-oxo-2-phenyl-2,3-dihydro-4-pyrazolyl)butane-1- sulfonamide
- 4,N-Dimethyl-n-phenyl- benzenesulfonamide
- N-(2,4-Dimethyl-phenyl)-4-hydrazino-3-nitro- benzenesulfonamide
- (2,4-Dimethylphenyl)(isocyano)methyl 4-methylphenyl sulfone
- N-(2,6-Dimethylphenyl)-4- methylbenzenesulfonamide
- 4-(3,4-Dimethyl-phenylsulfamoyl)-benzoic acid
- (2,4-Dimethylphenyl) thiourea
- 3,5 -Dimethylphenylthiourea
- N,N-Dimethylpiperidine-4- sulfonamide
- {4-[(2,2-dimethylpropoxy)sulfonyl]-3-methylphenyl}boronic acid
- 4-(3,5-Dimethyl-pyrazol-1-yl)- benzenesulfonamide
- N,N-Dimethylpyridine-3- sulfonamide
- N,N-Dimethylpyrrolidine-3- sulfonamide
- N,N-Dimethylpyrrolidine-3-sulfonamide hydrochloride
- N,N -Dimethylsulfamide
- 4 -(Dimethylsulfamoyl)benzamide
- 4-(Dimethylsulfamoyl)benzene-1-sulfonyl chloride
- 3-Dimethylsulfamoylbenzoic acid
- 4-[(Dimethylsulfamoyl)methyl]benzoic acid
- 3-(5-Dimethylsulfamoyl-1-methyl-1h-benzoimidazol-2-yl)-propionic acid
- [4-(Dimethylsulfamoyl)-3-methylphenyl]boronic acid
- 2-(Dimethylsulfamoyl)-5-methylphenylboronic acid
- 3-(2-N,N-Dimethylsulfamoylphenyl)-5-fluorobenzoic acid
- 4-(3-N,N- Dimethylsulfamoylphenyl)phenol
- 4-(4-N,N-Dimethylsulfamoylphenyl) phenol
- 4-[4-(Dimethylsulfamoyl)piperazin-1-yl]phenylboronic acid
- 1-(Dimethylsulfamoyl) pyrazole
- 1-(Dimethylsulfamoyl)pyrrole-2-boronic acid
- 4-(N,N-Dimethylsulfamoyl)-2-trifluoromethylphenylboronic acid
- 1-Dimethylsulfamoyl-3-(trifluoromethyl)pyrazole-5-boronic acid
- 5,6-Dimethyl-3H-thieno[2,3-d] pyrimidin-4-one
- Sulbactam ..
- 1,4-Dioxaspiro[4.5]decan-8-yl 4-methylbenzenesulfonate
- 2-[(1,1-Dioxido-1,2-benzisothiazol-3-yl)amino]-3-methylbutanoic acid
- 4-(1,1-Dioxido-2-isothiazolidinyl)phenylboronic acid pinacol ester
- (1,1-Dioxidotetrahydro-3-thienyl)acetic acid
- 1-(1,1-Dioxidotetrahydrothien-3-yl)-3,5-dimethyl-1h-pyrazole-4- carbaldehyde
- N-(1,1-Dioxidotetrahydrothien-3-yl)-n-ethylamine hydrochloride
- (1,1-Dioxidotetrahydro-3-thienyl)hydrazine hydrochloride
- [(1,1-Dioxidotetrahydro-3- thienyl)methyl]amine
- 1-(1,1-Dioxidotetrahydrothien-3-yl)piperazine dihydrochloride
- 4-((1,4-Dioxo-1,4-dihydronaphthalen-2-yl)amino) benzenesulfonamide
- 2-(1,3-Dioxoisoindol-2-yl)-N,N- dimethylethanesulfonamide
- 2-(1,3-Dioxoisoindol-2-yl)-N- ethylethanesulfonamide
- 2-(1,3-Dioxoisoindol-2-yl)-N- isopropylethanesulfonamide
- 2-(1,3-Dioxoisoindol-2-yl) -N-propylethanesulfonamide
- 2-(1,1-Dioxo-tetrahydro-1lambda6-thiophen-3-yl-amino) -ethanol
- 2,4-Dioxo-1,2,3,4-tetrahydro-quinazoline-6-sulfonyl chloride
- (1,1-Dioxo-tetrahydrothiophen-3-yl)methylamine, hydrochloride
- 1,1-Dioxo-tetrahydro-thiopyran-4- one
- 4-(1,1-Dioxo-1,2-thiazolidin-2-yl)-2-methylphenylboronic acid pinacol ester
- N-[5-(Diphenylphosphinoylmethyl)-4-(4-fluorophenyl)-6-isopropylpyrimidin-2-yl]- n-methylmethanesulfonamide
- N,N-Dipropyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- 4-(Dipropylsulfamoyl)benzoic acid
- 2,2'- Dithiodianiline
- Di-p- toluenesulfonamide
- 1,2-Ethanedisulfonic acid
- Ethanesulfonamide .
- 3-Ethanesulfonamidobenzoic acid
- 4-(Ethanesulfonyl) aniline
- 1-(Ethanesulfonyl)azetidine-3-carboxylic acid
- 3-(Ethanesulfonyl)azetidine hydrochloride
- 2-(Ethanesulfonyl)-N,N-dimethyl-5- nitroaniline
- 1-(Ethanesulfonyl)-2-fluoro-4 -nitrobenzene
- 1- (Ethanesulfonyl)imidazole
- [(ethanesulfonyl)methyl] benzene
- 2-[(Ethanesulfonyl)methyl] benzonitrile
- 3-[(Ethanesulfonyl)methyl] benzonitrile
- 1-[(Ethanesulfonyl)methyl] -4-nitrobenzene
- 1-Ethanesulfonyl-3-methyl- piperazine
- 4-Ethanesulfonyl-3 -morpholinonitrobenzene
- 1-(Ethanesulfonyl)-2 -nitrobenzene
- 1-(Ethanesulfonyl)-4 -nitrobenzene
- 2-(Ethanesulfonyl)-3 -nitropyridine
- 2-(Ethanesulfonyl)-5- nitropyridine
- 2-(Ethanesulfonyl)-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid
- 2-Ethanesulfony) nicotinonitrile
- Ethenesulfonyl chloride
- Ethenesulfonyl fluoride
- N-(4-Ethenylphenyl) methanesulfonamide
- N-(4-Ethenylphenyl)-N-(4-methoxybenzyl) methanesulfonamide
- N-(6-Ethoxy-2-benzothiazolyl) benzenesulfonamide
- 2-Ethoxyethanesulfonyl chloride
- 2-Ethoxy-1-methanesulfonyl-4 -nitrobenzene
- N-{2-[(1S)-1-(3-Ethoxy-4-methoxyphenyl)-2-methanesulfonylethyl]-1,3-dioxo-2,3- dihydro-1H-isoindol-4-yl}acetamide
- 4-Ethoxy-3-methylbenzenesulfonyl chloride
- 5-(2-([2-(2-Ethoxyphenoxy)ethyl]amino)propyl)-2-methoxybenzenesulfonamide hydrochloride
- N-(4-Ethoxyphenyl) ethanesulfonamide
- N-(4-Ethoxyphenyl)- 4-isothiocyanatobenzenesulfonamide
- 5-[(4-Ethoxy-phenyl)-methyl-sulfamoyl]-2-methyl-benzoic acid
- N-Ethyl 3-Aminobenzenesulfonamide
- N-Ethyl 4-aminobenzenesulfonamide
- Ethyl 4-amino-2-sulfanylpyrimidine-5-carboxylate
- 3-(Ethylaminosulfonyl)benzoic acid
- 4-[(Ethylamino)sulfonyl]benzoic acid
- Ethyl 1-(4-(aminosulfonyl)phenyl)-3-(trifluoromethyl)-5-p-tolyl-1h-pyrazole-4-carboxylate
- 4- Ethylbenzenesulfonamide
- Ethyl 3-boronobenzenesulfonamide
- N-Ethyl 4-boronobenzenesulfonamide
- N-Ethyl 3-bromobenzenesulfonamide
- N-Ethyl 4-bromobenzenesulfonamide
- N-Ethyl 5-bromo-2-methoxybenzenesulfonamide
- N-Ethyl 3-bromo-4-methylbenzenesulfonamide
- N-Ethyl 3-bromo-5-methylbenzenesulfonamide
- N-Ethyl 4-bromo-3-methylbenzenesulfonamide
- Ethyl 2-(4-(4-bromophenylsulfonyl)piperazinoacetate
- N-Ethyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N-Ethyl 5-bromopyridine-3-sulfonamide
- N-Ethyl 2-Bromo-4-trifluoromethoxybenzenesulfonamide
- N-Ethyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N-Ethyl 4-bromo-3-trifluoromethylbenzenesulfonamide
- Chlorimuron -ethyl
- Ethyl 3-(chlorosulfonyl)isonicotinate
- Ethyl 4-(chlorosulphonyl)-2,5-dimethyl-3-furoate
- 1-Ethylcyclopropane-1- sulfonamide
- N-Ethyl 4-fluorobenzenesulfonamide
- N-Ethyl -4-isothiocyanatobenzenesulfonamide
- 2-(N-Ethylmethanesulfonamido)benzoic acid
- 2-(N-Ethyl-N -methanesulfonyl)amino-3-nitropyridine
- Ethyl 4-methanesulfonylcinnamate
- Ethyl 2-methanesulfonylpyridine-4-carboxylate
- 5-Ethyl-2-methoxy-benzenesulfonyl chloride
- N-Ethyl-N -(4-methoxybenzyl)benzenesulfonamide
- N-Ethyl-2-methoxy- 5-nitrobenzenesulfonamide
- Ethyl 2-(N-methylmethanesulfonamido)acetate
- N-Ethyl-2-methyl -5-nitrobenzenesulfonamide
- N-Ethyl-4-methyl- 3-nitrobenzenesulfonamide
- 4-[2-[(3-Ethyl-4-methyl-2-oxo-3-pyrrolin- 1-yl)carboxamido]ethyl]benzenesulfonamide
- Ethyl n-(methylsulfonyl)glycinate
- Ethyl 3-(methylsulfonyl)propanoate
- Ethyl methyl sulfoxide
- 1-Ethyl-5-(morpholin-4-ylsulfonyl)-1h- benzimidazole-2-thiol
- N-Ethyl 3-nitrobenzenesulfonamide
- N-Ethyl- 1-(3-nitrophenyl)methanesulfonamide
- N-Ethyl-N -(2-nitrophenyl)methanesulfonamide
- N-Ethyl-N -(3-nitrophenyl)methanesulfonamide
- N-Ethyl-N- (4-nitrophenyl)methanesulfonamide
- N-Ethyl-1 ,3-propanesultam
- N-Ethylpyridine-3- sulfonamide
- thiazoles
- 5-Acetyl- 2,4-dimethylthiazole
- 2- Amino-4-(4-bromophenyl)-5-methylthiazole
- 6-Amino-7- bromobenzothiazole
- 1- (2-Amino-benzothiazol-4-yl)-ethanone
- 2-Amino- benzothiazol-6-ol
- 2- Amino-benzothiazole-6-carbonitrile
- 4-(1-Adamantyl)- 1,3-thiazol-2-amine
- N- (2-Amino-benzothiazol-6-yl)-acetamide
- N-Allylbenzothiazolium bromide
- (Z)-2-Amino-alpha-[1-(tert-butoxycarbonyl)]-1-methylethoxyimino-4-thiazolacetic acid
- 2-Amino-5-bromothiazole hydrochloride
- 2-Amino-6-bromo -4-fluorobenzothiazole
- 2-Acetylamino-benzothiazole-6-carboxylic acid
- 2-Amino- 6-benzothiazolol hydrochloride
- N- (5-Acetyl-1,3-thiazol-2-yl)-2,2-dimethylpropanamide
- 2- Amino-4-(3-bromophenyl) thiazole
- 2- Amino-5-bromo-4-methylthiazole hydrochloride
- 2-Acetylamino-4-(chloromethyl) thiazole
- (2-Amino-benzothiazol-6-yl)-acetic acid methyl ester
- 2-Amino-5-bromothiazole-4-carboxylic acid
- 2-Amino- 5-bromobenzothiazole
- 2-Amino- 4-bromo-6-fluorobenzothiazole
- 2-Amino- 5-bromobenzothiazole 3-oxide
- (2-Aminobenzothiazol-6-yl) acetic acid
- 2-Amino-4-bromo- 7-fluorobenzothiazole
- 2-Acetamido-4- (4-hydroxyphenyl)thiazole
- 2-[4-(Aminocarbonyl) phenoxy]-4-methyl-1,3-thiazole-5-carboxylic acid
- 2-Amino-5-(carboxymethyl)-1,3-thiazole-4-carboxylic acid hydrobromide
- 2-Amino- 4-chlorothiazole
- 2-Amino-N-(2-chloro-6-methylphenyl) thiazole-5-carboxamide
- 2-Amino-5-(4-chlorophenyl)- 1,3-thiazole-4-carboxylic acid
- 2-Amino-5-(2-chlorophenyl)- 1,3-thiazole-4-carboxylic acid
- 2-Amino-5-chlorothiazole -4-carboxylic acid methyl ester
- 2-Amino-4-(5-chlorothien-2-yl) thiazole
- 2-Amino -5,6-dichlorobenzothiazole
- 2-Amino -4-(3,4-difluorophenyl) thiazole
- 2-Amino -4,6-difluorobenzothiazole
- 2 -Amino-4,5-dihydrobenzo[d]thiazol-6(7h)-one
- 1- (2-Aminobenzo[d]thiazol-6-yl)ethanone
- 4-(1-Adamantyl) -2-methylthiazole
- 2-Amino- 5-benzothiazolecarboxylic acid
- 6- Aminobenzothiazole
- 2-Amino-4-bromo-6- fluorobenzothiazole
- 2-Amino-6-bromo-1,3- benzothiazole
- 2-Amino-5-bromo- 4-isopropylthiazole
- 2-(2-Aminobenzo[d]thiazol-6-yl)acetonitrile hydrobromide
- (2-Amino-benzothiazol-6-yl)-acetic acid ethyl ester acetate
- 2-Amino- 5-bromo-4-t-butylthiazole
- (2-Amino-benzothiazol-6-yl)-acetic acid ethyl ester
- 2-Amino-5-bromothiazole hydrobromide
- 2-Amino- 5-bromo-4-cyclopropylthiazole
- 2-Amino- 5-cyano-4-phenylthiazole
- 2 -Amino-5,6-dimethylbenzothiazole
- 2-Amino- 6,7-dichlorobenzothiazole
- 2- Amino-5,6-dihydro-4h-benzothiazol-7-one HBr
- 2-Amino-4-chlorothiazole- 5-carbaldehyde
- 2-Amino- 5-chloro-4-methylthiazole
- 2- Amino-5,6-dihydro-4h-cyclopentathiazole
- 2-Amino -4-(3,5-difluorophenyl)-1,3-thiazole
- 2-Amino- 5-chlorothiazole hydrochloride
- 2 -Amino-5,5-dimethyl-5,6-dihydro-4h-benzothiazol-7-one HBr
- 2- Amino-6-ethoxybenzothiazole
- 2-Amino -5-cyanothiazole
- 2- Amino-5,6-dihydro-4h-benzothiazol-7-one
- 2-Amino- 4,5-dimethylthiazole hydrobromide
- 2- Amino-4,5-dimethylthiazole hydrochloride
- 2-(2-Aminobenzo[d] thiazol-6-yl)acetonitrile
- 2 -Amino-5,5-dimethyl-5,6-dihydro-4h-benzothiazol-7-one
- 2 -Amino-6,7-dihydrothiazolo[5,4-c] pyridin-4(5h)-one
- 2-Amino-5-(carboxymethyl) -1,3-thiazole-4-carboxylic acid hydrobromide
- 2-Amino- 5,6-dibromobenzothiazole
- 2-Amino-5-Cbz-4,5,6,7-tetrahydro- 1,3-thiazolo[5,4-c]pyridine
- 2-Amino -4,6-dichlorobenzothiazole
- 2-Amino-6-chloro-4-(trifluoromethyl) benzo[d]thiazole
- 2-Amino- 6-chlorobenzo[d]thiazole
- 2-Amino-4-chloro-1,3-thiazole-5-carboxylic acid , 2-boc protected
- 2-Amino-4-chloro- 1,3-thiazole-5-carboxylic acid
- 2-Amino-4-chloromethythiazole hydrochloride
- 2- Amino-6,7-dihydro-5h-thiazolo[5,4-b]pyridine-4-carboxylic acid tert-butyl ester
- 2- Amino-6,7-dihydro-4h-thiazolo[4,5-c]pyridine-5-carboxylic acid tert-butyl ester
- 2- Amino-4-(3-bromophenyl)thiazole
- 2-(5-acetyl-4-methyl-1,3-thiazol-2-yl) guanidine
- 2 -Amino-4-(4-bromophenyl)-5-methylthiazole
- 2-Amino-6-bromothiazolo [4,5-b]pyrazine
- 2-Acetamido-5-bromo -4-ethylthiazole
- Abafungin .
- 2-Acetamido-5-bromo-4- ethylthiazole
- 2-Acetamido-4-(4-bromophenyl) thiazole
- 2-Acetamido- 5-bromothiazole
- 2-Acetamido- 5-chlorothiazole
- 2-Acetamido-4- (2-hydroxyphenyl)thiazole
- 2-Acetamido -4-methylthiazole
- 2-Acetamido- 5-methylthiazole
- 2-Acetamido-4-methylthiazole-5-sulfonyl chloride
- 2-Acetamido -5-nitrothiazole
- 2- Acetamidothiazole
- 2-acetamido-1,3-thiazole-5-carboxylic acid
- (2-acetamido-1,3-thiazol-4-yl)acetic acid
- Acetone-benzothiazolyl -2-hydrazone
- Acetone-benzothiazoly l-2-hydrazone
- 2-(Acetylamino)-1,3-benzothiazole-6-carboxylic acid
- 2-acetamido-1,3-benzothiazole-6-carboxylic acid
- [2-(([4-(Acetylamino)phenyl]sulfonyl)amino)-1,3-thiazol-4-yl]acetic acid
- 2-Acetylamino-5-thiazolecarboxylic acid methyl ester
- 2-Acetylamino-thiazole-5-sulfonyl chloride
- 2- Acetylbenzothiazole
- 6- Acetylbenzothiazole
- N-(5-Acetyl-4-methyl- 1,3-thiazol-2-yl)acetamide
- 1 -(5-Acetyl-4-methylthiazol-2-yl)imidazolidin-2-one
- 2- (1-Acetylpyrrolidin-2-yl)-5-bromo-4-methyl-1,3- thiazole
- 2- Acetylthiazole
- 2-Acetylthiazole-5-carboxylic acid
- 4-Acetyl-thiazole-2-carboxylic acid ethyl ester
- Acotiamide hydrochloride
- Acotiamide hydrochloride trihydrate
- T16Ainh- a01
- N-Allyl-4-(4-methylphenyl)- 1,3-thiazol-2-amine
- Alpha-(2-thiazolylamino)- o-cresol
- 2- Aminobenzo[d ]thiazole-6-carbaldehyde
- 2 Aminobenzo[d]thiazole-5-carbonitrile
- 6-Amino- 2-benzothiazolecarbonitrile
- 2-Aminobenzothiazole- 6-carboxamide
- 2-Aminobenzothiazole- 6-carboxylic acid
- 2-Aminobenzo[d] thiazole-7-carboxylic acid
- 2-Amino- 1,3-benzothiazole-6-carboxylic acid hydrochloride
- 2-Amino-benzothiazole -6-carboxylic acid 2-methoxy-ethyl ester
- 2-Amino benzothiazole- 5-carboxylic acid methyl ester
- 2-Amino-benzothiazole- 6-carboxylic acid methyl ester
- 2-Amino- 1,3-benzothiazole-6-sulfonamide
- 2-Amino- 1,3-benzothiazol-7-ol
- 2- Aminobenzo[d]thiazol-5-ol
- 2-Amino- 4-chlorobenzothiazole
- 6-Amino- 2-chlorobenzothiazole
- Laboratory Chemicals
- Home Page
- Company Profile
- Contact Us
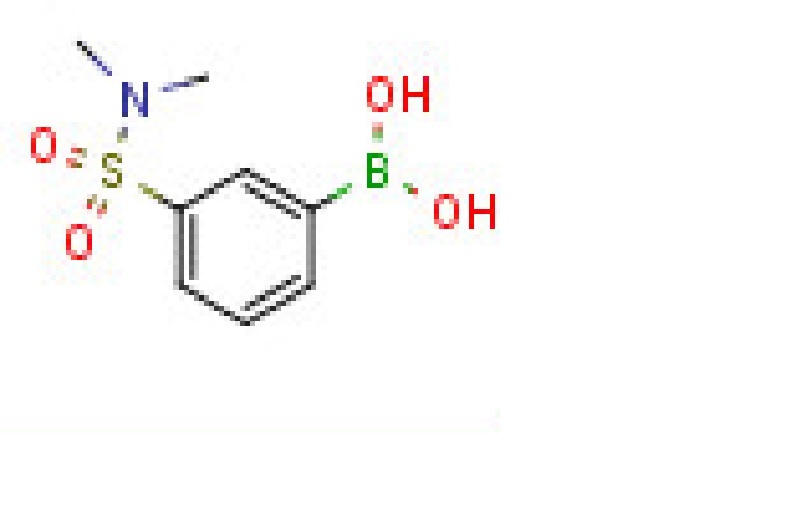
- Home
- Products
- Sulfonamides
- N,N-Dimethyl 3-boronobenzenesulfonamide
N,N-Dimethyl 3-boronobenzenesulfonamide
Product Details:
- Ph Level Not determined
- Shape Crystalline powder
- Molecular Formula C8H12BNO2S
- Application Organic synthesis, Suzuki-Miyaura cross-coupling reactions, pharmaceutical and agrochemical intermediates
- CAS No 871329-59-8
- Shelf Life 24 months (unopened, proper conditions)
- Usage Research, chemical synthesis, industrial applications
- Click to view more
-
Share Your Product:
X
N,N-Dimethyl 3-boronobenzenesulfonamide Price And Quantity
- 1 Number
- Stable at recommended storage conditions; hydrolyzes under strong acidic/basic conditions
- Not determined
- 3-(N,N-Dimethylsulfamoyl)phenylboronic acid
- Sealed bottles or drums, packed under inert atmosphere
- Not dangerous goods; transport in tightly sealed containers
- Avoid contact with skin and eyes
- For research and industrial use only; not for drug, food, or household use
- No significant hazard under normal laboratory conditions
N,N-Dimethyl 3-boronobenzenesulfonamide Product Specifications
- White to off-white solid
- Slightly soluble in polar organic solvents (e.g. DMSO, DMF); insoluble in water
- >=98.0% (HPLC)
- 197.07 g/mol
- 29309099
- Organic Intermediate
- Not determined
- Non-poisonous under normal conditions
- 149-153C
- N,N-Dimethyl 3-boronobenzenesulfonamide
- See product documentation or chemical databases
- Odorless
- Laboratory and Industrial Grade
- Organic synthesis, Suzuki-Miyaura cross-coupling reactions, pharmaceutical and agrochemical intermediates
- Not determined
- C8H12BNO2S
- Crystalline powder
- Airtight container; store under inert atmosphere; cool, dry place
- Powder
- Stable chemical; sensitive to moisture; organoboron sulfonamide compound
- 871329-59-8
- Research, chemical synthesis, industrial applications
- 24 months (unopened, proper conditions)
- Stable at recommended storage conditions; hydrolyzes under strong acidic/basic conditions
- Not determined
- 3-(N,N-Dimethylsulfamoyl)phenylboronic acid
- Sealed bottles or drums, packed under inert atmosphere
- Not dangerous goods; transport in tightly sealed containers
- Avoid contact with skin and eyes
- For research and industrial use only; not for drug, food, or household use
- No significant hazard under normal laboratory conditions
N,N-Dimethyl 3-boronobenzenesulfonamide Trade Information
- mumbai
- Telegraphic Transfer (T/T), Cash in Advance (CID), Cash Advance (CA)
- 1000 Number Per Month
- 7 Days
- Yes
- Contact us for information regarding our sample policy
- Eastern Europe, Africa, Asia, North America, Western Europe, Middle East, Central America, Australia, South America
- All India
Product Description
Chemical Name:N,N-Dimethyl 3-boronobenzenesulfonamide
Synonyms : 3-N,N-Dimethylsulfamoylphenylboronic acid
CAS No : 871329-59-8
Purity : 98%
MolecularFormula : C8H12BNO4S
Molecular Weight:229.1
Appearance : SolidOptimized for Organic Synthesis
This compound's boronic acid functionality makes it highly effective in Suzuki-Miyaura cross-coupling reactions. Chemists rely on N,N-Dimethyl 3-boronobenzenesulfonamide for producing pharmaceutical and agrochemical intermediates, as well as for advanced research in organic synthesis. Its versatility and high purity ensure consistent and reproducible results in laboratory and industrial settings.
Safe Handling and Storage
N,N-Dimethyl 3-boronobenzenesulfonamide is stable when stored in a tightly sealed, airtight container under an inert atmosphere in cool, dry conditions. Although considered non-hazardous under normal laboratory operations, direct contact with skin or eyes should be avoided. It should be stored away from moisture and strong acidic or basic environments to maintain product integrity.
FAQ's of N,N-Dimethyl 3-boronobenzenesulfonamide:
Q: How should N,N-Dimethyl 3-boronobenzenesulfonamide be stored for maximum stability?
A: Store the compound in an airtight container under an inert atmosphere, in a cool, dry location. This prevents moisture uptake and maintains stability for up to 24 months if unopened and properly stored.Q: What are the main applications of N,N-Dimethyl 3-boronobenzenesulfonamide?
A: It is primarily used in organic synthesis, such as Suzuki-Miyaura cross-coupling reactions. Additionally, it serves as an intermediate in pharmaceutical and agrochemical production and for research purposes.Q: When might this compound hydrolyze, and how can it be prevented?
A: Hydrolysis occurs when the compound is exposed to strong acids, bases, or moisture. To prevent degradation, always store it under inert gases and avoid contact with humid air or reactive chemicals.Q: Where is this compound typically utilized?
A: N,N-Dimethyl 3-boronobenzenesulfonamide is utilized in research laboratories, chemical synthesis facilities, and industrial settings focused on developing pharmaceuticals, agrochemicals, and advanced organic molecules.Q: What are the benefits of using this compound in chemical reactions?
A: Its high purity and organoboron structure enable efficient and selective cross-coupling reactions, supporting the synthesis of complex molecules with precision and reliability.Q: Is special handling required during transport or use?
A: While not classified as dangerous goods, the compound should be transported and handled in tightly sealed containers to avoid moisture exposure and contamination. Direct skin or eye contact should be avoided.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Name
Comapny Name
Phone Number
Email Id
City / State
Confirm Your Requirement
Verification Code
Did not receive yet?
Resend OTP

You’re Done!
We have received your requirements and will reply shortly with the best price.
Products You May Like
Other Products in 'Sulfonamides' category
×
Add a mobile number to receive call from "NATIONAL ANALYTICAL CORPORATION - CHEMICAL DIVISION"
×
Enter OTP
×
Share additional details for a quick response
×
Thank You!
Thank You for your valuable time. We have received your details and will get back to you shortly.
×
Get a price quote for N,N-Dimethyl 3-boronobenzenesulfonamide
×
Contact Details
- Kesarkunj, Rm No. 9, Vasanji Lalji Road, Kandivali (W), Mumbai - 400067, Maharashtra, India
- Phone : 08071630254
- Send Inquiry
NATIONAL ANALYTICAL CORPORATION - CHEMICAL DIVISION
GST : 27AWSPS9814L1ZF
GST : 27AWSPS9814L1ZF
- Mr Nilesh Shah (Director)
- Mobile : 08071630254
- Mr Dharmik Shah
+919987787082
NATIONAL ANALYTICAL CORPORATION - CHEMICAL DIVISION
All Rights Reserved.(Terms of Use)
Developed and Managed by Infocom Network Private Limited.
Developed and Managed by Infocom Network Private Limited.

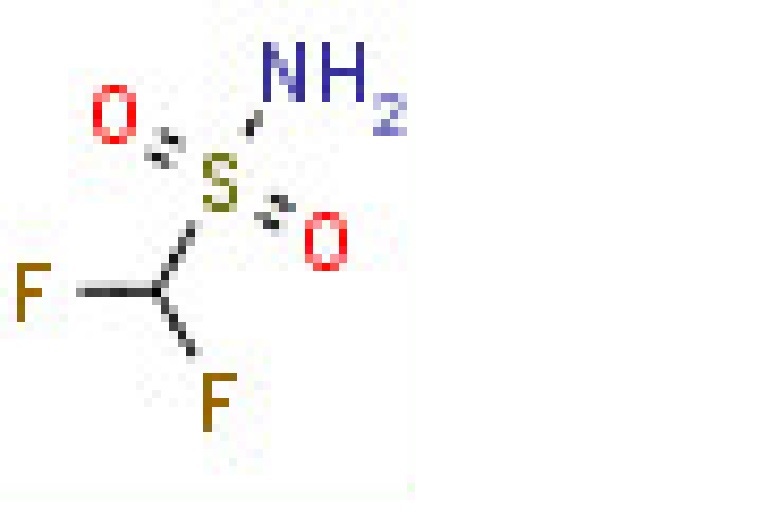
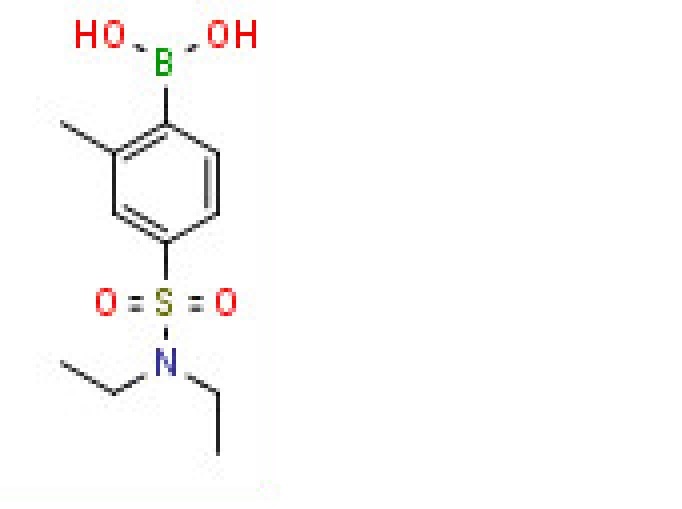
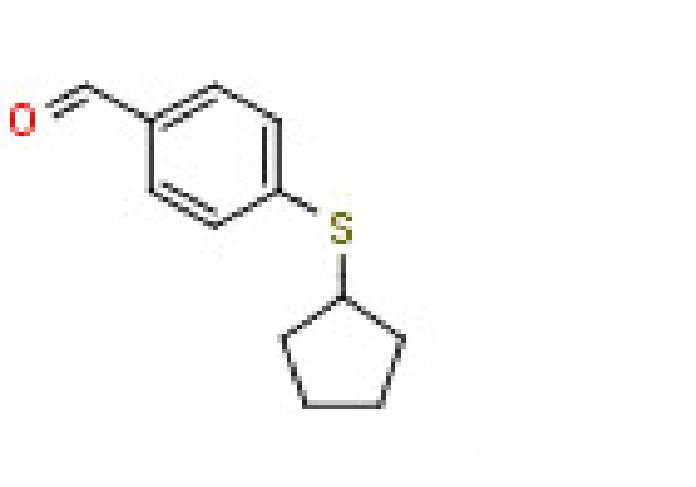
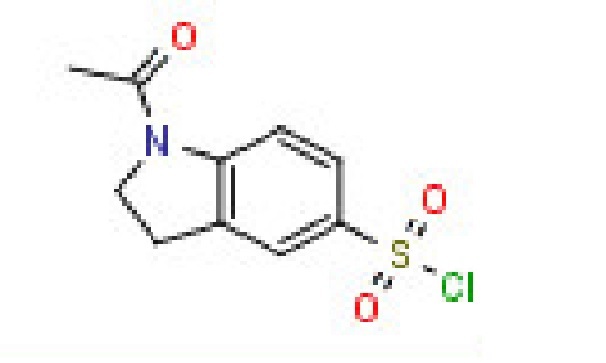
 Call Me Free
Call Me Free
