-
Our Products
- Laboratory Chemicals
- O-Bromoaniline Chemicals
- Solar Glycol
- Formic Acid
- 1-Chlorobutane For Liquid Chromatography
- 3-3-Dimethylbutyraldehyde Chemicals
- Ethyl--D-Glucuronide Chemicals
- Pure N-Heptane Chemical
- Nitric Acid
- Liquid Nitric Acid
- Tert Butyl Methyl Ether
- HACH Chemicals
- 1-Butanol Chemical
- HMB Calcium
- Zinc Sulphate Solution
- Acetonitrile Hypergrade For LC-MS
- Acetonitrile Sigma Aldrich
- Silver Iodide
- 1-Chlorobutane Chemicals
- Neutragel Neo Chemical
- Acetone Chemicals
- 3-Octanone Chemicals
- Ginkgolide C Chemicals
- Goldbio Chemicals
- 4-Aminophenol Acetaminophen RCK
- 1-Amino-3-(benzyloxy)cyclobutanecarboxylic acid hydrochloride
- 4-Aminopyrrolo[2,1-f][1,2,4]triazine-5-carboxylic acid
- 2-Amino-2- methylpropanenitrile
- Specialty Chemicals and Research Chemicals
- Hazardous and Non Hazardous Bulk Chemicals
- Aldehydes
- 3-Allylsalicylaldehyde 95%
- 1-Allyl-1h-pyrrole-2-carbaldehyde 95%
- 1-Allyl-1h-pyrazole-4-carbaldehyde 95%
- 3-Allyl-4-(2-propyn-1-yloxy)benzaldehyde 95%
- 4-(Allyloxy)-3-methoxybenzaldehyde 95%
- 4-(Allyloxy)-3-iodo-5-methoxybenzaldehyde 95%
- 4-(Allyloxy)-3-ethoxybenzaldehyde 95%
- 4-(Allyloxy)-3-chloro-5-methoxybenzaldehyde 95%
- 4-(Allyloxy)-2-chloro-5-methoxybenzaldehyd 95%
- 4-(Allyloxy)-3-chloro-5-ethoxybenzaldehyde 95%
- 2-(2-Hydroxyethoxy) Acetaldehyde 95%
- 4 - Acetamidobenzaldehyde 95%
- Gossypol-acetic Acid 95%
- Acetic acid 2-formyl-quinolin-8-yl Ester
- 3-Formylphenyl Acetate 98%
- 5-Bromo-4-formyl-2-methoxyphenyl Acetate 98%
- 4-Acetoxy-3,5-dimethoxybenzaldehyde 97%
- Acetylferulic Acid 95%
- 5-Acetoxymethyl-2-furaldehyde 98%
- 4-Acetoxy-4-methyl-1-pentanal 95%
- 2 - Acetylbenzaldehyde
- 3-Acetylbenzaldehyde 98%
- 4 Acetylbenzaldehyde 96%
- 4 Acetyl-biphenyl-4-carbaldehyde 95%
- N-Acetyl-D-Galactosamine 95%
- N-Acetyl-D-glucosamine 96%
- N-Acetylindole-3-carboxaldehyde 98%
- N-Acetyl-D-Mannosamine 97%
- 5-Acetyl-2-methoxybenzaldehyde 95%
- 4-Acetyl-1-methyl-1h-pyrrole-2-carbaldehyde 95%
- N-Acetyl-Muramic Acid 95%
- 1-(3-Acetylphenyl)-2,5-dimethyl-1h-pyrrole-3 Carbaldehyde
- 5-(4-Acetyl-phenyl)-furan-2-carbaldehyde 95%
- 1-(4-Acetylphenyl)-1h-pyrrole-2-carbaldehyde 95%
- 4-(4-Acetylpiperazin-1-yl)benzaldehyde 95%
- 1-Acetylpiperidine-4-carbaldehyde 95%
- 4-Acetyl-1h-pyrrole-2-carbaldehyde 98%
- 1-acetyl-1H-pyrrolo[2,3-b]pyridine-3 Carbaldehyde
- Acetylspiramycin 95%
- 5-Acetylthiophene-2-carbaldehyde 95%
- 5-Acetylthiophene-3-carbaldehyde 95%
- 3-Acetyl-2,4,6-trimethylbenzaldehyde 95%
- 3-(1-Adamantyl)-4-hydroxy-5-methoxybenzaldehyde 95%
- 3-(1-Adamantyl)-2-hydroxy-5-methylbenzaldehyde 95%
- 3-(1-Adamantyl)-1-phenyl-1h-pyrazole-4- carbaldehyde
- Adipaldehyde 1M in Water
- Alcaftadine 95%
- Ald-ch2-peg8-azide 95%
- Ald-ch2-peg10-t-butyl ester 95%
- Ald-ch2-peg4-ch2co2tbu 95%
- Aldehydo-D-glucose; keto-D-fructose 95%
- Ald-peg3-azide 95%
- Ald-peg1-t-butyl ester 96%
- Ald-peg4-t-butyl ester 95%
- Ald-peg5-t-butyl ester 95%
- Ald-peg5-t-butyl Ester
- Ald-peg4-nhs ester 95%
- Ald-peg4-pfp 95%
- Ald-ph-peg2-acid 95%
- Ald-Ph-PEG4-acid 95%
- Ald-ph-peg6-acid 95%
- Ald-ph-peg3-amine 95%
- Ald-ph-peg2-amine tfa salt 95%
- Ald-ph-peg2-t-butyl Ester
- Ald-ph-peg2-t-butyl Ester
- Ald-ph-peg4-t-butyl Ester
- Ald-ph-peg6-t-butyl Ester
- Ald-ph-peg2 nhboc
- Ald-ph-peg3-nh boc
- Ald-ph-peg3-o-nh boc
- Ald-ph-peg2 nhs
- All-trans Retinal
- 3-Allyl-4-(allyloxy)benzaldehyde 95%
- 3-Allyl-4-(allyloxy)-5 ethoxybenzaldehyde
- 3-Allyl-4-(allyloxy)-5 methoxybenzaldehyde
- 3-Allyl-4-butoxy-5 methoxybenzaldehyde
- -[(2E)-3-Allyl-4-chloro-5-formyl-1,3-thiazol-2(3h)-ylidene]thiophene-2-sulfonamide 95%
- 3-Allyl-4-(allyloxy)-5-methoxybenzaldehyde 95%
- 3-Allyl-4-butoxy-5-methoxybenzaldehyde 95%
- N-[(2E)-3-Allyl-4-chloro-5-formyl-1,3-thiazol-2(3h)-ylidene]thiophene-2-sulfonamide 95%
- 2-(Allyloxy)-5-chlorobenzenecarbaldehyde 95%
- 4-(Allyloxy)-2-chloro-5-ethoxybenzaldehyde 95%
- 3-Allyl-4,5-diethoxybenzaldehyde 95%
- 1-Allyl-2,3-dihydro-1h-indole-5 carbaldehyde
- 3-Allyl-4,5-dimethoxybenzaldehyde 95%
- 1-Allyl-3,5-dimethyl-1h-pyrazole-4 carbaldehyde
- 3-Allyl-4-ethoxybenzaldehyde 95%
- 3-Allyl-5-ethoxy-4-hydroxybenzaldehyde 95%
- 3-Allyl-4-ethoxy-5-methoxybenzaldehyde 95%
- 3-Allyl-5-ethoxy-4 methoxybenzaldehyde
- 3-Allyl-5-ethoxy-4 propoxybenzaldehyde
- 3-Allyl-5-ethoxy-4-(2-propyn-1-yloxy) benzaldehyde
- 2-Allyl-3-hydroxybenzaldehyde 95%
- 3-Allyl-4-hydroxybenzaldehyde 95%
- 2-Allyl-3-hydroxy-4-methoxybenzaldehyde 95%
- 3-Allyl-4-hydroxy-5-methoxybenzaldehyde 95%
- 5-Allyl-2-hydroxy-3-methoxybenzaldehyde 95%
- 1-Allyl-1h-indole-3-carbaldehyde 95%
- 3-Allyl-4-isopropoxybenzaldehyde 95%
- 3-Allyl-4-isopropoxy-5-methoxybenzaldehyde 95%
- 3-Allyl-4-methoxybenzaldehyde 95%
- 3-Allyl-5-methoxy-4-propoxybenzaldehyde 95%
- 1-Allyl-3-methyl-1h-pyrazole-4-carbaldehyde 95%
- 1-Allyl-5-methyl-1h-pyrazole-4 carbaldehyde
- 2-Allyloxybenzaldehyde 98%
- 3-(Allyloxy)benzaldehyde 95%
- 4-Allyloxybenzaldehyde 95%
- 2-(Allyloxy)-5-bromobenzaldehyde 95%
- 4-(Allyloxy)-3-bromobenzaldehyde 95%
- 4-(Allyloxy)-3-bromo-5-methoxybenzaldehyde 95%
- Fluorides
- 1-(Benzyloxy)-4-bromo-2-(trifluoromethyl) benzene
- 2-Amino-4-bromo-6-fluorobenzothiazole 95%
- 5-Acetyl-2, 2-difluoro-1,3-benzodioxole
- 1-(benzyloxy)-3-chloro-5- (trifluoromethoxy)benzene
- 2-Azaspiro[3.3]heptan-6-ol trifluoroacetate
- 3-Amino-4-mercaptobenzotrifluoride hydrochloride 95%
- 2-(benzyloxy)-1-chloro-4 -fluorobenzene
- Benzyl chlorofluoroacetate
- 1-(Azetidin-3-yl)-3,3-difluoropyrrolidine dihydrochloride
- 4-(Benzyloxy)-3,5-difluorobenzoic acid
- 2-(Benzyloxy)-1-bromo-5-fluoro -3-nitrobenzene
- 3-Amino-3-(3-fluorophenyl)propanoic acid...97%
- Benzyl N-[4-(4-cyano-3-fluorophenyl)phenyl]carbamate
- Benzyl n-[(3s,4r)-3-fluoropiperidin-4-yl]carbamate hydrochloride
- [2-Amino-1-(2-fluorophenyl)ethyl]dimethylamine 95%
- 2-Amino-3-fluoropyridine 98%
- Benzonitrile,4-(methylsulfonyl) -3-(trifluoromethyl)-
- 1-Benzyl-3-bromo-5-(trifluoromethyl) pyridin-2(1H)-one
- 2-Amino-6-fluorobenzonitrile 98%
- 1-(Azetidin-3-yl)-4-fluoropiperidine dihydrochloride
- 4-(Benzyloxy)- 2-fluorobenzonitrile
- 3-(Benzylcarbamoyl)-5-fluorophenylboronic acid
- 2-(Benzyloxy)- 5-fluorobenzonitrile
- 1-(4-Amino-3-fluorophenyl)ethanone 98%
- Benzyl (3-fluoro-4-morpholinophenyl)carbamate
- 4-Amino-2-fluoropyridine 98%
- 1-(benzyloxy)-3,5-dichloro- 2-fluorobenzene
- 5-Amino-2-fluoropyridine 97%
- 4-(Allyloxy) -2-fluorobenzonitrile 95%
- 1-(Azetidin-3-yl)-4,4-difluoropiperidine ditrifluoroacetic acid salt
- 3-Amino-2-hydroxy-5-(trifluoromethyl)pyridine 98%
- 4-(Azetidin-3-yl)-2,2-dimethylmorpholine ditrifluoroacetate
- 4-Amino-3-fluorophenol 97%
- 4-Amino-2,5-difluorobenzonitrile 97%
- 4- Benzyloxybenzotrifluoride
- (R)-2-Acetamido-2-(2-fluorophenyl)propanoic acid
- 3-(Benzylcarbamoyl)-4-fluorophenylboronic acid
- Benzyl 3-fluoropropanoate
- 4-Acetamido-2-fluoro-5-methylbenzenesulfonyl chloride
- N-Benzyl-2-chloro-n-(4-fluorophenyl) acetamide
- 1-Benzyloxy-4-(difluoromethyl) benzene
- 1-(Azetidin-3-yl)-3-methylazetidin-3-ol di-trifluoroacetate
- 2-Amino-5-(3,5-difluorophenyl)pyridine 98%
- 4-(benzyloxy)-2-chloro-1- fluorobenzene
- 2-Amino-5-fluoronicotinic acid 97%
- 2-(Benzyloxy)-3,5-difluorophenylboronic acid
- 5-Amino-2-fluorobenzyl alcohol 96%
- 2-Benzyloxy-1-bromo-4- fluorobenzene
- 2-Amino-4-fluorobenzo[d]thiazole 96%
- 4-Amino-2-fluoro-5-methylbenzonitrile 98%
- 1-Benzenesulfonyl-5-fluoro-1h-pyrrolo [2,3-b]pyridine
- 2H-1,4-Benzoxazin-3(4h)-one, 6,7-difluoro
- N-(1H-1,2,3-Benzotriazol-1-ylmethyl) -4-fluoroaniline
- benzyl N-(4-chloro-3-fluorophenyl)carbamate
- N-Benzyl-4-bromo-5-fluoro-2- nitroaniline
- 6-Amino-2-bromo-3-fluorophenol 97%
- 3-(1H-Benzoimidazol-2-yl) -1,1,1-trifluoro-propan-2-one
- 1-(Azetidin-3-ylcarbonyl)-4-methylpiperazine bis(trifluoroacetate)
- 4-Amino-2-fluoro-5-methylbenzenesulfonamide F310
- Benzyl ((4-fluoroazepan-4-yl)methyl)carbamate hydrochloride
- 2-Amino-3-bromo-5-fluorobenzoic acid 96%
- : {[(2R,3S,4S,5R)-5-(6-Amino-2-fluoro-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid 97%
- 1-Acetoxy -2-fluorobenzene
- (S)-1-(Azetidin-3-yl)-3-fluoropyrrolidine dihydrochloride
- 1-Benzoyl-3-(2,4-difluorophenyl) thiourea
- 3-Benzyloxy-2-chloro-6-fluorophenylboronic acid
- 4-Amino-2-fluorobenzonitrile 98%
- 2-Amino-6-fluorobenzothiazole 98%
- 2-Amino-6-fluorobenzaldehyde 98%
- 2-Amino-5-(4-fluorobenzylthio)-1,3,4-thiadiazole 95%
- 5-Amino-2-fluorophenylacetic acid 98%
- 2-Benzyloxy-1, 3-difluorobenzene
- 4-Acetyl-2,2-difluoro-1, 3-benzodioxole
- 1-Benzyl-5-bromo-6- fluorobenzimidazole
- benzyl N-(3-chloro-5-fluorophenyl)carbamate
- 2-Amino-5-fluoro-4-picoline 95%
- 4-Amino-2-fluorobenzoic acid 98%
- 4-(benzyloxy)-2, 3-difluorophenol
- 6-Benzyl-2,4-dichloro-8,8-difluoro-5,6,7,8-tetrahydropyrido[4,3-d]pyrimidine hydrochloride
- 3-Amino-5-fluoropyridin-2-ol 96%
- 2-Amino-5-(4-fluorophenyl)isonicotinic acid 97%
- 3-Amino-2-fluoro-6-methylpyridine 98%
- 4-Acetamido-3-fluorobenzoic acid
- 2-Benzoxazol-2-yl-1-(3-trifluoromethylphenyl) ethanol
- 1-(Azetidin-3-yl)-3-fluoropyrrolidine dihydrochloride
- 1-Acetamido-3, 5-difluorobenzene
- [3-Acetyl-5-(4-fluorophenyl)-2-methyl-1h-pyrrol-1-yl]acetic acid 95%
- 3-(Benzyloxy)-6-bromo-2- fluoroanisole
- 4-(Azetidin-3-ylcarbonyl)morpholine trifluoroacetate
- 3-Amino-6-fluoropicoline.... 98%
- Benzyl (2,4-difluorophenyl)carbamate
- 1-[2-(Benzyloxy)ethoxy]-3-bromo- 5-fluorobenzene
- Benzyl 3,3-difluorocyclobutylcarbamate
- 2'-Amino-5'-chloro-2,2,2-trifluoroacetophenone 98%
- Azetidin-1-yl(azetidin-3-yl)methanone trifluoroacetate
- 4-(Benzyloxy)-2,3-difluorophenylboronic acid
- 1-Benzyl-5-bromo-6- fluorobenzotriazole
- 2-Amino-3-fluorophenol hydrochloride....98%
- 2-Amino-4-bromo-5-fluorophenol 96%
- 2-Amino-6-fluorobenzamide 98%
- 5-Amino-2-methoxy-3-(trifluoromethyl) pyridine
- 1-(Azetidin-3-yl)pyrrolidin-2-one trifluoro acetate
- 3-(4-Benzyl-3-oxo-7-trifluoromethyl-3,4-dihydro-quinoxalin-2-yl)-propionic acid
- 4-Amino-2-fluoro-5-methoxybenzonitrile 98%
- Benzoic acid, 3,4,5-trifluoro-, 1,1-dimethylethyl ester
- 1-({[(Benzyloxy)carbonyl]amino}amino)- 2,2,2-trifluoroethanone
- 4-Amino-2-fluorophenylboronic acid pinacol ester...97%
- Benzoyl-1,1,1- trifluoroacetone
- 2-Acetamido -3-bromo-5-fluoropyridine
- 3-Amino-7-fluoro-1-methylindazole 96%
- benzyl N-(5-chloro-2-fluorophenyl)carbamate
- Acetamide, n-[4-[2,2,2-trifluoro-1-hydroxy-1-(trifluoromethyl)ethyl]phenyl]-
- 1-Benzoyl-3-(2,5-difluorophenyl) thiourea
- 5-Amino-8-fluoroquinoline 98%
- 5-Amino-2-fluoro-3-picoline 98%
- 2-Acetamido -6-trifluoromethylpyridine
- 3-Amino-3-(3-fluorophenyl)propanoic acid 97%
- 5-Benzyloxy- 2-fluoroaniline
- 5-Amino-2-fluorobenzonitrile 97%
- 2-Amino-5-methoxybenzotrifluoride 98%
- Benzyl N-(4-chloro-2-fluorophenyl)carbamate
- Benzenepropanoic acid, 2,3,6-trifluoro-
- ((2R,3R,5R)-3-(Benzoyloxy)-4,4-difluoro-5-((methylsulfonyl)oxy)tetrahydrofuran-2-yl)methyl benzoate
- 2-(benzyloxy)-5-chloro-1,3- difluorobenzene
- 4-Acetamido -3-bromobenzotrifluoride
- 5-Amino-2-methyl benzotrifluoride
- (4-Amino-2-fluorophenyl)acetic acid 98%
- Benzyl N-(5-bromo-2,3-difluorophenyl)carbamate
- 4- Acetylbenzotrifluoride
- 1-(Benzyloxy)- 4-fluorobenzene
- 2-Amino-5-fluorobenzonitrile 97%
- 5-Amino-2-fluorobenzamide 98%
- N-Acetyl-3-(2,5-difluorophenyl) -d-alanine
- 2-Aminomethyl-4-fluorophenylboronic acid, pinacol ester
- 2-(Benzylaminomethyl) -4-bromo-1-fluorobenzene
- 2-Amino-3-methoxy-4-fluorobenzoic acid 98%
- 3-Amino-2-fluoropyridine 97%
- N6-Benzoyl-2'-fluoro-2'- deoxyadenosine
- 4-Amino-2-fluorobenzotrifluoride 97%
- 2-Amino-5-bromo-3-trifluoromethylpyridine 98%
- 1-(Benzyloxy)-2,4-dibromo-5- fluorobenzene
- 4-Benzyloxy-2-fluoroaniline hydrochloride
- 2-Amino-4- trifluoromethyloxazole
- Benzyl[(5-fluoropyridin-2-yl) methyl]amine
- Benzyl (2-(4-((4-fluorobenzyl)carbamoyl)-5-hydroxy-6-oxo-1,6-dihydropyrimidin-2-yl)propan-2-yl)carbamate
- 3-Amino-5-fluorobenzotrifluoride 98%
- N-(4-(Benzyloxy)benzylidene) -4-fluoroaniline
- (3R)-1-(3-Azetidinyl)-3-fluoro-pyrrolidine dihydrochloride
- 1-(Azetidin-3-yl)-3-fluoroazetidine oxalate
- 3-(benzyloxy)-1-chloro-2,4-difluoro- 5-methylbenzene
- Benzyl N-[2-(3-fluoropyridin-4-yl)ethyl]carbamate
- Benzyl [(1r)-2-(3,5-bis(trifluoromethyl)phenyl)-1-methyl-2-oxo-ethyl]carbamate
- Benzyl N-(4-bromo-2-fluorophenyl)carbamate
- 1-Benzyl-3,3-difluoropiperidin-4-one oxime
- 2-Amino-5-fluorobenzotrifluoride 98%
- 2-Acetamido-5-fluorobenzoic acid
- 3-Amino-4-fluorobenzoic acid hydrochloride 98%
- 3-Amino-4-fluorophenol 97%
- 5-Amino-3-fluorobenzonitrile 97%
- N-Benzyl-3- fluorobenzamide
- Benzyl 5-fluoropiperidin-3-ylcarbamate hydrochloride
- 3-Amino-5-bromo-2-fluoropyridine 98%
- 3-(Benzyloxy)-2,6-difluorophenylboronic acid
- 2-Amino-5-fluoropyrimidine 98%
- 1-Benzyl-3,3-difluoro-4- phenylpiperidin-4-ol
- 4-(Benzyloxy)-3-chloro-5-fluorophenylboronic acid
- (1-Benzyl-4-fluoropiperidin-3-yl)methyl acetate
- 2-Amino-3-nitro-5- fluoropyridine
- 2-Acetamido -1-nitro-3,5,6-trifluorobenzene
- 2-Aminomethyl-4-fluorophenylboronic acid hydrochloride
- (1-Benzyl-4-fluoropiperidin-3-yl) methanol
- (4R,12As)-n-(2,4-difluorobenzyl) -7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-[1,3]oxazino[3,2-d]pyrido[1,2-a]pyrazine-9-carboxamide
- 3-Amino-6-fluoro-1-methylindazole 98%
- (S)-2-(2-((S)-2-Acetamido-4-methylpentanamido)acetamido) -n-(4-methyl-2-oxo-2h-chromen-7-yl)-6-(2,2,2-trifluoroacetamido)hexanamide
- Benzyl N-[2-(4-fluorophenyl)propan-2-yl]carbamate
- 4-Amino-3- nitrobenzotrifluoride
- Benzyl N-(3,3-difluoro-1-methylcyclobutyl)carbamate
- 1-(benzyloxy)-3-chloro-2,4- difluorobenzene
- Benzoyl trifluoromethanesulfonate
- 2-(Benzyloxy)-5-chloro-3- fluoropyridine
- 4-Amino-3-(trifluoromethoxy) benzonitrile
- 3-Amino-2-(methylamino)-5-(trifluoromethyl) pyridine
- N4-Benzoyl-2'-fluoro-2'- deoxycytidine
- 5-Aminomethyl-4-2luorophenylboronic acid, pinacol ester
- [(3S,4R)-1-Benzyl-4-(3-fluorophenyl)pyrrolidin-3-yl] methanol
- 4-(6-Aminopyridin-3-yl)-N-ethyl -2-fluorobenzamide
- Azido-peg4-pfp ester
- 3-Amino-2-fluorobenzotrifluoride 98%
- 2-Amino-4-fluorobenzamide 98%
- 5-Amino-3-(2-fluorophenyl)-1-methylpyrazole ...96%
- [1-(Azetidin-3-yl)-4-piperidyl]methanol di trifluoro acetic acid salt
- 2-Benzyloxy- 6-fluorobenzonitrile
- 3-(Benzyloxy)-6-bromo-2- fluorophenol
- Azetidin-3-yl acetate 2,2,2-trifluoroacetate
- Benzyl N-(3-bromo-2-fluorophenyl)carbamate
- 4-(1H-Benzimidazol-2-yl)-1-[3-(trifluoromethyl)phenyl] pyrrolidin-2-one
- 3-Amino-4-(trifluoromethyl)phenylboronic acid
- 4-Aminophenylsulfur pentafluoride
- 2-Amino-4-(trifluoromethyl)phenol hydrochloride
- 2-Amino-5-chloro-3-fluoropyridine 98%
- 1-(benzyloxy)-5-chloro-2, 4-difluorobenzene
- L-Asparagine 7-amido-4-methylcoumarin trifluoroacetate
- 5-Amino-3-[3-(trifluoromethyl)phenyl] isoxazole
- 2-(benzyloxy)-4-chloro-1- (trifluoromethyl)benzene
- 4-Amino-2,6-difluoropyridine 97%
- 1-(1,3-Benzodioxol-5-yl) -4,4,4-trifluorobutane-1,3-dione
- 2-Amino-5-fluorothiazole hydrochloride 97%
- Benzyl 2-fluoro-6-nitrobenzoate
- Benzoyl -1,1,1-trifluoroacetone
- 5-Amino-8- trifluoromethoxyquinoline
- 3- Benzyloxybenzaldehyde
- 2-Amino-5- nitrobenzotrifluoride
- Acetic acid 3-fluoro-4-methyl-phenyl ester
- 2-Amino-3,4-difluorobenzoic acid 98%
- 6-Benzyl-8,8-difluoro-5,6,7,8-tetrahydropyrido[4,3-d] pyrimidine-2,4-diol
- Benzyl [2-(3,5-bis(trifluoromethyl)phenyl)-1-methyl-2-oxo-ethyl]carbamate
- 3-(Benzyloxy)-5-fluorobenzoic acid
- : 3-(Benzyloxycarbonylamino)-4-fluorophenylboronic acid, pinacol ester
- 2-Benzyloxy- 4-fluoroaniline
- 3-Amino-4-fluorobenzoic acid 98%
- 4-(3-Aminophenyl)-N-ethyl-2- fluorobenzamide
- 2-(Benzyloxy)-1-bromo-3,5- difluorobenzene
- 4-Amino-2-ethoxy-5-fluorobenzonitrile 96%
- 2-Amino-3,5-difluoropyridine 97%
- (R)-1-(Azetidin-3-yl) -3-fluoropiperidine
- 1-Benzyl-3,3-difluoropiperidin -4-one
- 2-Acetamido-4-fluorobenzoic acid
- 3-Amino-3-(2-fluorophenyl)propan-1-ol hydrochloride 98%
- 4-(Benzyloxy)- 2-fluoroaniline
- 2-Amino-5-fluoro-3-iodopyridine 95%
- (1-Benzyl-4,4-difluoropiperidin-3-yl) methanol
- Benzyl 2-(benzyloxy)-4-fluorobenzoate
- 3-(benzyloxy)-1-chloro-2,4-difluoro-5- methylbenzene
- 2-Amino-2'-fluoro-5-nitrobenzophenone 98%
- 2-Amino-5-fluorophenol 95%
- (3-Amino-2-fluorophenyl)methanol.... 97%
- Benzyl (3,3-difluoro-2-hydroxypropyl)carbamate
- 4-(1,3-Benzodioxol-5-yl)-6-(trifluoromethyl) pyrimidine-2-thiol
- 2H-1,4-Benzoxazine, 7-fluoro-3,4-dihydro-3-methyl-
- 2-Amino-4'-fluoroacetophenone hydrochloride 97%
- 4-Amino-5-chloro-2-ethoxy-N-({4-[(4-fluorophenyl)methyl]morpholin-2-yl}methyl)benzamide 98%
- N-(2-Amino-3-fluorophenyl)methanesulfonamide 96%
- 7-[(4aS,7aS)-Octahydro-1H-pyrrolo[3,4-b]pyridin-6-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride
- 2-Amino-5-fluoropyridine 97%
- 2-Amino-4-fluorobenzonitrile 98%
- Benzyl (5-fluoroazepan-4-yl)carbamate hydrochloride
- 5-(Benzyloxy)-2- bromobenzotrifluoride
- 5-(4-Aminophenyl)-2-fluorobenzoic acid
- 2-(Benzofuran-2-yl) -4-fluorobenzonitrile
- N-(4-Acetylphenyl)-2,2,2-trifluoroacetamide 95%
- 1-Acetamido-4-bromo-2- (trifluoromethoxy)benzene
- 2-Amino-4-fluoropyridine 98%
- 4-Amino-3-fluorobenzoic acid 98%
- Azetidin-3-yl(3-fluorophenyl) methanone
- 1-(benzyloxy)-2-chloro-5-fluoro- 4-methylbenzene
- 1-(benzyloxy)-2,3-dichloro- 4-fluorobenzene
- Benzyl ((3-fluoroazepan-3-yl)methyl)carbamate
- 2-(benzyloxy)-1-chloro-4- fluorobenzene
- 5-Amino-2-fluoro-4-hydroxybenzoic acid 95%
- 7-[(4aS,7aS) -Octahydro-1H-pyrrolo[3,4-b]pyridin-6-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid hydrochloride
- 2-(Benzyloxy)- 5-fluorobenzamide
- 2-(Benzyloxy)-3-bromo-5- fluoroaniline
- 4-Amino-2,3,5,6- tetrafluoropyridine
- 2-Amino-5-(trifluoromethoxy) benzonitrile
- 2-Amino-4,5,6- trifluorobenzothiazole
- 5-Amino-2-pyrrolidin -1-yl-n-(3-trifluoromethyl-phenyl)-benzenesulfonamide
- 4-(6-Aminopyridin-3-yl)-2-fluorobenzoic acid
- 3-Amino-4-(trifluoromethoxy) benzonitrile
- 2-Amino-4-(trifluoromethoxy) phenol
- 2-Amino-6-(trifluoromethoxy) phenol
- 4-Amino-3-(trifluoromethoxy)phenylboronic acid, pinacol ester
- (2R)-2-Amino-3-[4-(trifluoromethoxy)phenyl]propanoic acid
- 1-Benzyl-3,3-difluoro-4, 4-piperidinediol
- 2-(benzyloxy)-1,4-dichloro- 3-fluorobenzene
- (3Ar,4r,5r,6as) -4-((e)-3,3-difluoro-4-phenoxybut-1-en-1-yl)-5-hydroxyhexahydro-2h-cyclopenta[b]furan-2-one
- Benzoic acid, 3-(3-fluoropropoxy)-
- 4-Amino-2-(trifluoromethyl)benzoic acid
- ((3Ar,5r,6s,6ar)-2,2-dimethyl-6-(((trifluoromethyl)sulfonyl)oxy)tetrahydrofuro[2,3-d][1,3]dioxol-5-yl)methyl 4-methylbenzoate
- 2-Amino-5-(trifluoromethyl) benzamide
- 1-(Azetidin-3-ylcarbonyl)azepane trifluoroacetate
- (3Ar,4r,5r,6as)-4- ((e)-3,3-difluoro-4-phenoxybut-1-en-1-yl)-5-hydroxyhexahydro-2h-cyclopenta[b]furan-2-one
- (3S,11Ar)-n-(2,4-difluorobenzyl) -6-methoxy-3-methyl-5,7-dioxo-2,3,5,7,11,11a-hexahydrooxazolo[3,2-d]pyrido[1,2-a]pyrazine-8-carboxamide
- (3As,4r,6ar) -6-((benzyloxy)methyl)-5-fluoro-2,2-dimethyl-4,6a-dihydro-3ah-cyclopenta[d][1,3]dioxol-4-ol
- 4-Amino-2-(trifluoromethyl) phenol
- 1-(benzyloxy)-2-chloro-3 -fluorobenzene
- 6-Amino-3-(4-fluorophenyl)picolinic acid ....98%
- 5-Amino-4-cyano-1-(4-fluorophenyl)-3-methylpyrazole 97%
- 2-Amino-5-(4-trifluoromethoxyphenyl) pyridine
- 2-[4-Amino-3-(trifluoromethyl) phenyl]acetonitrile
- 1-[(Ammoniooxy)methyl]-3-(trifluoromethyl)benzene chloride
- Amyltriethyl ammonium
- Ammonium perfluoro(2-methyl-3-oxahexanoate)
- 2-Amino-4-trifluoropyrimidine-5-boronic acid pinacol ester
- 2-Aminomethy-4-(4-fluorobenzyl) morpholine
- 2-Amino-4-(trifluoromethyl)thiazole-5-carboxylic acid
- 4-Amino-3-(trifluoromethoxy)benzoic acid
- 3-Amino-1,1, 1-trifluoropropan-2-ol
- 2-Aminomethyl-3-chlorophenylboronic acid, pinacol ester
- N-Benzyl-4- fluorobenzenesulfonamide
- 3-Azetidineacetic acid trifluoroacetate
- 2-Amino-6-(trifluoromethyl) pyridine
- Ammonium hexafluorozirconate
- 3-Amino-5-chlorobenzotrifluoride 98%
- 5-Amino-2-fluorobenzoic acid 95%
- 4-Acetoxy-2,3,5,6-tetrafluorobenzoic acid
- 1-Benzoyl-3- (2,3-difluorophenyl)thiourea
- 3-Amino-4-fluorophenylboronic acid, pinacol ester 96%
- Benzyl N-[1-(4-fluorophenyl)cyclobutyl]carbamate
- (2R,3R,4S,5R)-2-[(Benzoyloxy)methyl]-5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-4-fluorooxolan-3-yl benzoate
- 4-Amino-5-bromo-2-(trifluoromethyl)benzonitrile 97%
- 3-Amino-2-fluorobenzonitrile 97%
- 1-Benzyl-4,4- difluoropiperidine
- 2-(benzyloxy)-1-chloro-3,5- difluorobenzene
- Benzyl N-[3,5-bis(trifluoromethyl)phenyl]carbamate
- 1-(Azetidin-3-yl) -4,4-difluoropiperidine
- 1-(Azetidin-3-yl) -3-fluoroazetidine
- 2-Amino-5-trifluoromethyl -1,3,4-thiadiazole
- 2-Amino-5-(trifluoromethyl)benzoic acid
- 2-Amino-2-(4-fluoro-phenyl)-propionic acid 95%
- 6-Amino-2-fluoropyridine-3-boronic acid pinacol ester 95%
- 2-Aminohexafluoropropan-2-ol 95%
- 4-(Benzyloxy)-3-fluoroaniline hydrochloride
- 2-Amino-3-fluorobenzoic acid 98%
- (2R,3R,4R-3-Benzoyloxy-4-fluoro-4-methyl-5-oxotetrahydrofuran-2-yl)methyl benzoate
- 3-Amino-2-methyl benzotrifluoride
- 5-Amino-2-fluoro-4-picoline 95%
- 1-[(Ammoniooxy)methyl]-4-fluorobenzene chloride
- 3-(benzyloxy)-1-bromo-2,4-difluoro-5 -methylbenzene
- (3R)-3-Amino-1-[3-(trifluoromethyl) -5H,6H,7H,8H-[1,2,4]triazolo[4,3-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one
- 2-Amino-4,6-difluoropyrimidine 96%
- 3-Benzyloxy-4-chloro-2-fluorophenylboronic acid
- [3-(Benzyloxy)-2,4-difluoro-5-methylphenyl]boronic acid
- 1-(Benzyloxy)-3-bromo-5- fluorobenzene
- 2-Amino-2-(2-fluorophenyl)ethanol 95%
- 4-(Aminomethyl)-3-(trifluoromethyl) aniline
- 2-(N-Benzylamino) -5-chloro-3-fluoropyridine
- 6-Amino-3-Bromo-2-trifluoromethylpyridine 98%
- 4-Amino-5-(2-fluorophenyl)-4h-1,2,4-triazole-3-thiol 95%
- 2-Amino-5-[4-fluoro-3-(trifluoromethyl)phenyl]pyridine 98%
- 2-Acetamido -5-fluorotoluene
- (S)-3-Amino-4-(2-trifluoromethylphenyl)butanoic acid hydrochloride
- 3-Amino-3-(2-fluorophenyl)propanoic acid 98%
- 2-Acetamido-4-(trifluoromethyl)phenylboronic acid, pinacol ester
- N-Benzyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- 4-(Benzyloxy)-1-chloro- 2-fluorobenzene
- 3-(2-Amino-5-fluoropyridin-3-yl)prop-2-yn-1-ol 95%
- Benzyl(ethyl)dimethylammonium bis(trifluoromethanesulfonyl)imide
- 1-Acetyl -2-bromo-4-fluorobenzene
- 1-(4-Azepan-1-yl-3-fluorophenyl) ethanone
- (4R,12As)-n- (2,4-difluorobenzyl)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-[1,3]oxazino[3,2-d]pyrido[1,2-a]pyrazine-9-carboxamide
- 1-Benzyl-5-bromo-6-fluoro-2-methyl-1, 3-benzodiazole
- 2-Amino-3-(trifluoromethyl)benzoic acid
- 4-Amino-3-(trifluoromethyl)benzoic acid
- 3-Amino-4-fluorobenzotrifluoride 98%
- 2-(Benzyloxy)-1-chloro-3 -fluorobenzene
- 2-Aminomethyl-5-fluorophenylboronic acid hydrochloride
- 2-Amino-5-fluorobenzaldehyde 96%
- 3-Amino-n-(3-trifluoromethyl-phenyl) -benzenesulfonamide
- 2-Amino-4-trifluoromethylphenyl disulfide
- 3-Amino-2-(2-fluorophenoxy)pyridine 95%
- 6-Acetamido-2, 3-difluoroanisole
- Benzyl 3-fluoro-4-oxopiperidine-1-carboxylate
- 2-Amino-4'-fluorobenzophenone 97%
- Benzeneacetamide, 4-fluoro-n-[4-(5-oxazolyl)phenyl]-
- 5-(Benzyloxy)-2 -bromobenzotrifluoride
- [2-Amino-1-(4-fluorophenyl)ethyl]dimethylamine 96%
- 2-Amino-3-(trifluoromethoxy)benzoic acid
- 2-(Azetidine-1-carbonyl) -4-fluoroaniline
- 5-Amino-3-(2-fluorophenyl)-1-methylpyrazole .....96%
- 3-Amino-5-fluoro-1-methylindazole 98%
- 5-Amino-3-(3-fluorophenyl)-1-methylpyrazole ....95%
- Benzyl (5,5-difluoropiperidin-3-yl)carbamate hydrochloride
- (S)-3-Amino-3-(3-trifluoromethylphenyl)propionic acid
- 1-(benzyloxy)-2,4-difluoro- 5-nitrobenzene
- 2-Amino-4-fluorobenzaldehyde 96%
- 1-(benzyloxy)-4-chloro-2 -fluorobenzene
- 2-Amino-5-(4-fluoro-3-methoxyphenyl)pyridine F308
- 1-Benzhydryl-3-(trifluoromethyl) azetidin-3-ol
- 3-Amino-5-bromo-2-fluorobenzoic acid 96%
- 4-Amino-5-fluoro-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]-1,2-dihydropyrimidin-2-one 98%
- 1-(benzyloxy)-3, 5-difluorobenzene
- (+/-)-Alpha-methoxy-alpha-trifluoromethylphenylacetic acid 98%
- 3-Amino-4- (trifluoromethyl)pyridine
- 2-Amino-6-(trifluoromethoxy) benzo[d]thiazole
- 4-Amino-n-[4-(trifluoromethyl) phenyl]benzenesulfonamide
- 3-Amino-5-trifluoromethylphenylboronic acid, pinacol ester
- 4-amino-3-(trifluoromethyl) benzonitrile
- 2-Amino-1-(4-(trifluoromethyl)phenyl)ethanone hydrochloride
- 3-Amino-3-(3-trifluoromethylphenyl)propanoic acid
- 5-Amino-3-(trifluoromethyl) pyridin-2-ol
- [4-Amino-2-(trifluoromethyl)phenyl] (4-methyl-1-piperazinyl)methanone
- (R)-3-Amino-3-(3-trifluoromethylphenyl)propanoic acid
- 2-Amino-4-[(trifluoromethyl)sulfonyl] phenylamine
- 2-Amino-3- trifluoromethylpyridine
- (S)-3-Amino-3-(4-trifluoromethylphenyl)propionic acid
- N-Benzyl-5-chloro-2- fluorobenzamide
- 4-Amino-2- trifluoromethylpyridine
- 1-(benzyloxy)-3-chloro-5- fluorobenzene
- 2-Amino-4-(trifluoromethyl) pyrimidine
- 5-Amino-2- trifluoromethylpyridine
- 2-Amino-4-(trifluoromethyl)pyridine-5-boronic acid pinacol ester
- 3-Amino-5-(trifluoromethyl) pyridin-2(1H)-one
- 2-Amino-4,4,4-trifluoro-n-butyric acid hydrochloride
- 2-Amino-3- trifluoromethylbenzonitrile
- Benzoic acid, 4-[(1,2,2-trifluoroethenyl)oxy]-
- 3-Amino-n-(2,2,2-trifluoroethyl) benzamide
- 4-Amino-2-fluoro-N-methylbenzamide 98%
- N-Benzyl 2-bromo-5-trifluoromethoxybenzenesulfonamide
- 5-Amino-6-Chloro-2,2-difluorobenzodioxole 97%
- N-(4-Acetylphenyl)-2,2,3,3,4,4,4- heptafluorobutanamide 95%
- 4-Amino-2-(trifluoromethyl) benzonitrile
- 2-Amino-6-(trifluoromethyl) benzonitrile
- 2-Amino-5- trifluoromethylbenzonitrile
- 3-Amino-5-(trifluoromethyl) benzonitrile
- Ammonium trifluoroacetate
- Benzyl N-[2-(4,4-difluoropiperidin-1-yl)-2-oxoethyl]carbamate
- 2-Amino-3-fluorobutyric acid 95%
- 2-Amino-5-fluoro-4-methylphenylboronic acid, pinacol ester 97%
- Benzyl 1-(4-fluorophenyl)cyclopropylcarbamate
- 2-Benzyloxy-4- bromobenzotrifluoride
- 1-Benzyloxy-2-chloro-4- fluorobenzene
- 2-Amino-4-(4-fluorophenyl)thiazole 98%
- 2-Amino-5-fluoro-6-methylpyridine 98%
- 2-Amino-5-chlorobenzotrifluoride 98%
- 3-Amino-n-(2-fluoro-phenyl)-benzenesulfonamide 95%
- 2-(4-Aminophenyl) -6-fluorobenzothiazole
- 3-Amino-5-methyl benzotrifluoride
- 1-Benzoyl-3-(3-fluorophenyl) thiourea
- 5-Fluorocytosine 98%
- 4-(benzyloxy)-1-chloro-2-(trifluoromethyl) benzene
- N-benzyl-3-chloro-5- fluorobenzamide
- 4-Amino-3-fluorobenzonitrile 98%
- Aluminum trifluoromethanesulfonate 95%
- benzyl N-(3-chloro-2-fluorophenyl)carbamate
- 4-Benzyloxy-3,5-difluorophenylboronic acid
- 1-Benzoyl-3-[4-(trifluoromethyl) phenyl]thiourea
- 2-Amino-4-chlorobenzotrifluoride 97%
- (2S,3R,4S,5S,6R)-2-(3-(Benzo[b]thiophen-2-ylmethyl)-4-fluorophenyl) -6-(hydroxymethyl)-2-methoxytetrahydro-2h-pyran-3,4,5-triol
- 3-[3-Acetyl-5-(4-fluorophenyl)-2-methyl-1h-pyrrol-1-yl]propanoic acid 95%
- 4-Acetamido-3-fluorophenylboronic acid, pinacol ester
- 4-Azidosulfonyl-benzoic acid pentafluorophenyl ester
- 3-Amino-4-methyl-n-[3-(trifluoromethyl) phenyl]benzenesulfonamide
- 3-(Benzyloxycarbonylamino)-4-fluorophenylboronic acid, pinacol ester
- 1-(2-Aminophenyl) -2,2,2-trifluoroethan-1-one
- Benzyl ((4,4-difluoropiperidin-3-yl)methyl)carbamate hydrochloride
- 1,3-Benzodioxole, 4-chloro-2,2-difluoro-
- 2-Amino-4-fluorobenzoic acid 98%
- 2-Amino-3-chloro-5-trifluoromethylpyridine 98%
- 2-Amino-3-fluorobenzonitrile 98%
- 5-Amino-2,2-difluorobenzodioxole 97%
- 2-Amino-6-fluorobenzoic acid 98%
- 2-Acetoxy-2', 4'-difluoroacetophenone
- 4-(4-Aminophenyl)-N-ethyl-2- fluorobenzamide
- 5-(benzyloxy)-1-chloro-2-fluoro-3- (trifluoromethyl)benzene
- N-(2-Benzoyl-4-chlorophenyl) -3,4-difluorobenzamide
- Benzyl N-[1-(2-fluorophenyl)cyclopropyl]carbamate
- 2-Acetamido-5-fluorophenylboronic acid, pinacol ester
- Benzoic acid pentafluorophenyl ester
- 2-Amino-4-fluoro-5-methoxybenzoic acid 98%
- N-Benzyl-2-fluoroaniline hydrochloride
- 2-Acetamido-4,4,4-trifluorobutanoic acid
- 4-(Benzyloxy)-3-bromo-5- fluoroaniline
- N-Acetyl-3-(2,5-difluorophenyl) -l-alanine
- 3-Acetyl-4-fluorobenzoic acid
- 4-Acetyl-3-fluorobenzoic acid
- 4-Acetyl-3-fluoro-benzoic acid benzyl ester
- 4-Acetyl-3-fluoro-benzoic acid methyl ester
- 4-Acetyl-3-fluoro-benzoic acid tert-butyl ester
- 2-Acetyl-5- fluorobenzonitrile
- N-Acetyl 5-fluoro-2-methylaniline
- N-Acetyl 2-fluoro-4-(3-hydroxy-3-methylbut-1-ynyl)aniline
- 3-Acetyl-7-fluorochromen -2-one
- 4-Acetyl-n-(4-fluorobenzyl) benzenesulfonamide
- 4-Acetyl -n-(4-fluorobenzyl)benzenesulfonamide
- 2'-Acetyl-4-fluoro-3- methylbiphenyl
- 5-Acetyl-2-fluorophenylboronic acid, pinacol ester
- 2-Acetyl-3-fluoropyridine 98%
- 3-Acetyl-2-fluoropyridine 97%
- 2-(4-Acetylphenyl)-2,2-difluoroacetic acid 95%
- N-(3-Acetylphenyl)-2-fluorobenzamide 95%
- N-(3-Acetylphenyl)-3-fluorobenzamide 95%
- N-(3-Acetylphenyl)-4-fluorobenzamide 95%
- N-(4-Acetylphenyl)-2-fluorobenzamide 95%
- N-(4-Acetylphenyl)-3-fluorobenzamide 95%
- N-(3-Acetylphenyl)-2,2,3,3,4,4,4-heptafluorobutanamide 95%
- N-(3-Acetylphenyl)-2,2,2-trifluoroacetamide 98%
- N-(4-Acetylphenyl) -2,2,2-trifluoroacetamide 95%
- 1-(4-Acetylphenyl)-2,2,2-trifluoroethanone 95%
- 6-Acetyl-2,2,3,3-tetrafluorobenzo-1,4-dioxene 95%
- S-Acetylthioglycolic acid pentafluorophenyl ester 95%
- 4-Acetyl-2-trifluoromethyl-benzoic acid 95%
- 4-Acetyl-2-trifluoromethyl-benzoic acid methyl ester 95%
- 3-Acetyl-4'-(trifluoromethyl)biphenyl 95%
- N-(1-Adamantylmethyl)-4-fluoroaniline 95%
- 1-(1-Adamantyl)-4,4,4-trifluorobutane-1,3-dione 95%
- L-Alanine-7-amido-4-methylcoumarin trifluoroacetate salt 98%
- 1-Allyl-1h-benzimidazole-2-thiol 95%
- Allyl chlorodifluoroacetate 95%
- Allyl 2-chloro-1,1,2-trifluoroethyl ether 95%
- Allyl 1h,1h-heptafluorobutyl ether 95%
- Allyl 1,1,2,3,3,3-hexafluoropropyl ether 95%
- 1-Allyl-1-methylpyrrolidinium bis(trifluoromethanesulfonyl)imide 98%
- 1-(Allyloxymethyl)-2-fluorobenzene 98%
- 2-Allyloxy-1,1,2,2-tetrafluoroethanesulfonyl fluoride 95%
- Allyl 2,2,3,3,3-pentafluoropropyl ether 97%
- Allyl 1h,1h-perfluorooctyl ether 95%
- 4-Allyl-2,3,5,6-tetrafluorobenzoic acid 90%
- Allyl 1,1,2,2-tetrafluoroethyl ether 95%
- Allyl 2,2,3,3-tetrafluoropropyl ether 95%
- N-Allyl-2,2,2-trifluoroacetamide 95%
- 2-Allyl-4-(trifluoromethoxy)aniline 95%
- 1-Allyl-2-(trifluoromethyl)benzimidazole 98%
- N-Allyl-2-(3-(trifluoromethyl)-4,5,6,7-tetrahydroindazol-1-yl)acetamide 95%
- Alpha-chloro-4-fluorobenzaldoxime 96%
- Alpha-fluorophenylacetic acid 95%
- (Alpha-fluoro)phenylacetic acid methyl ester 95%
- (R)-(+)-Alpha-methoxy-alpha-trifluoromethylphenylacetic acid 97%
- (S)-(-)-Alpha-methoxy-alpha-(trifluoromethyl)phenylacetic acid 98%
- (+)-Alpha-methoxy-alpha-(trifluoromethyl)phenylacetic anhydride 95%
- Alpha-methyl-3-(trifluoromethyl)benzyl alcohol 97%
- Alpha-methyl-m-trifluoromethylbenzyl bromide 98%
- Alpha-(p-toluenesulfonyl)-4-fluorobenzylisonitrile 98%
- (R)-Alpha-(4-trifluoromethyl-benzyl)-proline hydrochloride 95%
- (S)-Alpha-(4-trifluoromethyl-benzyl)-proline hydrochloride 95%
- Alpha-(trifluoromethyl)-gamma-butyrolactone 95%
- Alpha-(trifluoromethyl)styrene 95%
- Aluminium trifluoroacetate 95%
- 4-Aminobenzotrifluoride 98%
- 4-Amino-3',5'-bis(trifluoromethyl)biphenyl 95%
- 5-Amino-2-(4-Boc-piperazino)benzotrifluoride 98%
- 3-Amino-2-bromobenzotrifluoride 97%
- 3-Amino-4-bromobenzotrifluoride 97%
- 3-Amino-2-bromo-6-chloropyridine 97%
- 5-Amino-6-bromo-2,2-difluorobenzodioxole 98%
- 2-Amino-4-bromo-3-fluorobenzoic acid 96%
- 2-Amino-5-bromo-4-fluorobenzoic acid 95%
- 3-Amino-5-bromo-4-fluorobenzoic acid 97%
- 2-Amino-5-bromo-4-fluorophenol 97%
- 2-Amino-6-bromo-4-fluorophenol 95%
- 2-Amino-3-bromo-5-fluoropyridine 98%
- 5-Amino-3-bromo-2-fluoropyridine 97%
- 6-Amino-3-bromo-2-fluoropyridine 98%
- 4-Amino-3-bromo-5-nitrobenzotrifluoride 98%
- 2-Amino-3-Bromo-6-trifluoromethylpyridine 98%
- 3-Amino-2-bromo-6-(trifluoromethyl)pyridine 98%
- 5-Amino-2-chloro-4-fluorobenzoic acid 98%
- 5-Amino-4-chloro-2-fluorobenzoic acid 95%
- 2-Amino-5-chloro-2'-fluorobenzophenone 98%
- 2-amino-5-chloro-4-fluorophenol 95%
- 4-Amino-2-chloro-5-fluoropyrimidine 97%
- 4-Amino-6-chloro-5-fluoropyrimidine 98%
- 2-Amino-5-chloro-3-nitrobenzotrifluoride 95%
- 2-Amino-5-chloro-3-nitrobenzotrifluoride 97%
- 4-Amino-3-chloro-5-nitrobenzotrifluoride 97%
- 3-Amino-2-chloro-6-(trifluoromethyl)pyridine 98%
- 2-Amino-4-chloro-6-(trifluoromethyl)pyrimidine 97%
- : 5-Amino-4-cyano-1-(2,5-difluorophenyl)-3-methylpyrazole 96%
- 5-Amino-4-cyano-1-(2-fluorophenyl)-3-methylpyrazole 97%
- 5-Amino-4-cyano-3-methyl-1-(2,3,5,6-tetrafluorophenyl)pyrazole 98%
- 5-Amino-4,6-dibromo-2,2-difluorobenzodioxole 96%
- 2-Amino-3,5-dibromo-4-fluorobenzoic acid 98%
- 2-Amino-3,5-dibromo-6-trifluoropyridine 95%
- 2-Amino-3,5-dichlorobenzotrifluoride 97%
- 5-Amino-1-(2,6-dichloro-4-trifluoromethylphenyl)-3-cycano pyrazole 98%
- 5-Amino-1-[2,6-dichloro-4-(trifluoromethyl)phenyl]-4-(trifluoromethane)sulfinyl-1H-pyrazole-3-carbonitrile 98%
- 2-Amino-2',4'-difluoroacetophenone hydrochloride 97%
- 4-Amino-2,2-difluoro-1,3-benzodioxole 96%
- 2-Amino-3,5-difluorobenzoic acid 97%
- 2-Amino-4,5-difluorobenzoic acid 98%
- 3-Amino-2,6-difluorobenzoic acid 95%
- 4-Amino-2,5-difluorobenzoic acid 96%
- 5-Amino-2,4-difluorobenzoic acid 97%
- 2-Amino-3,4-difluorobenzonitrile 98%
- 3-Amino-2,6-difluorobenzonitrile 98%
- 4-Amino-2,6-difluorobenzonitrile 96%
- 2-Amino-4,6-difluorobenzothiazole 98%
- 4-Amino-2,5-difluorobenzotrifluoride 98%
- 4-Amino-1-[(2R,4R,5R)-3,3-difluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,2-dihydropyrimidin-2-one 98%
- 4-Amino-2,6-difluorophenol hydrochloride 96%
- (6-Amino-2,3-difluorophenyl)acetic acid 95%
- 5-Amino-2,3-difluorophenylboronic acid 98%
- 3-Amino-4,5-difluorophenylboronic acid, pinacol ester 96%
- 2-Amino-5-(2,3-difluorophenyl)pyridine 98%
- 4-Amino-2-fluoro-5-nitrobenzonitrile 95%
- 2-Amino-3-fluorophenol 97%
- 2-Amino-4-fluorophenol 97%
- N-(2-Amino-4-fluorophenyl)methanesulfonamide .....98%
- N-(2 Amino 4 fluorophenyl)methanesulfonamide
- N-(2-Amino-5-fluorophenyl)methanesulfonamide ....98%
- 2-Amino-3-(trifluoromethoxy) benzoic acid
- 2-Amino-6- trifluoromethylbenzothiazole
- 2-Amino-5-(trifluoromethyl) nicotinaldehyde
- 2-Amino-5- trifluoromethylpyridine
- Amino-3,3,3-trifluoropropanoic acid hydrochloride
- (4R,12As)-7-(benzyloxy) -n-(2,4-difluorobenzyl)-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-[1,3]oxazino[3,2-d]pyrido[1,2-a]pyrazine-9-carboxamide
- 4-[4-Azido-2-(trifluoromethyl)benzoyl] morpholine
- (2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidine 3',5'-dibenzoate
- 3-Benzyloxy-5- bromobenzotrifluoride
- 4-(Benzyloxy)-5-bromo-2-fluorophenylboronic acid
- 1-(Benzyloxy)-2-bromo-4-(trifluoromethyl) benzene
- 1-(benzyloxy)-4-chloro-2,3- difluorobenzene
- 2-(benzyloxy)-1,5-dichloro- 3-fluorobenzene
- 1-Benzyloxy- 3-fluorobenzene
- Carboxes
- 8-Aminoquinoline-3-carboxylic acid
- 4-Acetoxymethylbenzoic acid
- 2-Acetylsulfanyl-2-methylpropionic acid ethyl ester
- 3-Acetyl-4-hydroxy-6-methyl-2h- pyran-2-one
- Acetic acid 2-ethylphenyl ester
- Acetic acid 3-buten-1-yl ester
- 2-Azaspiro[3.3]heptane-6-carboxylic acid trifluoroacetate
- Ammonium sebacate
- Ammonium 2,4-dichlorophenoxyacetate
- 8-Aminoquinoline-4-carboxylic acid
- 5-Aminopyrazolo[1,5-a]pyridine-3-carboxylic acid hydrochloride
- 5-Amino-2-phenyl-2h-1,2,3-triazole-4-carboxylic acid
- Aceclofenac ethyl ester.
- Acepromazine maleate
- 2-Acetamidoacrylic acid
- 5-Acetamidoanthranilic acid
- 2-acetamidoisophthalic acid
- 4-Acetamidobenzoic acid
- 4-Acetamidobicyclo[2.2.2]octane-1-carboxylic acid
- 2'-Acetamidobiphenyl-3-carboxylic acid
- 3'-Acetamidobiphenyl-3-carboxylic acid
- 4'-Acetamidobiphenyl-3-carboxylic acid
- 4-Acetamidobutyric acid
- 2-Acetamido-5-chlorobenzoic acid
- 4-Acetamido-5-chloro-2-methoxybenzoic acid
- 1-Acetamidocyclopentane-1-carboxylic acid
- 2-Acetamido-2-deoxy-1-thio-beta-D-glucopyranose 3,4,6-triacetate
- (1S,2S,3R,4R)-3-((S)-1-Acetamido-2-ethylbutyl)-4-guanidino-2-hydroxycyclopentane-1-carboxylic acid trihydrate
- N-(2-Acetamido)iminodiacetic acidmonosodium salt
- 4-Acetamido-2-methylbenzoic acid
- 3-Acetamido-3-methylbutanoic acid
- S-Acetamidomethyl-3-mercaptopropionic acid
- 3-Acetamido-2-methylphenyl acetate
- (4-Acetamidophenoxy)acetic acid
- 2-(2-Acetamidophenoxy)acetic acid
- 4-Acetamidophenylacetic acid ethyl ester
- 4-(4-Acetamidophenyl)-4-oxobutanoic acid
- 5-Acetamidopyridine-2-carboxylic acid
- 4-Acetamido-1h-pyrrole-2-carboxylic acid
- 3-Acetamido-2,4,6-triiodo-5-(N-methylacetamido)benzoic acid
- Acetic acid acetoxy-(4-chlorosulfonylphenyl)methyl ester
- Acetic acid 4-chloro-3-methylphenyl ester
- Acetic acid, 2-[[4-(4-cyclopropyl-1-naphthalenyl)-4h-1,2,4-triazol-3-yl]thio]-, methyl ester
- Acetic acid-d
- Acetic acid 4-ethylphenyl ester
- Acetic acid n-heptadecyl ester
- Acetic acid 5-hexen-1-yl ester
- Acetic acid, 2-hydrazinyl-2-imino-, ethyl ester
- Acetic acid 7-methoxy-4-oxo-1,4-dihydro-quinazolin-6-yl ester
- ACETIC ACID 8-NONEN-1-YL ESTER
- tert-Butyl 3-carbamimidoylazetidine-1-carboxylate acetate
- Acetic acid trans-2-hepten-1-yl ester
- Acetic acid tridecyl ester
- Acetic acid 2-vinylphenyl ester
- 3-Oxo- N- phenylbutanamide
- Acetoacetic acid n-butyl ester
- Acetoacetic acid hexyl ester
- Acetoacetic acid isoamyl ester
- Acetoacetic acid n-octyl ester
- Acetoacetic acid n-propyl ester
- Acetoacetic acid sec-butyl ester
- Acetoacetyl-l-carnitine chloride
- Acetochloro - alpha- d- mannose
- diethyl 2-(2-oxopropyl)malonate
- Diethyl 2-(2-oxopropyl)succinate
- Acetoxime benzoate
- Acetoxyacetic acid
- Acetonyl acetate
- 2-Acetylphenyl acetate
- 3-acetylphenyl acetate
- Acetoxyacetyl chloride
- 3-Acetoxybenzoic acid
- 4-Acetoxybenzoic acid
- (2-cyanophenyl) acetate
- 6-Acetoxy-n-caproic acid methyl ester
- 3-chloro-2-oxopropyl acetate
- 2-Acetoxy-2-(4-chlorophenyl)acetic acid
- 3-(4-ACETOXYPHENYL)ACRYLIC ACID
- 4-Acetoxy-3-(cyclopropylmethoxy)benzoic acid
- 2-(p-Tolyloxy)ethyl acetate
- 7-Acetoxy-1- heptene
- (R)-2-Acetoxy-3-(hexadecyloxy)propyl (2-(trimethylammonio)ethyl) phosphate
- 2,3-Dihydro-1H-inden-1-yl acetate
- 1-Chloro-2-methyl-1-oxopropan-2-yl acetate
- 4-(Acetyloxy)-3-methoxybenzoic acid
- ((2R,3S)-3-Acetoxy-6-methoxy-3,6-dihydro-2h-pyran-2-yl)methyl acetate
- 3-acetyloxy-2-methylbenzoic acid
- 7-Acetoxy-4- methylcoumarin
- (2-methylnaphthalen-1-yl) Acetate
- (Z)-4-Acetoxy-3-(4-(methylsulfonyl) phenyl)-2-phenylbut-2-enoic acid
- (2S,3S,4S,5R,6R)-6-(Acetoxymethyl)-3-(((trifluoromethyl)sulfonyl)oxy)tetrahydro-2H-pyran-2,4,5-triyl triacetate
- 6-Acetoxy-2-naphthoic acid
- 8-Acetoxy-1- octene
- 3-Acetoxy-2-phenylpropanoic acid
- (+/-)-2-Acetoxypropionic acid
- 2-Acetoxypropionyl chloride
- Succinimidyl acetate
- Acetrizoic acid
- Acetylacrylic acid methyl ester
- 5'-O- Acetyladenosine
- 3-[(Acetylamino)methyl]adamantane-1-carboxylic acid
- 4-(3-Acetylaminophenyl)benzoic acid
- 1-[3-(Acetylamino)phenyl]-5-oxopyrrolidine-3-carboxylic acid
- 1-[4-(Acetylamino)phenyl]-5-oxopyrrolidine-3-carboxylic acid
- 3-(Acetylamino)thiophene-2-carboxylic acid
- 4-(4-Acetylanilino)-4-oxobutanoic acid
- 1-Acetylazepane-4-carboxylic acid
- 1-Acetyl-azetidine-3-carboxylic acid
- 1-Acetyl-azetidine-3-carboxylic acid methyl ester
- 1-Acetylazetidin-3-yl acetate
- 2-Acetylbenzenecarbo nitrile
- 3-Acetylbenzo[f] coumarin
- 2-Acetylbenzoic acid
- 3-Acetylbenzoic acid
- 4-Acetylbenzoic acid
- 3- Acetylbenzonitrile
- 4- Acetylbenzonitrile
- 5-Acetyl-2-benzyloxy-benzoic acid
- 3'-Acetylbiphenyl-3-carboxylic acid
- 4'-Acetylbiphenyl-3-carboxylic acid
- 1'-Acetyl-[1,4'-bipiperidine]-4-carboxylic acid
- 4-Acetylbutyric acid
- 2- Acetylbutyrolactone
- Acetyl-l-carnitine hydrochloride
- N'-Acetyl-2- chloroacetohydrazide
- 4-Acetyl-2-chlorobenzoic acid
- 4-Acetyl-3-chlorobenzoic acid
- 3-Acetyl-3-chloro-dihydro-furan- 2-one
- Acetylcholine bromide
- Acetylcholine perchlorate
- 3- Acetylcoumarin
- 1-Acetyl-cyclopropanecarboxylic acid methyl ester
- 3-O-Acetyl-1,2:5,6-di-o-isopropylidene-alpha-d- glucofuranose
- 3-Acetyl-2,4-dimethyl-5- carbethoxypyrrole
- (1S,3R)-3-Acetyl-2,2-dimethylcyclobutane-1-carboxylic acid
- 5-Acetyl-2,2-dimethyl-1,3-dioxane-4,6- dione
- 4-Acetyl-3,5-dimethyl-1h-pyrrole-2-carboxylic acid
- 5-Acetyl-2,4-dimethyl-1h-pyrrole-3-carboxylic acid
- 4-O-Acetyl-3,6-di-o-(tert-butyldiphenylsilyl)-d- galactal
- 5-Acetyl-2-ethoxybenzoic acid
- 5-Acetyl-2-ethoxy-benzoic acid methyl ester
- 4-(2-Acetylhydrazino)-4-oxobutanoic acid
- 3-Acetyl-4-hydroxy-2H-chromen- 2-one
- 6-O-Acetyl-4-hydroxy-7-methoxy quinazoline
- 3-Acetylindolizine-1-carboxylic acid
- 3-Acetyl-4-isopropoxybenzoic acid
- 5'-Acetyl-2',3'- isopropylideneadenosine
- O-Acetyl mandelic acid
- 5-Acetyl-2-(4-methoxy-benzyloxy)-benzoic acid methyl ester
- 3-Acetyl-7-methoxychromen-2- one
- 4-Acetyl-2-methoxyphenyl acetate
- 5-Acetyl-2-methoxyphenyl acetate
- (5-Acetyl-2-methoxyphenyl)acetic acid
- 3-[3-Acetyl-5-(4-methoxyphenyl)-2-methyl-1h-pyrrol-1-yl]propanoic acid
- N- Acetyl-2-methylalanine
- 2-[Acetyl(methyl)amino]benzoic acid
- 1-Acetyl-2-methylazepane-2-carboxylic acid
- 1-Acetyl-4-methylazepane-4-carboxylic acid
- 4-Acetyl-2-methylbenzoic acid
- 4-Acetyl-2-methyl-benzoic acid benzyl ester
- 3-Acetyl-7-methyl-2h-chromen-2- one
- 3-Acetyl-6-methyl-2-oxo-2h-pyran-4-yl difluoridoborate
- (3-Acetyl-2-methyl-5-phenyl-1h-pyrrol-1-yl)acetic acid
- 3-(3-Acetyl-2-methyl-5-phenyl-1h-pyrrol-1-yl)propanoic acid
- 6-Acetyl-7-methylpyrazolo[1,5-a]pyrimidine-3-carboxylic acid
- 4-Acetyl-1-methyl-1h-pyrrole-2-carboxylic acid
- 1-Acetyloxy-2,5-dioxopyrrolidine-3-sulfonic acid
- (2-Acetyl-phenoxy)-acetic acid
- (3-Acetylphenoxy)acetic acid
- 4-Acetylphenoxyacetic acid
- (3-Acetyl-phenoxy)-acetic acid tert-butyl ester
- (4-Acetyl-phenoxy)-acetic acid tert-butyl ester
- 4-(4-Acetyl-phenoxy)-butyric acid
- 2-(4-Acetylphenoxy)-n,n- diethylacetamide
- 2-(4-Acetylphenoxy)-n,n- dimethylacetamide
- 5-[(2-Acetylphenoxy)methyl]-2-furoic acid
- 5-[(4-Acetylphenoxy)methyl]-2-furoic acid
- 2-(4-Acetylphenoxy)-n- phenylacetamide
- 4-Acetylphenyl acetate
- 2-([(3-Acetylphenyl)amino]carbonyl)cyclohexanecarboxylic acid
- 2-([(4 Acetylphenyl)amino]carbonyl) cyclohexanecarboxylic acid
- 6-([(3-Acetylphenyl)amino]carbonyl)cyclohex-3-ene-1-carboxylic acid
- 6-([(4-Acetylphenyl)amino]carbonyl)cyclohex-3-ene-1-carboxylic acid
- 2-(3-Acetylphenyl)benzoic acid
- 3-(2-Acetylphenyl)benzoic acid
- 4-(2-Acetylphenyl)benzoic acid
- 4-(3-Acetylphenyl)benzoic acid
- 4-(4-Acetylphenyl)benzoic acid
- 3-(3-Acetylphenyl) benzonitrile
- 3-(4-Acetylphenyl) benzonitrile
- 4-(2-Acetylphenyl)-2-chlorobenzoic acid
- 4-(4-Acetylphenyl)-2-chlorobenzoic acid
- 4-Acetylphenyl (3,4-dichlorophenyl) carbamate
- 4-Acetylphenyl dimethylcarbamate
- 2-(4-Acetylphenyl)-1,3-dioxoisoindoline-5-carboxylic acid
- 3-Acetylphenyl ethyl(methyl) carbamate
- 3-(3-Acetylphenyl)-4-fluorobenzoic acid
- 3-(4-Acetylphenyl)-4-fluorobenzoic acid
- 5-(4-Acetylphenyl)-2-furoic acid
- (4-Acetylphenyl) n-methylcarbamate
- 1-Acetyl-4-piperidineacetic acid
- 1-Acetylpiperidine-4-carbonyl chloride
- (S)-1-Acetylpiperidine-3-carboxylic acid
- 1-Acetylpiperidine-3-carboxylic acid
- 1-Acetylpiperidine-4-carboxylic acid
- 6-[(1-Acetylpiperidin-4-yl)amino]pyrimidine-4-carboxylic acid lithium salt
- 5-Acetylpyrazine-2-carboxylic acid
- 3-Acetyl-1h-pyrazole-5-carboxylic acid
- 5-Acetyl-2H-pyrazole-3-carboxylic acid ethyl ester
- 6-Acetyl-2-pyridinecarboxylic acid
- 4-Acetyl-1h-pyrrole-2-carboxylic acid
- 1-Acetyl-2- pyrrolidone
- 6-Acetylquinoline-3-carboxylic acid
- 7-Acetylquinoline-3-carboxylic acid
- Acetylsalicylic anhydride
- Acetylsalicylsalicylic acid
- 4-(Acetylsulfanyl)butanoic acid
- S-Acetylthioacetic acid
- 2-[(Acetylthio)methyl]-3-phenylpropionic acid
- (2S)-3-Acetylthio-2-methylpropionic acid
- 2-(Acetylthio)succinic anhydride
- 3'-O- Acetylthymidine
- 9-Acridinecarboxylic acid
- 2- Propenamide
- 6-Acrylamidohexanoic acid
- Acrylic acid barium salt monomer
- Acrylic acid isoamyl ester
- Acrylic acid tetradecyl ester
- Acrylic anhydride
- 4-(6-Acryloxy-hex-1-yl-oxy)phenyl 4-(hexyloxy) benzoate
- (3-Acryloxypropyl)tris (trimethylsiloxy)silane
- N- Acryloxysuccinimide
- 4- Acryloyloxybenzophenone
- 4-((6-(Acryloyloxy)hexyl)oxy)benzoic acid
- 4'-[(6-Acryloyloxy)hexyloxy]-4- biphenylcarbonitrile
- 1-Adamantaneacetic acid
- 1- Adamantanecarboxamide
- Adamantane-2-carboxylic acid
- 1-Adamantanecarboxylic acid
- Adamantane-1-carboxylic acid methyl ester
- 1,3-Adamantanediacetic acid
- 1,3-Adamantanedicarboxylic acid
- 2-Adamantanone-5-carboxylic acid
- (E)-3-((1r,3r,5R,7S)-Adamantan-2-ylidene)-4- (1-(2,5-dimethylfuran-3-yl)ethylidene)dihydrofuran-2,5-dione
- Adamantan-1-ylmethyl acetate
- [2-(1-Adamantyl)ethoxy]acetic acid
- 5-(1-Adamantyl)-2-furoic acid
- 1-Adamantyl methacrylate
- 3-(1-Adamantyl)-4-methoxybenzoic acid
- 5-(1-Adamantyl)-2-methoxybenzoic acid
- 1-(2-Adamantyl)-4-nitro-1h-pyrazole-3-carboxylic acid
- (1-Adamantyloxy)acetic acid
- [4-(1-Adamantyl)phenoxy]acetic acid
- 4-(1-Adamantyl)phenyl acetate
- 3-(1-Adamantyl)-1-phenyl-1h-pyrazole-4-carboxylic acid
- (2E)-3-[3-(1-Adamantyl)-1-phenyl-1h-pyrazol-4-yl]acrylic acid
- 3-(1-Adamantyl)propanoic acid
- 2-(1-Adamantyl)quinoline-4-carboxylic acid
- (1-Adamantylthio)acetic acid
- Adipic acid dibenzyl ester
- Adipic acid monobenzyl ester
- Adipic acid monoethyl ester
- Adipic anhydride
- Adipic dihydrazide
- Z-Aeeac-oh dcha
- Afatinib dimaleate
- Ahu-377 hemicalcium salt
- Alantola ctone
- Allyl acetoacetate
- Allyl amyl glycolate
- Allyl benzoate
- 3-Allyl-3-hydroxyazetidine-1-carboxylic acid tert-butyl ester
- 3-Allyl-4-methoxybenzoic acid
- Allyl 2-oxocyclohexanecarboxylate
- Allyl 2-oxocyclopentanecarboxylate
- 1-Allyl-5-oxopyrrolidine-3-carboxylic acid
- 3-Allyloxy-azetidine-1-carboxylic acid tert-butyl ester
- 4-(Allyloxy)-1-benzofuran-6-carboxylic acid
- 3-(Allyloxy)benzoic acid
- 4-(Allyloxy)benzoic acid
- 4-(Allyloxy) benzonitrile
- 4-Allyloxycarbonylamino-bicyclo[2.2.2]octane-1-carboxylic acid
- 1-Allyloxycarbonylamino-cyclopentanecarboxylic acid
- 1-Allyloxycarbonylamino-cyclopropanecarboxylic acid
- (2-Allyloxy-ethoxy)-acetic acid benzyl ester
- 3-Allyloxyphenylacetic acid
- 3-Allyloxypropionic acid
- 2-(4-Allyloxy-pyrazol-1-yl)-2-methyl-propionic acid
- [(4-Allyl-5-phenyl-4h-1,2,4-triazol-3-yl)thio]acetic acid
- 1-Allylpiperidine-3-carboxylic acid
- 1-Allylpiperidine-4-carboxylic acid
- Allylsuccinic anhydride
- 2-Allyl-1-(tert-butoxycarbonyl)-2,3-dihydro-1H-benzo[d]imidazole-2-carboxylic acid
- 1-Allyl 4-tert-butyl piperazine-1,4-dicarboxylate
- (Allylthio)acetic acid
- 2-(Allylthio)propanoic acid
- Aloc-o2oc-oh dcha
- Alpha- acetoxyphenylacetonitrile
- (1Alpha,8alpha,9alpha)-bicyclo[6.1.0]non-4-ene-9-carboxylic acid ethyl ester
- (1Alpha,2alpha,4alpha)-1,2,4-cyclohexanetricarboxylic acid
- Alpha,alpha'- dilaurin
- Alpha,alpha-dimethyl-gamma- butyrolactone
- Alpha- angelicalactone
- Alpha-d-cellobiose octaacetate
- Alpha-cyanocinnamic acid
- Alpha-cyanocinnamic acid ethyl ester
- alpha-Cyclopentylmandelic acid
- Alpha-cyclopentylphenylacetic acid
- Alpha-(Fmoc-amino)-omega-(succinimidyl propionate) tetra(ethylene glycol)
- Alpha-d-galacturonic acid hydrate
- 2-O-Alpha-d-glucopyranosyl-l-ascorbic acid
- Alpha-hydroxy-gamma- butyrolactone
- Alpha-ketobutyric acid sodium salt
- Alpha-ketoglutaric acid sodium salt
- H-Alpha-me-leu- oh
- (R)-2-AMINO-2,4-DIMETHYLPENTANOIC ACID
- Alpha-methoxyphenylacetic acid sodium salt
- Alpha-methylcinnamic acid
- Alpha-methylene-gamma- butyrolactone
- Alpha-methylene-gamma- valerolactone
- Alpha-methyl-gamma- butyrolactone
- Alpha-phenylcinnamic acid
- Alpha-(phenylthio)phenylacetic acid
- L-Alpha- phosphatidylinositol
- 2H-Pyran- 2-one
- D-Alpha-tocopheryl acetate
- 4-Amidinobenzoic acid hydrochloride
- 2-Aminoadamantane-2-carboxylic acid
- 3-Amino-1-adamantanecarboxylic acid
- 3-Amino-1-adamantanecarboxylic acid hydrochloride
- 6-Amino-3-aza-bicyclo[3.1.1]heptane-3-carboxylic acid
- 6-Amino-2-azaspiro[3.3]heptane-6-carboxylic acid
- 6-Amino-2-azaspiro[3.3]heptane-6-carboxylic acid dihydrochloride
- 6-Amino-2-aza-spiro[3.3]heptane-2-carboxylic acid tert-butyl ester
- 6-Amino-2-aza-spiro[3.3]heptane-2,6-dicarboxylic acid 2-tert-butyl ester
- 2-Amino-7-azaspiro[3.5]nonane-7-carboxylic acid tert-butyl ester
- 2-Amino-6-aza-spiro[3.4]octane-6-carboxylic acid tert-butyl ester
- 3-Amino-azepane-1-carboxylic acid tert-butyl ester
- 3-Aminoazetidine-3-carboxylic acid hydrochloride
- 4-Aminobenzene-1,3-dicarboxylic acid
- 4-Amino-1-benzofuran-2-carboxylic acid
- 5-Amino-1-benzofuran-2-carboxylic acid
- 5-Amino-1h-benzoimidazole-2-carboxylic acid methyl ester
- 2-Aminobenzophenone-2'-carboxylic acid
- 5-Amino-1-benzothiophene-2-carboxylic acid
- 3-(4-Aminobenzylamino)pyrrolidine-1-carboxylic acid tert-butyl ester hydrochloride
- 3-Amino-5-(4-benzyloxyphenyl)thiophene-2-carboxylic acid methyl ester
- 3-Amino-1-benzylpyrrolidine-3-carboxylic acid
- 5-Amino-1-benzyl-1,2,3,6-tetrahydro-pyridine-4-carboxylic acid ethyl ester
- 5-Amino-1-benzyl-1h-1,2,3-triazole-4-carboxylic acid
- 3-Aminobicyclo[2.2.1]hept-5-ene-2-carboxylic acid hydrochloride
- 4-Aminobicyclo[2.2.2]octane-1-carboxylic acid
- 4-Amino-bicyclo[2.2.2]octane-1-carboxylic acid cyclopropylamide, hydrochloride
- 4-Amino-bicyclo[2.2.2]octane-1-carboxylic acid cyclopropylmethyl-amide, hydrochloride
- 4-Amino-bicyclo[2.2.2]octane-1-carboxylic acid methylamide, hydrochloride
- 3-Aminobicyclo[1.1.1]pentane-1-carboxylic acid hydrochloride
- 1'-Amino-1,1'-bi(cyclopropyl)-1-carboxylic acid hydrochloride
- 2'-Aminobiphenyl-3-carboxylic acid
- 3'-Aminobiphenyl-3-carboxylic acid
- 4'-Aminobiphenyl-3-carboxylic acid
- 2'-Amino-[1,1'-biphenyl]-4-carboxylic acid hydrochloride
- 3'-Amino-[1,1'-biphenyl]-3-carboxylic acid hydrochloride
- 4'-Amino-[1,1'-biphenyl]-4-carboxylic acid hydrochloride
- 3'-Amino-biphenyl-3-carboxylic acid methyl ester hydrochloride
- (1S,3R,4R)-4-Amino-3-(boc-amino)-cyclohexanecarboxylic acid ethyl ester
- 4-Amino-1-Boc-4-piperidinedicarboxylic acid ethyl ester
- (3R,4S)-4-Amino-1-boc-3-pyrrolidinecarboxylic acid ethyl ester hydrochloride
- 2-Amino-4- bromobenzamide
- 3-Amino-6-bromo-benzofuran-2-carboxylic acid methyl ester
- 3-Amino-5-bromobenzoic acid
- 4-Amino-5-bromo-2-methoxybenzenecarboxylic acid
- 4-Amino-8-bromo-6-methylquinoline-3-carboxylic acid ethyl ester
- 5-Amino-1-(4-bromophenyl)-1H-pyrazole-4-carboxylic acid
- 2-Amino-3-bromopyrazine-5-carboxylic acid
- 3-Amino-4-bromo-1h-pyrazole-5-carboxylic acid
- 4-Amino-5-bromopyridine-2-carboxylic acid
- 3-Amino-6-bromopyridine-2-carboxylic acid ethyl ester
- 3-Amino-5-bromopyridine-2-carboxylic acid hydrochloride
- 3-Amino-5-bromo-pyridine-2-carboxylic acid isopropyl ester
- 4-Amino-6-bromoquinoline-3-carboxylic acid
- 4-Amino-8-bromoquinoline-3-carboxylic acid ethyl ester
- 3-Amino-5-bromo-thiophene-2-carboxylic acid methyl ester hydrochloride
- 2-Amino-3-carbamoyl-4,7-dihydro-5h-thieno[2,3-c]pyridine-6-carboxylic acid benzyl ester
- 2-Amino-3-carbamoyl-4,7-dihydro-5h-thieno[2,3-c]pyridine-6-carboxylic acid tert-butyl ester
- N-[3-(Aminocarbonothioyl) phenyl]cyclopropanecarboxamide
- 4-[(Aminocarbonyl)amino]benzoic acid
- (1S,2R,3R,4S)-3-(Aminocarbonyl)bicyclo[2.2.1]hept-5-ene-2-carboxylic acid
- 3-Aminocarbonyl-1-Boc- pyrrolidine
- 2-(Aminocarbonyl)cyclopropanecarboxylic acid
- 2-[4-(Aminocarbonyl)phenoxy]quinoline-4-carboxylic acid
- 3-(3-Aminocarbonylphenyl)benzoic acid
- 4-(3-Aminocarbonylphenyl)benzoic acid
- 1-[4-(Aminocarbonyl)phenyl]-5-oxopyrrolidine-3-carboxylic acid
- 5-(Aminocarbonyl)thiophene-2-carboxylic acid
- 4-Amino-4-carboxytetrahydrothiopyran hydrochloride
- 3-Amino-4-chloro-benzofuran-2-carboxylic acid methyl ester
- 4-Amino-2- chlorobenzonitrile
- 4-Amino-3-([(2-chlorobenzyl) amino]carbonyl ) isothiazole-5-carboxylic acid
- 6-Amino-5-chloro-2-cyclopropylpyrimidine-4-carboxylic acid
- 4-Amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid
- 5-Amino-1-(2-chloro-5-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-1-(4-chloro-2-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- 6-Amino-5-(4-chloro-3-methylphenyl)pyridine-3-carboxylic acid
- 1-[2-Amino-1-(2-chloro-phenyl)-ethyl]-pyrrolidine-3-carboxylic acid
- 5-Amino-1-(4-chlorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-4-chloro-1h-pyrazole-3-carboxylic acid
- 2-Amino-6-chloropyridine-4-carboxylic acid
- 3-Amino-2-chloroisonicotinic acid
- 3-Amino-5-chloropyridine-2-carboxylic acid
- 3-Amino-6-chloropyridine-2-carboxylic acid
- 5-amino-2-chloropyridine-3-carboxylic acid
- 3-Amino-5-chloropyridine-2-carboxylic acid hydrochloride
- 3-Amino-6-chloropyridine-2-carboxylic acid hydrochloride
- 2-Amino-5-chloropyridin-3- ol
- 6-Amino-5-chloro-4-pyrimidinecarboxylic acid
- 2-Amino-5-chloropyrimidine-4-carboxylic acid hydrochloride
- 3-Amino-6-chloro-thieno[3,2-b]pyridine-2-carboxylic acid methyl ester
- 4-Amino-5-chlorothiophene-2-carboxylic acid methyl ester
- 2-Amino-5- cyanobenzamide
- 3-Aminocyclobutanecarboxylic acid
- 1-Amino-cyclobutane-carboxylic acid ethyl ester hydrochloride
- 1-Aminocyclobutanecarboxylic acid hydrochloride
- 3-Aminocyclobutanecarboxylic acid hydrochloride
- 1-Amino-cyclobutane-carboxylic acid methyl ester hydrochloride
- 3-Aminocyclobutanecarboxylic acid tert-butyl ester
- 7-Amino-2-cyclobutyl[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 1-Amino-1-cycloheptanecarboxylic acid
- (1R,2R)-2-Aminocyclohexanecarboxylic acid
- (1S,2S)-2-Aminocyclohexanecarboxylic acid
- (1S,3S)-3-Aminocyclohexanecarboxylic acid
- 3-Aminocyclohexanecarboxylic acid
- (1R,3S)-3-Aminocyclohexanecarboxylic acid hydrochloride
- (1S,3R)-3-Aminocyclohexane-1-carboxylic acid hydrochloride
- (1S,3S)-3-Aminocyclohexane-1-carboxylic acid hydrochloride
- 2-Aminocyclohexane-1-carboxylic acid hydrochloride
- (1R,2S)-(+)-2-Aminocyclohex-3-enecarboxylic acid hydrochloride
- (1S,2S)-2-Aminocyclopentanecarboxylic acid
- (1R,2S)-(-)-2-Amino-1-cyclopentanecarboxylic acid hydrochloride
- (1R,3R)-3-Aminocyclopentanecarboxylic acid hydrochloride
- (1S,3S)-3-Aminocyclopentane-1-carboxylic acid hydrochloride
- (1S,4R)-4-Aminocyclopent-2-enecarboxylic acid hydrochloride
- 5-Amino-3,6-dichloropyrazine-2-carboxylic acid
- 3-Amino-2,6-dichloropyridine-4-carboxylic acid
- 1-Amino-3,3-difluorocyclobutanecarboxylic acid
- 1-Amino-4,4-difluorocyclohexane-1-carboxylic acid
- (1R)-1-Amino-2,2-difluorocyclopropane-1-carboxylic acid
- 1-Amino-2,2-difluorocyclopropane-1-carboxylic acid
- (2S,4R)-4-Amino-2-difluoromethyl-pyrrolidine-1-carboxylic acid tert-butyl ester
- 5-Amino-1-(2,4-difluorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-1-(3,5-difluorophenyl)-1h-pyrazole-4-carboxylic acid
- 2-Amino-4,7-dihydro-5-[2-[4-(carboxy)phenyl]ethyl]-4-oxo-3h-pyrrolo[2,3-d]pyrimidine hydrate
- 6-Amino-1,4-dihydro-4-oxo-2-quinolinecarboxylic acid methyl ester
- 2-Amino-7,8-dihydro-5h-pyrido[4,3-d]pyrimidine-6-carboxylic acid benzyl ester
- 2-Amino-5,8-dihydro-6h-pyrido[3,4-d]pyrimidine-7-carboxylic acid tert-butyl ester
- 2-Amino-7,8-dihydro-6h-pyrido[3,2-d]pyrimidine-5-carboxylic acid tert-butyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyran-3-carboxylic acid tert-butyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyridine-3,6-dicarboxylic acid 6-benzyl ester 3-ethyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyridine-3,6-dicarboxylic acid 6-benzyl ester 3-methyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]pyridine-3,6-dicarboxylic acid di-tert-butyl ester
- 2-Amino-4,7-dihydro-5h-thieno[2,3-c]thiopyran-3-carboxylic acid ethyl ester
- 2-Amino-5,8-dimethoxy-1,2,3,4-tetrahydronaphthalene-2-carboxylic acid
- 3-Amino-4,6-dimethylthieno[2,3-b]pyridine-2-carboxylic acid
- 2-Amino-4,5-dimethylthiophene-3-carboxylic acid Synonyms : 2-amino-4,5-dimethylthiophene-3-carboxylic acid
- 2-Amino-4,5-dimethyl-thiophene-3-carboxylic acid methyl ester
- 1-(2-Aminoethyl)cyclopropanecarboxylic acid
- 1-(2-Aminoethyl)cyclopropane-1-carboxylic acid hydrochloride
- 4-(2-Aminoethyl)-5-(2-furyl)-1,2-dihydro-3h- pyrazol-3-one
- 2-Amino-4-ethyl-5-methylthiophene-3-carboxylic acid methyl ester
- 2-Amino-5-ethylthiophene-3-carboxylic acid methyl ester
- 4-Amino-2-ethylthiopyrimidine-5-carboxylic acid
- 4-Amino-2-(ethylthio)-5-pyrimidinecarboxylic acid ethyl ester
- 1-(2-Aminoethyl)-1h-1,2,3-triazole-4-carboxylic acid hydrochloride
- 2-Amino-5- fluorobenzamide
- 3-Amino-4-fluoro-benzofuran-2-carboxylic acid methyl ester
- 3-Amino-6-fluoro-benzofuran-2-carboxylic acid methyl ester
- 3-Amino-2-fluorobenzoic acid
- 4-Amino-6-fluorocinnoline-3-carboxylic acid
- 1-Amino-3-fluoro-cyclobutanecarboxylic acid
- (2R,4R)-4-Amino-6-fluoro-2-methylchroman-4-carboxylic acid
- 3-Amino-5-(3-fluoro-2-methylphenyl)benzoic acid
- 1-[2-Amino-1-(4-fluoro-phenyl)-ethyl]-pyrrolidine-3-carboxylic acid
- 5-Amino-1-(2-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- 5-Amino-1-(4-fluorophenyl)-1h-pyrazole-4-carboxylic acid
- (1R,3S,4S)-3-Amino-4-hydroxy-cyclohexanecarboxylic acid ethyl ester hydrochloride
- (1S,3R,4R)-3-Amino-4-hydroxy-cyclohexanecarboxylic acid ethyl ester hydrochloride
- 2-Amino-4-hydroxy-7,8-dihydro-5h-pyrido[4,3-d]pyrimidine-6-carboxylic acid tert-butyl ester
- 5-Amino-1-(2-hydroxyethyl)pyrazole-4-carboxylic acid
- 1-Amino-3-(hydroxymethyl)cyclobutane-1-carboxylic acid
- 4-Amino-2-hydroxypyrimidine-5-carboxylic acid hydrobromide
- 3-Amino-3-imino-propanoic acid, ethyl ester hydrochloride
- 1-Aminoindan-1-carboxylic acid
- 2-Amino-2-indancarboxylic acid
- 3-Amino-1H-indazole-5-carboxylic acid
- 3-Amino-1h-indazole-4-carboxylic acid
- 3-Amino-1h-indazole-6-carboxylic acid
- 5-Amino-1H-indazole-3-carboxylic acid
- 6-Amino-1h-indazole-7-carboxylic acid
- 4-Amino-5-iodo-2-methoxybenzenecarboxylic acid
- 3-Amino-6-iodopyrazine-2-carboxylic acid
- 5-Amino-6-iodo-2-pyrazinecarboxylic acid
- N-(6-Amino-1-isobutyl-2,4-dioxo-1,2,3,4-tetrahydro-pyrimidin-5-yl) -4-chloro-n-(2-methoxy-ethyl)-butyramide
- 3-Aminoisoquinoline-6-carboxylic acid
- 4-Aminoisoquinoline-6-carboxylic acid
- 4-Aminoisoquinoline-8-carboxylic acid
- 7-Aminoisoquinoline-4-carboxylic acid
- 8-Aminoisoquinoline-5-carboxylic acid
- 3-Aminoisoquinoline-8-carboxylic acid hydrochloride
- 4-Amino-2-mercaptopyrimidine-5-carboxylic acid
- 3-Amino-4- methanesulfonylbenzonitrile
- 2-Amino-5-(4-methoxy-phenylcarbamoyl)-4-methyl-thiophene-3-carboxylic acid ethyl ester
- 1-[2-Amino-1-(2-methoxy-phenyl)-ethyl]-pyrrolidine-3-carboxylic acid
- 4-Amino-1-(4-methoxyphenyl)-1h-pyrazole-3-carboxylic acid
- 5-Amino-1-(4-methoxyphenyl)-1h-pyrazole-4-carboxylic acid
- 6-Amino-5-methoxy-pyridine-2-carboxylic acid methyl ester
- 4-Amino-6-methoxyquinoline-3-carboxylic acid ethyl ester
- 2-Amino-6-methoxy-1,2,3,4-tetrahydro-naphthalene-2-carboxylic acid
- 2-Amino-8-methoxy-1,2,3,4-tetrahydro-naphthalene-2-carboxylic acid
- 2-Aminomethyl-7-azaspiro[3.5]nonane-7-carboxylic acid tert-butyl ester
- 2-Amino-3- methylbenzamide
- 3-(Aminomethyl)cyclobutanecarboxylic acid hydrochloride
- 1-Aminomethyl-cyclohexanecarboxylic acid methyl ester hydrochloride
- 1-Aminomethyl-3,4-dihydro-1h-isoquinoline-2-carboxylic acid tert-butyl ester oxalate
- 3-Aminomethyl-3-fluoropiperidine-1-carboxylic acid tert-butyl ester
- 5-(Aminomethyl)furan-2-carboxylic acid hydrochloride
- 5-Amino-1-methyl-1h-indazole-3-carboxylic acid
- (R)-3-Aminomethyl-morpholine-4-carboxylic acid tert-butyl ester
- 3-Aminomethyl-morpholine-4-carboxylic acid tert-butyl ester
- 1-Aminomethyl-2-oxa-5-aza-bicyclo[2.2.1]heptane-5-carboxylic acid tert-butyl ester
- 7-Aminomethyl-6-oxa-2-aza-spiro[3.4]octane-2-carboxylic acid tert-butyl ester
- 5-Amino-1-(4-methylphenyl)-1H-pyrazole-4-carboxylic acid
- 4-Amino-1-methyl-4-piperidinecarboxylic acid
- 4-Amino-1-methylpiperidine-4-carboxylic acid hydrochloride
- 4-Amino-1-methyl-4-piperidinecarboxylic acid methyl ester
- 3-Amino-5-methylpyrazine-2-carboxylic acid
- 3-Amino-1-methyl-1h-pyrazole-4-carboxylic acid
- 3-Amino-1-methyl-1h-pyrazole-5-carboxylic acid
- 4-Amino-1-methyl-1h-pyrazole-3-carboxylic acid
- 4-Amino-3-methyl-1h-pyrazole-5-carboxylic acid
- 5-Amino-1-methyl-1h-pyrazole-3-carboxylic acid
- 4-Amino-1-methyl-1h-pyrazole-5-carboxylic acid hydrochloride
- 4-Amino-3-methyl-pyrazole-1-carboxylic acid tert-butyl ester
- 5-(Aminomethyl)pyrazolo[1,5-a]pyridine-3-carboxylic acid hydrochloride
- 7-Amino-2-methylpyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 7-Amino-3-methylpyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 2-Amino-4-methylpyridine-3-carboxylic acid
- 2-Amino-6-methyl-3-pyridinecarboxylic acid
- 3-Amino-2-methylpyridine-5-carboxylic acid
- 3-Amino-4-methylpyridine-2-carboxylic acid
- 3-Amino-5-methyl-pyridine-2-carboxylic acid amide
- 5-Amino-6-methyl-3-pyridinecarboxylic acid ethyl ester
- 5-Amino-2-methylpyridine-4-carboxylic acid hydrochloride
- 2-Amino-4-methyl-pyrimidine-5-carboxylic acid
- 4-Amino-6-methyl-pyrimidine-2-carboxylic acid methyl ester
- 4-Amino-1-methyl-1h-pyrrole-2-carboxylic acid hydrochloride
- 2-Aminomethyl-pyrrolidine-1-carboxylic acid benzyl ester
- 2-Amino-7-methyl-7h-pyrrolo[2,3-d]pyrimidin-6-carboxylic acid methyl ester
- 7-Amino-2-(methylsulfanyl)[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 3-(Aminomethyl)-1-[(tert-butoxy)carbonyl]azetidine-3-carboxylic acid hydrochloride
- 4-Aminomethyltetrahydropyran-4-carboxylic acid
- 4-(Aminomethyl)tetrahydro-2h-pyran-4-carboxylic acid methyl ester hydrochloride
- 2-Amino-5-methylthiophene-3-carboxylic acid
- 3-Amino-4-methylthiophene-2-carboxylic acid
- 4-Amino-5-methylthiophene-3-carboxylic acid
- 4-Amino-2-(methylthio)pyrimidine-5-carboxylic acid
- 7-Amino-2-methyl[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 5-Amino-1-methyl-3-(trifluoromethyl)-1h-pyrazole-4-carboxylic acid
- 2-Amino-3- nitrobenzonitrile
- 3-Amino-5-(3-nitro-phenyl)-thiophene-2-carboxylic acid ethyl ester
- 3-Amino-7-oxabicyclo[2.2.1]hept-5-ene-2-carboxylic acid hydrochloride
- 1-Amino-4-oxocyclohexanecarboxylic acid ethylene ketal
- 3-Amino-5-oxo-4,5-dihydro-pyrazine-2-carboxylic acid methyl ester
- 6-Amino-4-oxo-1,4-dihydroquinoline-2-carboxylic acid
- 2-Amino-4-oxo-1,5,7,8-tetrahydro-4h-pyrido[4,3-d]pyrimidine-6-carboxylic acid tert-butyl ester
- 4-(4-Aminophenethyl)piperazine-1-carboxylic acid tert-butyl ester
- 3-Amino-5-phenylbenzoic acid
- 2-(3-Aminophenyl)-6-bromoquinoline-4-carboxylic acid
- 2-(3-Aminophenyl)-6-chloroquinoline-4-carboxylic acid
- 2-(4-Aminophenyl)-6-chloroquinoline-4-carboxylic acid
- 1-(3-Amino-phenyl)-cyclopropanecarboxylic acid amide
- 2-(3-Aminophenyl)-6,8-dimethylquinoline-4-carboxylic acid
- 1-(3-Aminophenyl)-5-oxo-4,5-dihydro-1h-pyrazole-3-carboxylic acid
- 1-(4-Aminophenyl)-2-oxopyridine-3-carboxylic acid
- 4-Amino-1-phenyl-1h-pyrazole-3-carboxylic acid
- 5-Amino-1-phenyl-1h-pyrazole-4-carboxylic acid
- 2-Amino-4-phenylpyrimidine-5-carboxylic acid
- 2-(3-Aminophenyl)quinoline-4-carboxylic acid
- 2-(4-Amino-phenyl)-quinoline-4-carboxylic acid
- 3-Amino-5-phenylthiophene-2-carboxylic acid
- 4-Aminopiperidine-4-carboxylic acid
- 1-Aminopropan-2-one hydrochloride
- 4-(3-Amino-propionyl)-piperazine-1-carboxylic acid tert-butyl ester
- 1-(3-Aminopropyl)-5-tert-butyl-2-methylpyrrole-3-carboxylic acid
- 3-Amino-1H-pyrazole-5-carboxylic acid
- 5-Amino-1h-pyrazole-4-carboxylic acid
- 5-Amino-1h-pyrazole-3-carboxylic acid hydrochloride
- 5-Amino-pyrazole-1,4-dicarboxylic acid 1-tert-butyl ester 4-ethyl ester
- 2-Aminopyrazolo[1,5-a]pyrimidine-3-carboxylic acid
- 7-Aminopyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 3-(5-Amino-1h-pyrazol-3-yl)-azetidine-1-carboxylic acid tert-butyl ester
- 5-Amino-pyridazine-4-carboxylic acid
- 6-Aminopyridazine-3-carboxylic acid
- 6-Aminopyridine-2-carboxylic acid ethyl ester
- 4-Aminopyridine-2-carboxylic acid monohydrate
- 4-Aminopyridine-2,6-dicarboxylic acid
- 4-(6-Amino-pyridin-3-yloxy)-pyridine-2-carboxylic acid methylamide
- 5-Amino-pyrimidine-4-carboxylic acid
- 3-Amino-pyrrolidine-1,3-dicarboxylic acid 1-tert-butyl ester
- 5-Amino-1h-pyrrolo[2,3-b]pyridine-2-carboxylic acid
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-5-carboxylic acid
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-6-carboxylic acid
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-5-carboxylic acid methyl ester
- 2-Amino-7h-pyrrolo[2,3-d]pyrimidin-6-carboxylic acid methyl ester
- 4-Amino-7h-pyrrolo[2,3-d]pyrimidine-5-carboxylic acid
- 2-Aminoquinoline-3-carboxylic acid
- 2-Aminoquinoline-6-carboxylic acid
- 3-Aminoquinoline-5-carboxylic acid
- 3-Aminoquinoline-6-carboxylic acid
- 3-Aminoquinoline-7-carboxylic acid
- 3-Aminoquinoline-8-carboxylic acid
- 4-Aminoquinoline-6-carboxylic acid
- 5-Aminoquinoline-3-carboxylic acid
- 7-Aminoquinoline-5-carboxylic acid
- 3-Amino-1-(tert-butoxycarbonyl)-3-azetidinecarboxylic acid
- 5-Amino-2,3,4,5-tetrahydro-benzo[b]azepine-1-carboxylic acid tert-butyl ester
- 2-Amino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylic acid
- 2-Amino-4,5,6,7-tetrahydro-benzo[b]thiophene-3-carboxylic acid isopropyl ester
- 2-Amino-4,5,6,7-tetrahydro-benzo[b]thiophene-3-carboxylic acid methyl ester
- 2-Amino-4,5,6,7-tetrahydro-benzo[b]thiophene-3-carboxylic acid tert-butyl ester
- 3-Aminotetrahydrofuran-3-carboxylic acid
- 3-Aminotetrahydro-3-furancarboxylic acid butyl ester
- 1-Amino-1,2,3,4-tetrahydro-1-naphthalenecarboxylic acid hydrochloride
- 2-Amino-1,2,3,4-tetrahydro-naphthalene-2-carboxylic acid hydrochloride
- 3-Amino-5,6,7,8-tetrahydrothieno[2,3-b]quinoline-2-carboxylic acid
- 3-Amino-tetrahydro-thiophene-3-carboxylic acid
- 2-Amino-6h-[1,3,4]thiadiazine-5-carboxylic acid ethyl ester hydrochloride
- 2-Amino-6h-1,3,4-thiadiazine-5-carboxylic acid hydrochloride
- 4-Aminothieno[3,2-d]pyrimidine-7-carboxylic acid
- 5-Aminothieno[2,3-d]pyrimidine-6-carboxylic acid
- 4-Aminothiophene-2-carboxylic acid
- 3-Aminothiophene-2-carboxylic acid potassium salt
- 3-Amino-thiophene-2-carboxylic acid sodium salt
- 3-Amino-3- thioxopropanamide
- 6-Amino-[1,2,4]triazine-5-carboxylic acid
- 5-Amino-4h-1,2,4-triazole-3-carboxylic acid
- 3-Amino[1,2,4]triazolo[4,3-a]pyridine-6-carboxylic acid
- 7-Amino[1,2,4]triazolo[1,5-a]pyrimidine-6-carboxylic acid
- 6-(5-Amino-1h-1,2,4-triazol-3-yl)cyclohex-3-ene-1-carboxylic acid
- Amino-tri-(carboxyethoxymethyl)- methane
- 4-Amino-3,5,6-trichloropyridine-2-carboxylic acid
- 5-Amino-1-[4-(trifluoromethoxy)phenyl]-1h-pyrazole-4-carboxylic acid
- 2-Amino-3,3,3-trifluoropropanoic acid hydrochloride
- 2-[(3-Amino-2,4,6-triiodophenyl)methyl]butanoic acid
- (1R,2R,3S,5R)-2-Amino-2,6,6-trimethyl-bicyclo[3.1.1]heptane-3-carboxylic acid methyl ester hydrochloride
- Ammonium adipate
- Ammonium 2-(1,3-benzodioxol-5-yloxy)-2-methylpropanoate
- Ammonium carbamate
- Ammonium citrate dibasic
- Ammonium dodecanedioate
- Ammonium ([2-(2-furyl)cyclohexyl]oxy)acetate
- Ammonium hydrogen maleate
- Ammonium succinate
- Amphotericin b
- Angelic acid
- Angelic acid isoamyl ester
- Angelic acid isobutyl ester
- Angelic anhydride
- 1,5-Anhydro-3-o-benzoyl-4,6-o-benzylidene-2-deoxy- 2-(n4-benzoylcytidin-1-yl)-d-altro-hexitol
- 2-Anilinobenzoic acid methyl ester
- 1[(Anilinocarbonyl)amino]cyclopropanecarboxylic acid
- 4-[5-(Anilinocarbonyl)-1,3,4-thiadiazol-2-yl]butanoic acid
- 3-[5-(Anilinocarbonyl)-1,3,4-thiadiazol-2-yl]propanoic acid
- 2-Anilinonicotinic acid
- Ansamitocin p 3'
- 1-Anthracenecarboxylic acid
- 2-Anthracenecarboxylic acid
- 9-Anthracenecarboxylic acid
- (1R,2R)-2-(Anthracene-2,3-dicarboximido) cyclohexanecarboxylic acid
- (1S,2S)-2-(Anthracene-2,3-dicarboximido) cyclohexanecarboxylic acid
- 9-Anthracenylmethyl acrylate
- 9-Anthracenylmethyl methacrylate
- Anthraquinone-2-carboxylic acid
- Anthraquinone-2,3-dicarboxylic acid
- Anti-3-oxotricyclo[2.2.1.0(2,6)]heptane-7-carboxylic acid
- D-Araboascorbic acid
- Arachidic acid
- Arachidic acid ethyl ester
- Arachidic acid methyl ester
- Arachidic anhydride
- (3Ar*,6ar*)-benzylhexahydropyrrolo [3,2-b]pyrrole-1(2h)-carboxylate
- (3S,3Ar,5r,6ar,7r)-6-iodo-2-oxooctahydro-3,5-methanocyclopenta[b]pyrrole-7-carboxylic acid
- (4Ar,8ar)-rel-1-methyl-decahydro-1,5-naphthyridine, oxalic acid
- (3aR,6aR)-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-1(2H)-carboxylate
- (3aR,6aR)-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-5(1H)-carboxylate
- (4Ar,9ar)-tert-butyl octahydro-[1,4]oxazino[3,2-c]azepine-4(4ah)-carboxylate
- (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-6,10,10b-Trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-3-vinyldodecahydro-1H-benzo[f]chromen-5-yl acetate
- (3S,3Ar,5s,6as,7s)-2-oxooctahydro-3,5-methanocyclopenta[b]pyrrole-7-carboxylic acid
- (3Ar,5s,6as)-tert-butyl 5-hydroxyhexahydrocyclopenta [c]pyrrole-2(1H)-carboxylate
- (3Ar,6as)-tert-butyl 5-oxohexahydrocyclopenta [c]pyrrole-2(1h)-carboxylate
- (4Ar,7as)-tert-butyl tetrahydro-2h-[1,4]dioxino[2,3-c]pyrrole-6(3h)-carboxylate
- Ar-13324 hydrochloride
- (1S,7S,8S,8aR)-8-(2-((2R,4R)-4-Hydroxy-6-oxotetrahydro-2H-pyran-2-yl)ethyl)-7-methyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl (S)-2-methylbutanoate
- (3S,11Ar)-6-methoxy-3-methyl-5,7-dioxo-2,3,5,7,11,11a-hexahydrooxazolo[3,2-d]pyrido[1,2-a]pyrazine-8-carboxylic acid
- (9Ar)-octahydro-1h-pyrido[1,2-a]piperazine oxalic acid
- (6S,8Ar)-3-oxooctahydroindolizine-6-carboxylic acid
- (3aS,9aR,10aR,10bS,E)-6,9a- Dimethyl-3-methylene-3a,4,5,8,9,9a,10a,10b-octahydrooxireno[2',3':9,10]cyclodeca[1,2-b]furan-2(3H)-one
- (3As,4r,5s,6ar)-(+)-hexahydro-5-hydroxy- 4-(hydroxymethyl)-2h-cyclopenta[b]furan-2-one
- (3As,4s,6ar)-4-methoxytetrahydrofuro [3,4-b]furan-2(3h)-one
- (1R,4As,10ar)-methyl 7-isopropyl-1,4a-dimethyl-1,2,3,4,4a,9,10,10a-octahydrophenanthrene-1-carboxylate
- (2R,3aS,6S,6aS,7R,10R,11aR,11bS)-2-(Furan-3-yl)- 11b-methyloctahydro-4H-3a,6:7,10-dimethanofuro[2,3-c]oxepino[4,5-e]oxepine-4,8(6H)-dione
- (3As,5r,6s,6as)-6-hydroxy-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxole-5-carboxylic acid Synonyms : (3aS,5R,6S,6aS)-6-hydroxy-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxole-5-carboxylic acid
- (4As,8as)-tert-butyl hexahydro-1h-pyrido[3,4-b][1,4]oxazine-6(7h)-carboxylate
- (3As*,6as*)-tert-butylhexahydropyrrolo [3,4-c]pyrrole-2(1H)-carboxylate
- (3aS,6aS)-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-5(1H)-carboxylate
- L-Ascorbic acid
- L-Ascorbic acid 2-phosphate sesquimagnesium salt hydrate
- L-Ascorbyl 2,6-dibutyrate
- L-Ascorbyl 2,6-dipalmitate
- (4R,12As)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2h-pyrido[1',2':4,5]pyrazino[2,1-b][1,3]oxazine-9-carboxylic acid
- (9As)-octahydro-1h-pyrido[1,2-a]piperazine, oxalic acid
- Atractyloside potassium salt
- Atracurium oxalate
- Atropic acid
- 7- Geranyloxycoumarin
- Aurintricarboxylic acid
- AVERMECTIN B1A
- 7-Azabicyclo[2.2.1]heptane-2-carboxylic acid ethyl ester hydrochloride
- (1R*,2S*,4S*)-7-Aza-bicyclo[2.2.1]heptane-2-carboxylic acid hydrobromide
- 2-Azabicyclo[2.2.1]heptane-6-carboxylic acid hydrochloride
- 2-Azabicyclo[3.1.1]heptane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.1]heptane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[4.1.0]heptane-6-carboxylic acid hydrochloride
- 3-Azabicyclo[4.1.0]heptane-3-carboxylic acid, 6-(hydroxymethyl)-, 1,1-dimethylethyl ester
- 2-Aza-bicyclo[2.2.1]heptane-2,6-dicarboxylic acid 2-tert-butyl ester
- 2R-7-Aza-bicyclo[2.2.1]heptane-2,7-dicarboxylic acid 7-tert-butyl ester
- 2-Azabicyclo[2.2.1]heptane-2-propanoic acid
- 2-Azabicyclo[2.2.1]hept-5- en-3-one
- 3-Azabicyclo[3.1.0]hexane-1-carboxylic acid
- (1R,5S,6S)-3-Azabicyclo[3.1.0]hexane-6-carboxylic acid hydrochloride
- 2-Azabicyclo[2.1.1]hexane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.0]hexane-1-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.0]hexane-6-carboxylic acid hydrochloride
- 3-Azabicyclo[3.1.0]hexane-1-carboxylic acid, methyl ester, hydrochloride (1:1)
- 2-Azabicyclo[2.1.1]hexane-2,5-dicarboxylic acid 2-tertbutyl ester
- 8-Azabicyclo[3.2.1]octane-8-carboxylic acid, 3-(hydroxymethyl)-, 1,1-dimethylethyl ester, (3-endo)-
- 2-Azabicyclo[2.2.2]octane-2-carboxylic acid, 5-oxo-1,1-dimethylethyl ester
- 8-Azabicyclo[3.2.1]octane-3,8-dicarboxylic acid, 8-(1,1-dimethylethyl) ester, (3-endo)-
- 2-Azabicyclo[2.2.2]octane-2,6-dicarboxylic acid 2-tertbutyl ester
- 2-Azabicyclo[2.2.2]oct-7-ene-6-carboxylic acid
- 1-Azabicyclo[2.2.2]oct-2-ene-3-carboxylic acid methyl ester hydrochloride
- Azadibenzocyclooctyne acid
- 2-Azaspiro[4.5]decane-7-carboxylic acid
- 5-Azaspiro[2.4]heptane-1-carboxylic acid
- 2-Azaspiro[3.3]heptane-2-carboxylic acid tert-butyl ester
- 1-Azaspiro[3.3]heptane hemioxalate
- 2-Azaspiro[3.3]heptane hemioxalate
- 4-Azaspiro[2.4]heptane hemioxalate
- 2-(5-Azaspiro[2.4]heptan-7-yl)acetic acid
- 5-Azaspiro[2.3]hexane hemioxalate
- 2-Azaspiro[4.4]nonane-2-carboxylic acid, 7-hydroxy-, 1,1-dimethylethyl ester
- 2-Aza-spiro[4.4]nonane hemioxalate
- 2-Aza-spiro[4.4]non-7-ene-2-carboxylic acid tert-butyl ester
- 2-Azaspiro[3.4]octane hemioxalate
- 5-Azaspiro[3.4]octane hemioxalate
- 6-Azaspiro[3.4]octane hemioxalate
- 5-Azaspiro[3.4]octane oxalate
- 3-Azaspiro[5.5]undecane-3,9-dicarboxylic acid 3-(tert-butyl) ester
- Azaerythromycin A
- Azelaic acid monomethyl ester
- 3-(1-AZEPANYL)BUTANOIC ACID
- (([5-(Azepan-1-ylcarbonyl)-1,2,4-oxadiazol-3-yl]methyl)thio)acetic acid
- 3-(([5-(Azepan-1-ylcarbonyl)-1,2,4-oxadiazol-3-yl]methyl)thio)propanoic acid
- 2-Azepan-1-ylisonicotinic acid
- (2E)-3-[3-(Azepan-1-ylmethyl)-4-methoxyphenyl]acrylic acid
- 3-Azepan-1-yl-2-methylpropanoic acid
- Azepan-1-yl(oxo)acetic acid
- 3-Azepan-1-ylpropanoic acid
- 5-(Azepan-1-ylsulfonyl)-2-methylbenzoic acid
- Azetidine-3-carboxylic acid
- 1-Azetidinecarboxylic acid, 3-fluoro-3-(hydroxymethyl)-, phenylmethyl ester
- 1-Azetidinecarboxylic acid, 3-[4-(phenylmethyl)-1-piperazinyl], 1,1-dimethylethyl ester
- 1,3-Azetidinedicarboxylic acid, 3-amino-, 1-(1,1-dimethylethyl) 3-methyl ester
- 2-Azetidinepropanoic acid, 2-(methoxycarbonyl)-1-(phenylmethyl)-, methyl ester
- Azetidine-1,3,3-tricarboxylic acid 1-tert-butyl ester 3-ethyl ester
- 2- Azetidinone
- Azetidin-3-yl acetate
- Azetidin-3-yl-acetic acid
- 2-(Azetidin-3-yl)acetic acid hydrochloride
- 1-(Azetidin-3-yl)azetidin-3-ol oxalate
- 2-(Azetidin-3-yl)-2-methylpropanoic acid
- 3-(Azetidin-1-yl)propanoic acid
- 4-(Azetidin-3-yl)tetrahydro-2h-pyran-2-one hydrochloride
- 1-Azido-1-deoxy-beta-d-galactopyranoside tetraacetate
- O-(2-Azido-2-deoxy-3, 4,6-tri-o-acetyl-beta-d-galactopyranosyl)-trichloroacetimidate
- 2-[2-(2-Azidoethoxy)ethoxy]ethyl 2,3,4,6-tetra-o-acetyl-d-galactopyranoside
- Azido-peg12- acid
- 2-(Azido-peg3-amido)-1,3-bis(carboxylethoxy) propane
- 4-Azidosulfonyl-benzoic acid 2,5-dioxo-pyrrolidin-1-yl ester
- 11-Azido-3,6,9-trioxaundecanoic acid
- Azilsartan medoxomil
- 2-(((3-(Aziridin-1-yl)propanoyl)oxy)methyl)-2-ethylpropane-1,3-diyl bis(3-(aziridin-1-yl)propanoate)
- Azobenzene-4,4'-dicarboxylic acid
- Azobenzene-4,4'-dicarboxylic acid dimethyl ester
- 3-Azocan-1-ylpropanoic acid hydrochloride
- 3,3'-Azodibenzoic Acid
- Azoxybenzene-4,4'-dicarboxylic acid diethyl ester
- Balofloxacin dihydrate
- Bapta tetraethyl ester
- Docosanoic acid
- Behenic Anhydride
- Behenyl acrylate
- Bendamustine hydrochloride hydrate
- Benzal diacetate
- Benzamidine hydrochloride
- 2-Benzamidoethyl benzoate
- 2-(3-Benzamidophenyl)acetic acid
- (Z)-N'-Hydroxybenzimidamide hydrobromide
- Benzeneacetic acid, alpha,alpha-diphenyl-, rhodium(2+) salt (2:1)
- 1,4-Benzenediacrylic acid
- 1,2-Benzenedicarboxylic acid,4-chloro-,1,2-dimethyl ester
- Benzene-1,2,3,4,5,6-hexacarboxylic acid
- Benzenepentacarboxylic acid
- Benzenepropanoic acid, alpha-(hydroxymethyl)-, ethyl ester
- Benzenepropanoic acid, 4-methoxy-3-(3-methoxypropoxy)-a-(1-methylethyl)-, (ar)-
- 5-Benzenesulfonyl-5h-pyrrolo[3,2-d]pyrimidine-6-carboxylic acid
- 1,2,3-Benzenetricarboxylic acid
- 1,2,4-Benzenetricarboxylic acid
- 1,2,3-Benzenetricarboxylic acid hydrate
- 1,2,4-Benzenetricarboxylic acid tripropyl ester
- 1-Benzhydrylazetidin-3-yl 2-cyanoacetate
- 1H-Benzimidazole-1-carboxylic acid, 2,3-dihydro-2-oxo-, 1,1-dimethylethyl ester
- 1H-Benzimidazole-7-carboxylic acid, 1-[[2'-(2,5-dihydro-5-oxo-1,2,4-oxadiazol-3-yl)[1,1'-biphenyl]-4-yl]methyl]-2-ethoxy-, methyl ester
- Benzimidazole-5,6-dicarboxylic acid
- 3-(1H-Benzimidazol-1-yl)butanoic acid
- 3-(1H-Benzimidazol-2-yl)-2H-chromen -2-one
- 3-(1H-Benzimidazol-2-yl)-3-hydroxypropanoic acid
- Imidazoles
- 5-Amino-1,4-dimethylimidazole hydrochloride
- 5-Amino-1-((3ar,4r,6r,6ar)-6-(hydroxymethyl)-2,2-dimethyltetrahydrofuro[3,4-d][1,3]dioxol-4-yl)- 1h-imidazole-4-carboxamide
- 2- Aminobenzimidazole
- Ac-his-oh h2o
- 1-Aminobenzimidazole-2-sulfonic acid
- Acyclovir-side chain-2-3h
- 1-(4-Aminobenzyl)-n-[3-(trifluoromethyl)phenyl]- 1h-imidazole-4-carboxamide
- 2-Amino-1-Boc -imidazole
- 2-Amino-1h-benzo[d]imidazole- 5-carbonitrile
- 1-(1-Adamantyl)- 1h-benzimidazole-2-thiol
- 1-(4-Aminobenzyl)-n-(2,4-dimethylphenyl)- 1h-imidazole-4-carboxamide
- 2-[(2-Allylphenoxy)methyl] -1h-benzimidazole
- Acalabrutinib .
- 5-Amino-1h-imidazol- 4-carbonitrile
- Allylchloro[1,3-bis(2,6-di-i-propylphenyl) -4,5-dihydroimidazol-2-ylidene]palladium(ii)
- 1- Allylhydantoin
- 1-(5-Acetyl-4-methylthiazol- 2-yl)imidazolidin-2-one
- Allyl 1h-imidazole-1-carboxylate
- 2-amino-9-{[(1,3-dihydroxypropan-2-yl)oxy]methyl}-6, 9-dihydro-1H-purin-6-one
- 1-(4-Aminobenzyl)-n-(4-ethylphenyl)- 1h-imidazole-4-carboxamide
- N-Alpha-methyl-l-histidine hydrochloride
- N-Acetyl-l- carnosine
- 3-Amino-n-cyclopropyl-4-(1h-imidazol-1-yl) benzamide
- 1-(4-Aminobenzyl)-n-(3,4-dimethoxyphenyl)-1h-imidazole- 4-carboxamide
- 1-Allyl-3-methylimidazolium chloride
- 1-(4-Aminobenzyl)-n-(3-methoxyphenyl)- 1h-imidazole-4-carboxamide
- 1-(4-Aminobenzyl)-n-(2-methoxy-5-methylphenyl)- 1h-imidazole-4-carboxamide
- Z-Ala-his -ome
- 5- Aminobenzimidazolone
- 2-Amino-9-((1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylenecyclopentyl)-1,9-dihydro-6H-purin-6-one hydrate
- 2-Amino-9-[(1S,3R,4S)-4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl]-6, 9-dihydro-1H-purin-6-one
- Ac-His(1-Trt)- OH
- 1-Acetyl- 2-thiohydantoin
- (R)-(-)-Alpha-methylhistamine dihydrochloride
- Alpha-[5-(1,2-dithiolan-3-yl)pentamido]-omega-biotinyl undeca(ethylene glycol)
- Alpha-(2,4-dichlorophenyl) -1h-imidazole-1-ethanol
- 1-(4-Aminobenzyl)-n-(5-chloro-2-methylphenyl)- 1h-imidazole-4-carboxamide
- 1-(4-Aminobenzyl)-n-phenyl-1h-imidazole- 4-carboxamide
- 2-Amino-7-benzyl-1h-purin-6(7h)- one
- 2-Amino-9-(4-hydroxy-3-(hydroxymethyl)butyl)-1,9-dihydro-6H-purin-6- one
- 3-Amino-1,3-diaza-spiro[4.5]decane-2, 4-dione
- 1-(4-Aminobenzyl)-n-(2,4-difluorophenyl)- 1h-imidazole-4-carboxamide
- 3-Amino-5-ethyl-5-methylimidazolidine-2, 4-dione
- 2-Amino-9-((2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-1h-purin-6(9h)-one dihydrate
- 2-Amino-5, 6-dimethylbenzimidazole
- 1-Allyl-3-methylimidazolium dicyanamide
- 3-Amino-5,5-diphenylimidazolidine-2, 4-dione
- N-(6-Aminohexyl)-5-((3as,4s,6ar)-2-oxohexahydro-1h-thieno[3,4-d]imidazol-4-yl) pentanamide
- 2-Amino-9-[(2-hydroxyethoxy)methyl]- 6,9-dihydro-1H-purin-6-one
- 3-Acetyl-2-methylimidazo [1,2-a]pyridine
- 2-Amino-5, 6-dichlorobenzimidazole
- Ac-beta-ala-his-ser-his- oh
- Ac-his- nhme
- 1-Amino-1h-imidazole- 5-carboxamide
- 3-Amino-6-chloroimidazo[1,2-b] pyridazine
- 2-Amino-9-(4-hydroxy-3-(hydroxymethyl)butyl) -1,9-dihydro-6H-purin-6-one
- N-(2-Aminoethyl) biotinamide
- 2-[(4-Allyl-2-methoxyphenoxy)methyl] -1h-benzimidazole
- 3-(4-Aminobenzyl)-5,5-dimethylimidazolidine-2, 4-dione
- 8- Aminoguanosine
- 8- Aminoguanine
- 6-Amino-3-chloroimidazo[1,2-a] pyridine
- 2-Amino-7-bromo-1h,4h-imidazo[2,1-f][1,2,4] triazin-4-one
- 1-(4-Aminobenzyl)-n-(3,5-dimethylphenyl)- 1h-imidazole-4-carboxamide
- 4-Amino-7-bromoimidazo[2,1-f][1,2,4] triazine
- 6-Amino-3-bromo-imidazo[1,2-a] pyridine
- (2R)-2-Acetamido-3-(1h-imidazol-4-yl)propanoic acid
- N-Acetyl-dl-histidine monohydrate
- 2-[(4-Allyl-2-methoxyphenoxy)methyl]- 1h-benzimidazole
- 1-Allyl-3-methylimidazolium bromide
- 1-(4-Aminobenzyl)-n-(3-chlorophenyl)-1h-imidazole- 4-carboxamide
- 9-[(2-Acetoxyethoxy)methyl] -acetylguanine
- 1-Allyl-2-(trifluoromethyl) benzimidazole
- 1- Allylimidazole
- 1-(5-Acetyl-4-methylthiazol-2-yl) imidazolidin-2-one
- 1-Acetyl- 2-imidazolidinone
- 1-(2-Aminoethyl)- 2-imidazolidone
- Alpha-methyl-dl-histidine dihydrochloride
- 1-(4-Aminobenzyl)-n-(4-fluoro-2-methylphenyl)- 1h-imidazole-4-carboxamide
- 1-Allyl-3- methylimidazolium
- N-[2-[2-(2-Aminoethoxy)ethoxy]ethyl] biotinamide
- 3-Acetoxy- 5-bromoindole
- 4- Acetylimidazole
- 2-Amino-1,9-dihydro-9-[(1s,3r,4s)-4-(benzyloxy)-3-(benzyloxymethyl)-2-methylenecyclopentyl]- 6h-purin-6-one
- 5-Aminoimidazole-4-carboxamide 1-beta-D-ribofuranoside
- 2-Amino-4-bromo- 1-methylimidazole
- 1- Acetylimidazole
- N(Alpha), n-(im)-di-boc-l-histidine methyl ester
- (1-[3-(Acetylamino)phenyl]-2,5-dioxoimidazolidin-4-yl)acetic acid
- 4-Amino-1h-imidazole- 5-carbonitrile
- 2-Amino-9-[4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-1, 9-dihydro-6h-purin-6-one
- 5- Aminobenzimidazole
- 5-Amino-4-bromo- benzimidazole
- 3-Acetyl-6-chloroimidazo[1,2-b] pyridazine
- 3'-Amino-2', 3'-dideoxyguanosine
- Acetylene-PEG4-biotin conjugate
- 1-(4-Acetylphenyl) imidazole
- N-(4-Aminobutyl) biotinamide
- 4-Amino- 5-imidazolecarboxamide
- 1-(4-Aminobenzyl)-n-(4-methoxyphenyl)- 1h-imidazole-4-carboxamide
- 2-Amino-6-chloroimidazo[1,2-b] pyridazine
- D4476 CK1 Inhibitor
- 2- Acetylimidazole
- 2-Amino-6-ethyl-7-hydroxy-3-(4-methyl-1h-imidazol-2-yl)- 4h-chromen-4-one
- 2-Acetamido- 6-hydroxypurine
- Alpha-(2,3-dimethylphenyl)-1-(trityl)-1h-imidazole- 4-methanol
- 1-Allyl-1h-benzimidazole- 2-thiol
- Alpha-methyl-imidazo[1,2-a]pyridine- 3-methanol
- Deuterated
- 1-Bromo-3-(methoxy-d3) -5-nitrobenzene
- 1-(Benzyloxy)-3-bromo-5-(methoxy-D3) benzene
- Methyl 2-(methoxy-d3)benzoate
- Acetonitrile-d3 98%
- 5-Amino-1-(methyl-d3) pyridin-2-one
- 5-Amino-1-(methyl-d3)pyridin-2-one hydrochloride
- 1-(Benzyloxy)-3-chloro-5-(methoxy-d3) benzene
- 4-(Benzyloxy)-1-chloro-2-(methoxy-d3) -benzene
- 4-(Benzyloxy)-2-chloro-1-(methoxy-D3) benzene
- 1-(Benzyloxy)-3-(methoxy-d3)-5-(trifluoromethoxy) benzene
- 4-(Benzyloxy) -1-(methoxy-d3)-2-(trifluoromethyl)benzene
- 4- (Benzyloxy)-1-(methoxy-d3)-2-(trifluoromethyl)benzene
- (S)-1-Boc-2-(n-methyl-d3-aminomethyl)- pyrrolidine
- 3-Bromo-4-(methoxy-d3) aniline
- 3-Bromo-5-(methoxy-d3) aniline
- 4-Bromo-3-(methoxy-d3) aniline
- 3-Bromo-4-(methoxy-d3)benzoic acid
- 5-Bromo-2-(methoxy-d3)benzoic acid
- 3-Bromo-5-(methoxy-d3) phenol
- 5-Bromo-1-(methyl-d3) indole
- 1-(2-Bromophenyl)-4-(methyl-d3) piperazine
- 1-(3-Bromophenyl)-4-(trideuteriomethyl) piperazine
- 1-(4-Bromophenyl)-4-(methyl-d3) piperazine
- 1-Chloro-2,4-difluoro-5-(methoxy-d3) benzene
- 3-Chloro-5-(methoxy-d3) aniline
- 4-Chloro-3-(methoxy-d3) aniline
- 2-Chloro-4-(methoxy-d3) phenol
- 3-Chloro-4-(methoxy-d3) phenol
- 3-Chloro-5-(trideuteriomethoxy) phenol
- 4-Chloro-3-(methoxy-d3) phenol
- 5-Chloro-1-(methyl-d3) -4-nitro-1h-pyrazole
- 4-Cyano-N-(methyl-d3) benzenesulfonamide
- Dimethylsulfoxide- d6
- 2-Fluoro-5-(methoxy-d3) benzonitrile
- 3-Fluoro-5-(methoxy-d3) phenol
- [2-fluoro-5-(methoxy-d3) phenyl]methanamine
- 2-(Methoxy-d3)benzoic acid
- 3-(Methoxy-d3)benzoic acid
- 4-(Methoxy-d3) benzoic acid
- 2-(Methoxy-d3) naphthalene-1-carbaldehyde
- 3-(Methoxy-D3)5-(trifluoromethoxy) phenol
- Methyl 5-bromo-2-(methoxy-d3)benzoate
- Methyl 4-(methoxy-d3)benzoate
- 1-(Methyl-d3) -4-nitro-1H-pyrazole
- 1-(Methyl-d3) -2-oxopyridine-4-carbonitrile
- 1-(Methyl-d3)-6-oxopyridine-3-carboxylic acid
- 4-[Methyl-d3-sulfamoyl]benzoic acid
- Methyl-d3 p-toluenesulfonate
- Methyl-d3 triflate
- Methyl-d3 (2,2,2-trifluoroethyl)amine hydrochloride
- Pyrimidines
- 4-Amino-5-(2-methylphenyl)-4h-1,2, 4-triazole-3-thiol
- 5-Aminopyrimidine-2- carboxylic acid
- 2-Amino-4-hydroxy-6- phenylpyrimidine
- 2-Acetamido-5- bromopyrimidine
- 3-Acetamido-6- chloropyridazine
- 3-Acetamido-4,5-dibromo- 1H-pyrazole
- 2- Acetamidopyrimidine
- 2-Acetamidopyrimidine-5-boronic acid, pinacol ester
- 3- Acetylaminopyrazole
- 2-Acetyl-3,5(6)-dimethylpyrazine, mixture of isomers
- 5-Acetyl-1,3-dimethyl-1,2,3,4-tetrahydropyrimidine-2, 4-dione
- 2-Acetyl-3- methylpyrazine
- 5-Acetyl-4-methylpyrimidin-2(1h)- one
- 2-[(Acetyloxy)methyl]-4-(2-amino-9H-purin-9-yl)butyl acetate
- 1-(4-Acetylphenyl)-4-bromo-3,5- dimethylpyrazole
- 2- ACETYLPYRAZINE
- 1-(1H-Pyrazol-4-yl) ethanone
- 1-Acetyl-1H-pyrazole-4-boronic acid, pinacol ester
- 2- Acetylpyrimidine
- 5- Acetylpyrimidine
- 1-(1H-Pyrrol-2-yl)ethanone 2-acetyl pyrrole
- 1-quinoxalin-2- ylethanone
- 1-Acetyl-4-tert- butoxycarbonylpiperazine
- 5-Acetyl-1H-pyrimidine- 2,4-dione
- Adenine phosphate
- Adenine hemisulfate
- Alfuzosina. ..
- Adenosine ..
- Alfuzosin ..
- 5-Allyl-4,6- dichloropyrimidine
- 5-Allyl-2,6-dimethyl- 4-pyrimidinol
- 5-Allyl-2-mercapto-6- methylpyrimidin-4(3h)-one
- 4-Allyl-1-mercapto-4h-[1,2,4]triazolo[4,3-a] quinazolin-5-one
- 2-Allyl-6-(methylthio)-1h-pyrazolo [3,4-d]pyrimidin-3(2h)-one
- 4-(Allyloxy)-2-chloro-6- methylpyrimidine
- 5-Allylsulfanyl-[1,3,4]thiadiazol- 2-ylamine
- 2-Amidinopyrimidine hydrochloride
- 4-Amino-5-aminomethyl-2- methylpyrimidine
- 3-Amino-6-(aminomethyl)-1,2,4-triazin-5(4h)-one dihydrochloride
- 6-Amino-5-benzylamino-1-butyl- 1h-pyrimidine-2,4-dione
- 2-Amino-5- chlorobenzo[d]thiazole
- 4-Amino-2-chloro-5- fluoropyrimidine
- 2-Amino-4-chloro-6- hydroxypyrimidine
- 3-Amino-6-chloroimidazo [1,2-b]pyridazine
- 4-Amino-2-chloro-5- iodopyrimidine
- 2-Amino-4-chloro-6- isopropylpyrimidine
- 2-Amino-5-chloro-3- methoxypyrazine
- 2-Amino-4-chloro-6- methoxypyrimidine
- 4-Amino-6-chloro-5- methoxypyrimidine
- 2-Amino-3-chloro-5- methylpyrazine
- 2-Amino-4-chloro-6- methylpyrimidine
- 4-Amino-2-chloro-5- methylpyrimidine
- 4-Amino-6-chloro-2- methylpyrimidine
- 5-Amino-2-chloro-4- methylpyrimidine
- 4-Amino-6-chloro-2- (methylthio)pyrimidine
- 5-Amino-3-(4-chlorophenyl) isoxazole
- 2-Amino-6- chloropurine
- Cladribine ..
- 4-Amino-6-chloro-5 -fluoropyrimidine
- 3-Amino-5- hydroxypyrazole
- 2-Amino-3- hydroxypyrazine
- 4-Amino-6-hydroxy-2- methylpyrimidine
- 5-Amino-1-(2-hydroxyethyl) pyrazole
- 7-Amino-furo[2,3-b]pyrazine-6-carboxylic acid ethyl ester
- 2-Amino-5- fluoropyrimidine
- 4-Amino-5-(2-fluorophenyl) -4h-1,2,4-triazole-3-thiol
- 2-Amino-5-(4-fluorobenzylthio)- 1,3,4-thiadiazole
- 2-Amino-3- ethynylpyrazine
- 4-(2-Aminoethyl)-5-methyl-1,2- dihydro-3h-pyrazol-3-one
- 2-Amino-3-(ethylamino) pyrazine
- 2-Amino-5- ethoxypyrazine
- 4-Amino-5-(2-ethoxyphenyl)-4h-1, 2,4-triazole-3-thiol
- 6-Amino-1,3- dimethyluracil
- 5-Amino-1,3-dimethylpyrimidine-2, 4(1H,3H)-dione
- 4-Amino-1,5-dimethylimidazole hydrochloride
- 2-Amino-3-(dimethylamino) pyrazine
- 2-Amino-4,6-dimethoxy-1 ,3,5-triazine
- 4-Amino-N-(2,6-dimethoxypyrimidin-4-yl) benzene-1-sulfonamide
- 2-Amino-5- hydroxypyrimidine
- 4-Amino-6- hydroxypyrimidine
- 3-Amino-1H-indazole-7-boronic acid pinacol ester
- 2-Amino-4-iodo-6- methoxypyrimidine
- 2-Amino-5-iodo-4- methoxypyrimidine
- 4-Amino-6-iodo-2- methylpyrimidine
- 2-Amino-3- iodopyrazine
- 2-Amino-5- iodopyrazine
- 3-Amino-4-iodo- 1h-pyrazole
- 4-Amino-3-iodo-1H-pyrazolo [3,4-d]pyrimidine
- 2-Amino-5- iodopyrimidine
- 4-Amino-5- iodopyrimidine
- 4-Amino-6-iodopyri midine
- 5-Amino-1-isopropyl-1h- indazole
- 4-Amino-1-isopropylpyrazole hydrochloride
- 4-Aminoisoxazole hydrochloride
- 3-Aminoisoxazolo[4,5-b] pyrazine
- 4-Amino-5-(2-methoxy-5-methyl-phenyl)- 4h-[1,2,4]triazole-3-thiol
- 2-Amino-4-methoxy-6- methylpyrimidine
- 2-Amino-4-methoxy-6-methyl-1,3,5- triazine
- 3-Amino-5-(2-methoxyphenyl)- 1H-pyrazole
- 4-Amino-5-(2-methoxy-phenyl)-4h- [1,2,4]triazole-3-thiol
- 2-([4-Amino-5-(2-methoxyphenyl)-4h-1,2,4-triazol-3-yl]sulfanyl)acetic acid
- 2-Amino-3- methoxypyrazine
- 2-Amino-5- methoxypyrazine
- 2-Amino-6- methoxypyrazine
- 3-Amino-6- methoxypyridazine
- 2-Amino-4- methoxypyrimidine
- 2-Amino-5- methoxypyrimidine
- 4-Amino-2- methoxypyrimidine
- 4-Amino-6- methoxypyrimidine
- 4-Amino-N-(5-methoxypyrimidin-2-yl) benzene-1-sulfonamide
- 2-Amino-8- methoxyquinazoline
- 6-Aminomethyl-1H -benzotriazole
- 6-((4-(Aminomethyl)benzyl)oxy)- 7H-purin-2-amine
- 2-Aminomethyl-3- chloropyrazine
- 4-Amino-5-(2-methyl-3-furyl)-4h- 1,2,4-triazole-3-thiol
- 5-Amino-3-methyl-isoxazole-4-carboxylic acid ethyl ester
- 2-(Aminomethyl)- 5-methylpyrazine
- 5-(Aminomethyl)-2-methylpyrimidin-4-amine dihydrochloride
- 6-Amino-1-methyl-5- nitrosouracil
- 5-Amino-3-methyl-1- phenylpyrazole
- 3-Amino-2-methyl-5-phenyl-3h-thieno [2,3-d]pyrimidin-4-one
- 4-Amino-6-(4-methyl-1-piperazinyl) pyrimidine
- 4-Amino-2-methyl-5-propylpyrazole-3-carboxamide hydrochloride
- 2- Aminomethylpyrazine
- 2-Amino-3- methylpyrazine
- 2-Amino-6- methylpyrazine
- 3- (Aminomethyl)pyrazole
- 3-Amino-5- methylpyrazole
- 4-Amino-1-methyl-1H-pyrazole- 3-carbonitrile
- 5-Amino-1-methyl-1H-pyrazole- 4-carbonitrile
- 5-Amino-1-methyl-1H-pyrazole-4- carboxylic acid
- 3-Amino-4-methyl- pyridazine
- 3-Amino-6- methylpyridazine
- 2-Amino-4- methylpyrimidine
- 4-(Aminomethyl) pyrimidine
- 4-Amino-5- methylpyrimidine
- 5-Amino-4- methylpyrimidine
- 2-Amino-4-methylpyrimidine-5- carbonitrile
- 2-Aminomethylpyrimidine hydrochloride
- (4-Amino-2-methyl-5-pyrimidinyl) methanol
- 2-[4-(6-Amino-2-methylpyrimidin-4-yl)piperazin-1-yl] ethanol
- 4-Amino-2-(methylsulfanyl)pyrimidine-5- carbonitrile
- 4-Amino-N-(5-methyl-1,3,4-thiadiazol-2-yl)benzene-1- sulfonamide
- 5-Amino-2-(methylthio)pyrimidine-4-carboxylic acid
- 4-Amino-2-(methylthio)-6- pyrimidinol
- 4-Amino-6- morpholinopyrimidine
- 2-Amino-3-morpholin-4- ylpyrazine
- 2-Amino-4-morpholin-4-yl- pyrimidine
- 3-Amino-5- nitroindazole
- 5-Amino-1-(4-nitrophenyl)-1h-pyrazole-4- carbonitrile
- 4-Amino-5-(2-nitro-phenyl)-4h-[1,2,4] triazole-3-thiol
- 2-Amino-5- nitropyrimidine
- 2-((2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)methoxy)ethyl L-valinate hydrochloride
- (S)-2-(4-(2-(2-Amino-4-oxo-4,7-dihydro-1h-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl)benzamido) pentanedioic acid
- 4-(2-(2-Amino-4-oxo-4,7-dihydro-1h-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl)benzoic acid
- 2-(4-Aminophenyl) oxazole
- 3-(4-Aminophenyl) pyrazole
- 3-Amino-4-phenyl-1H- pyrazole
- 3-Amino-5- phenylpyrazole
- 5-Amino-1-phenylpyrazole-4- carbonitrile
- 2-(5-Amino-3-phenyl-1h-pyrazol-1-yl) ethanol
- 5- Aminophthalazine
- 2-Amino-4-piperidino-6- methylpyrimidine
- 4-Amino-6- piperidinopyrimidine
- 6-Amino-1-propyl-3H- pyrimidine-2,4-dione
- 1-((4-Amino-2-propylpyrimidin-5-yl)methyl)-2-methylpyridin-1-ium chloride hydrochloride
- 2-Aminopurine riboside
- Adenosine 5'-monophosphate
- {[2-(6-Amino-9H-purin-9-yl)ethoxy]methyl}phosphonic acid
- 5-Aminopyrazine-2- carbonitrile
- 3-Aminopyrazine-2-carboxylic acid
- 6-Aminopyrazine-2-carboxylic acid
- 3-Amino-2(1H)- pyrazinone
- 4-Amino-1H- pyrazole
- 3-Amino-1H-pyrazole-4- carbonitrile
- 3-Aminopyrazole-4- carboxylic acid
- 4-Aminopyrazole hydrochloride
- 3-Aminopyrazolo[1,5-a] pyrimidine
- 4-Aminopyrazolo[3,4-d] pyrimidine
- 4-(3-Amino-1H-pyrazol-4-yl) benzonitrile
- 2-(3-Amino-1H-pyrazol-1-yl) ethanol
- 2-(4-Amino-1h-pyrazol-1-yl) ethanol
- 2-(4-Amino-1H-pyrazol-1-yl)ethanol hydrochloride
- 4- Aminopyridazine
- 3-Aminopyridazine hydrochloride
- 4- Aminopyrimidine
- 5- Aminopyrimidine
- 2-Amino-4- pyrimidinecarbonitrile
- 2-Aminopyrimidine-5- carbonitrile
- 2-Amino-5- pyrimidinecarboxaldehyde
- 2-Aminopyrimidine-4-carboxylic acid
- 2-Aminopyrimidine-5-carboxylic acid
- 2-Aminopyrimidine-5- carboxylic acid
- 4-Aminopyrimidine-5-carboxylic acid
- 6-Amino-pyrimidine-4-carboxylic acid
- 2-Amino-pyrimidine-5-carboxylic acid ethyl ester
- 2-Amino-5- pyrimidinesulfonamide
- 2-Aminopyrimidin- 4-ol
- 5-Aminopyrimidin-4- ol
- 2-(2-Aminopyrimidin-5-yl) benzaldehyde
- 4-Amino-N-(pyrimidin-2-yl)benzene-1- sulfonamide
- 3-(2-Aminopyrimidin-5-yl)benzoic acid
- 4-(2-Aminopyrimidin-5-yl)benzoic acid
- 1-(2-Aminopyrimidin-4-yl) ethanone
- 1-Amino-1H-pyrrole-2- carboxamide
- 4-Amino-7H-pyrrolo[2,3-d] pyrimidine
- 4-Amino-7h-pyrrolo[2,3-d]pyrimidine hydrogen sulfate
- 6-Aminopyrrolo[1,2-f][1,2,4] triazin-4(3h)-one
- 2- Aminoquinazoline
- 6- Aminoquinazoline
- 4-Aminoquinazoline-8- carbonitrile
- 4-Amino-2-sulfanylpyrimidine-5- carbonitrile
- 3-Amino-5-tert- butylpyrazole
- 5-Amino-1-tert-butyl-1h-pyrazole-4- carbonitrile
- (7-Amino-3-((tetrahydrofuran-2-yl)methyl)-3h-[1,2,3]triazolo[4,5-d]pyrimidin-5-yl) methanol
- 2-Amino-5,6,7,8-tetrahydropyrido-[4,3-d]-pyrimidine dihydrochloride
- 4-[(5-Amino-1,3,4-thiadiazol-2-yl)methoxy] benzenesulfonic acid
- 2-(5-Amino-[1,3,4]thiadiazol-2-ylsulfanyl)-n-(1,5-dimethyl-3-oxo- 2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)-acetamide
- 2-(5-Amino-[1,3,4]thiadiazol-2-ylsulfanyl)-n-(1,5-dimethyl- 3-oxo-2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)-propionamide
- 2-Amino-5-(2-thienyl)-1,3,4- thiadiazole
- 6-Amino-2-thiouracil monohydrate
- 3-Amino-5-p- Tolylpyrazole
- 3-Amino-1,2,4- triazole
- 4-Amino-1,2,4- triazole
- 3-(5-Amino-4h-[1,2,4]triazol-3-yl)- propan-1-ol
- N-Benzyl-2-chloroquinazolin-4- amine
- N-Benzyl-2-chloroquinazolin- 4-amine
- 7-Benzyl-4-chloro-7h-pyrrolo[2,3-d] pyrimidine
- 4-Benzyl-2-chloro- pyrimidine
- 6-Benzyl-4-chloro-6,7-dihydro-5h-pyrrolo [3,4-d]pyrimidine
- 1-Benzyl-4-bromopyrazole-5-boronic acid, pinacol ester
- 1-Benzyl-4-bromo-1H- pyrazole
- Quinolines
- 4-(Benzylsulfanyl) benzaldehyde
- (2-Acetyl-1,2-dihydroisoquinolin-1-yl)acetic acid
- 2-Methylquinoline-6- carbonitrile
- 3-Amino-7-bromoisoquinoline-4- carbonitrile
- 5- Acetamidoisoquinoline
- 1-Acetyl-8-allyl-1,2,3,4- tetrahydroquinoline
- 5-Acetyl-8-(benzyloxy)quinolin- 2(1h)-one
- 3-Acetyl-6-bromo-4- phenylquinolin-2(1h)-one
- 3-Acetyl-6-bromo-4-phenylquinolin- 2(1h)-one
- (2-Acetyl-6,7-dimethoxy-1,2,3,4-tetrahydro-isoquinoline-1-yl)-acetic acid
- 5-Acetyl-8-hydroxy-1h-quinolin- 2-one
- 3- Acetylquinoline
- 6- Acetylquinoline
- 3-Acetyl-1h-quinolin- 2-one
- 2-Acetyl-1,2,3,4-tetrahydro-3-isoquinolinecarboxylic acid
- 1-Acetyl-1,2,3,4-tetrahydroquinolin-7-amine hydrochloride
- 1-Acetyl-1,2,3,4-tetrahydro-6- quinolinecarbonitrile
- 1-Acetyl-1,2,3,4-tetrahydroquinoline-6- sulfonyl chloride
- 6-Acetyl-1,2,3,4- tetrahydroquinolin-2-one
- 1-(1-Acetyl-2,2,4,6-tetramethyl-1,2,3,4- tetrahydroquinolin-7-yl)ethanone
- 1-(1-Acetyl-2,2,4,8-tetramethyl-1,2,3,4- tetrahydroquinolin-7-yl)ethanone
- 3-Allyl-1,5- naphthyridine
- 4-Allyl-5-(2-phenylquinolin-4-yl)-4h-1,2,4- triazole-3-thiol
- 4-Amino-2,8-bis(trifluoromethyl)- quinoline
- 4-Amino-3- bromoisoquinoline
- 8-Amino-5- bromoisoquinoline
- 4-Amino-3-bromo-8- methoxyquinoline
- 2-Amino-6-bromo-3- methylquinoline
- 4-Amino-6-bromo-2- methylquinoline
- 4-Amino-6-bromo-7- methylquinoline
- 6-Bromo-4-chloro-7- methoxyquinoline
- 4-Amino-8-bromo-2- methylquinoline
- 4-Amino-3-bromo-6- nitroquinoline
- 2-Amino-6-bromopyrido[2,3-d]pyrimidin-4 (3H)-one
- 1-Chloroisoquinoline-6- carbonitrile
- 1-Chloroisoquinoline-7- carbonitrile
- 6-Chloroisoquinoline-1- carbonitrile
- 4-chloroisoquinoline hydrochloride
- 1-Chloroisoquinoline-5-sulfonyl chloride
- 1-Chloroisoquinolin- 7-ol
- 1-Chloroisoquinolin-7- ol
- 3-Chloroisoquinolin-4- ol
- 3-Chloroisoquinolin-5- ol
- 1-Chloroisoquinolin -6-ol
- 3-Chloroisoquinolin- 4-ol
- 3-Chloroisoquinolin- 5-ol
- 1-Chloro-4- methoxyisoquinoline
- 3-Chloroisoquinolin- 6-ol
- 1-Chloroisoquinolin- 6-ol
- Methyl 1-chloroisoquinoline-7-carboxylate
- Methyl 3-chloroisoquinoline-6-carboxylate
- Methyl 3-chloroisoquinoline-7-carboxylate
- 2-Methylquinoline-4- carbohydrazide
- 2-Methyl-4- quinolinecarbonitrile
- 4-Methylquinoline-2- carbonitrile
- 2-Methylquinoline-4- carboxamide
- Methyl quinoline-2-carboxylate
- IMPURITY CHEMICALS
- Ascorbic Acid EP Impurity E (Oxalic Acid)
- Almotriptan Related Compound C (N-Desmethyl Almotriptan)
- Abacavir EP Impurity E (Dihydro Abacavir)
- 4-[2-(1,3-Dihydro-1,3-dioxo-2H-isoindol-2-yl)ethoxy]-3-oxobutanoic Acid Methyl Ester
- (2RS)-2-(3,4-Dimethoxyphenyl)-3- methylbutanenitrile
- Anastrozole Monoacid
- Acetazolamide EP Impurity D
- 3-Piperazinyl-1,2-benzisothiazole. HCl
- Artemether Related Compound B (-Artemether)
- 1-(3,4-Dimethoxyphenyl)-2- methylpropan-1-one
- Abacavir EP Impurity B/ Abacavir Related Compound D (O-Pyrimidine Derivative Abacavir)
- Abacavir EP Impurity C/ Abacavir Related Compound A (Descyclopropyl Abacavir)
- Abacavir EP Impurity C/ Abacavir Related Compound A (Descyclopropyl Abacavir)
- Abacavir EP Impurity F/ O-t-Butyl Derivative Abacavir (Abacavir t-Butyl Ether)
- Abacavir Related Compound B
- Abacavir Related compound C (Abacavir 6- Chloro Analog)
- 2-cyano-3- morpholinoacrylamide
- Abacavir Cyclopropyl Diamino Purine Impurity
- (1S,4R)-4-Amino-2-cyclopenten-1-carboxylic Acid
- N-(2-Amino-4,6-dichloro-5-pyrimidinyl) formamide
- (1S,4R)-(4-Aminocyclopent-2-enyl)methanol hydrochloride
- rac 2-Azabicyclo[2.2.1]hept-5-en-3-one
- Abiraterone Methyl Ether
- Acarbose EP Impurity A
- Acarbose EP Impurity C
- Acarbose EP Impurity E
- Acarbose EP Impurity F
- Acarbose EP Impurity G
- Acebutolol EP Impurity A (Acebutolol Epoxypropoxy Impurity)
- Acebutolol EP Impurity B/ Acebutolol Related Compound B (Diacetolol)
- Acebutolol EP Impurity C
- Acebutolol EP Impurity D
- Acebutolol EP Impurity E (Acebutolol Desacetyl Impurity)
- Acebutolol EP Impurity J
- Aceclofenac EP Impurity A (Diclofenac)
- Aceclofenac EP Impurity B (Diclofenac Methyl Ester)
- Aceclofenac EP Impurity C (Diclofenac Ethyl Ester)
- Aceclofenac EP Impurity D (Aceclofenac Methyl Ester)
- Aceclofenac EP Impurity E (Aceclofenac Ethyl Ester)
- Aceclofenac EP Impurity F (Aceclofenac Benzyl Ester)
- Aceclofenac EP Impurity G (Acetic Aceclofenac)
- Aceclofenac Impurity EP H (Diacetic Aceclofenac)
- Aceclofenac EP Impurity I (Diclofenac EP Impurity A/ Diclofenac USP Related Compound A/ Diclofenac Amide)
- Acemetacin EP Impurity A (Indomethacin EP Impurity A/ Indomethacin Related Compound B/ Bezafibrate EP Impurity B)
- Acemetacin EP Impurity B (Indomethacin)
- Acesulfame Potassium Impurity A/ Acetoacetamide
- Acetazolamide EP Impurity A
- Acetazolamide EP Impurity B
- Acetazolamide EP Impurity C
- Acetazolamide EP Impurity E
- Acetazolamide EP Impurity G
- Acetylsalicylic Acid EP Impurity A (Butyl Parahydroxybenzoate EP Impurity A/ p-Salicylic Acid/ 4-Hydroxybenzoic acid)
- Acetylsalicylic Acid EP Impurity B (4-Hydroxyisophthalic Acid)
- Acetylsalicylic Acid EP Impurity C (Salicylic Acid/ Lamivudine EP Impurity C/ Mesalazine EP Impurity H/ Sulfasalazine EP Impurity H)
- Acetylsalicylic Acid EP Impurity D (Acetylsalicylsalicylic acid)
- Acetylsalicylic Acid EP Impurity E (Salsalate/ Salicylsalicylic acid)
- Acetylsalicylic Acid EP Impurity F (Acetylsalicylic Anhydride)
- Salicylaldehyde Azine
- Acyclovir EP Impurity A (O-Acetyl Aciclovir/ Valaciclovir EP Impurity I/ Valaciclovir Related Compund I)
- Acyclovir EP Impurity B (Guanine/ Valaciclovir EP Impurity A/ Valaciclovir Related Compound A)
- Acyclovir EP Impurity C (Aciclovir N7-Isomer)
- Acyclovir EP Impurity F (N2-Acetyl Aciclovir)
- Acyclovir EP Impurity G (Aciclovir N,O-Diacetate)
- Acyclovir Impurity H
- Acyclovir EP Impurity J
- Acyclovir EP Impurity L
- Acyclovir EP Impurity M (N,O-Diacetate Isoacyclovir)
- Acyclovir EP Impurity P
- Acyclovir L-Alaninate (Valaciclovir EP Impurity H/ Valaciclovir Related Compund H/ Didesmethyl Valacyclovir)
- Acyclovir L-Isoleucinate (Valaciclovir EP Impurity J)
- Acyclovir N-Methylene Dimer (Valaciclovir EP Impurity L)
- Acyclovir D-Valinate (Valaciclovir EP Impurity R/ D-Valacyclovir)
- 2-amino-8-hydroxy-9- ((2-hydroxyethoxy)methyl)-1,9-dihydro-6H-purin-6-one
- Adapalene EP Impurity B (Adapalene 3-Hydroxy Impurity)
- Adapalene EP Impurity C/ Adapalene Related Compound C (o-Adamantylanisole)
- Adrenaline EP Impurity B
- Adrenaline Impurity C/ Epinephrine Impurity C (Adrenalone)
- Adrenaline EP Impurity D
- Adrenaline Impurity E/ Epinephrine Impurity E
- Adrenaline EP Impurity F
- Epinephrine Sulfonic Acid
- Agomelatine Dimer Acetamide
- Agomelatine Desmethyl Impurity
- 7-Methoxy-1- naphthaleneacetonitrile
- Albendazole EP Impurity A (Albendazole Amine/ Desmethoxycarbonyl Albendazole)
- Albendazole EP Impurity C (Albendazole Sulfone)
- Albendazole EP Impurity D (Desmethoxycarbonyl Albendazole Sulfone)
- Albendazole EP Impurity E (Carbendazim)
- Albendazole EP Impurity F
- Albendazole EP Impurity I (Oxibendazole)
- Albendazole EP Impurity J
- Albendazole EP Impurity K
- Albendazole EP Impurity L
- O-Desmethyl-O-ethyl Albendazole
- Alfuzosin EP Impurity A/ Alfuzosin Related Compund A (Alfuzosin Tetradehydro Impurity)
- Alfuzosin EP Impurity B (Prazosin EP Impurity A/ Terazosin EP Impurity A/ Doxazosin EP Impurity F/ Doxazosin Related Compund C)
- Alfuzosin EP Impurity C
- Alfuzosin EP Impurity D / Alfuzosin Related Compound D (Alfuzosin Aminopropyl Impurity)
- Alfuzosin EP Impurity E
- Alfuzosin EP Impurity F
- Alfuzosin EP Impurity G (Dimer)
- (2-Chloro-6,7-dimethoxyquinazoline-4-yl)- (3,4-dimethoxyphenyl)-amine
- Alimemazine EP Impurity C (Promethazine EP Impurity A/ Phenothiazine)
- Allopurinol EP impurity A/ Allopurinol Related Compound A
- Allopurinol EP impurity B/ Allopurinol Related Compound B
- Allopurinol EP impurity C/ Allopurinol Related Compound C
- Allopurinol EP impurity D/ Allopurinol Related Compound D
- Allopurinol EP Impurity E/ Allopurinol Related Compound E
- Almotriptan Related Compound A (N-Hydroxymethyl Almotriptan)
- Almotriptan Related Compound B (Almotriptan Didesmethyl Impurity)
- Almotriptan Related Compound D (Almotriptan N-Oxide)
- 6-Chloro-3- methyluracil
- Alogliptin 6-Chloro Impurity
- 2-Cyanobenzyl bromide
- Alogliptin S-Isomer
- 4,6-Dichloro-2- (methylthio)pyrimidine
- 4,6-Dichloro-2-(methylsulfonyl) pyrimidine
- 6-Chloro-2-(methylthio) pyrimidin-4-ol
- Altizide EP Impurity A (Hydrochlorothiazide EP Impurity B/ Hydrochlorothiazide Related Compound A/ Benzothiadiazine Related Compound A/ Salamide )
- Amantadine EP Impurity B (N-Acetyl Amantadine)
- Ambrisentan Hydroxy Acid Impurity
- Ambroxol EP Impurity A (Bromhexine EP Impurity A)
- Ambroxol EP Impurity B
- Ambroxol EP Impurity C (Dehydro Ambroxol)
- Ambroxol EP Impurity D (cis-Ambroxol HCl/ Acebrophylline Impurity D)
- Ambroxol EP Impurity E (Bromhexine EP Impurity B)
- 2-Amino-3-benzoyl--oxo-benzeneacetic Acid
- 7-Benzoylindoline- 2,3-dione
- 6-Aminocaproic Acid Dimer
- Amiodarone EP Impurity B/ Amiodarone Related Compound B (Desethyl Amiodarone)
- Amiodarone EP Impurity D/ Amiodarone Related Compound D
- Amiodarone EP Impurity E/ Amiodarone Related Compound E
- Amiodarone EP Impurity F/ Amiodarone Related Compound F
- Amiodarone EP Impurity H/ Amiodarone Related Compound H
- Amiodarone Methoxy Impurity
- 2-butyl benzofuran
- (2S)-2-Aminomethyl-1- ethylpyrrolidine
- 2,4- dimethylaniline
- Amlodipine Impurity-A (EP) /Amlodipine Related Compound D (Phthaloyl Amlodipine)
- Amlodipine Impurity-B (EP) (Methyl amino phthaloyl Amlodipinee)
- Amlodipine EP Impurity C (Bis(aminoethoxy) Amlodipine)
- Amlodipine Impurity-D (EP) /Amlodipine Related Compound A (Aromatic Amlodipine/ Dehydro Amlodipine)
- Amlodipine Impurity-E (EP) /Amlodipine Related Compound E (Amlodipine Diethyl Ester)
- Amlodipine Impurity-G (EP) /Amlodipine Related Compound C
- Amlodipine Impurity-H (EP) ( 2-Carboxybenzoyl Amlodipine)
- Amlodipine 4-Chloro Analogue (Para chloro Amlodipine)
- Amlodipine Dimethoxy intermediate Impurity
- Amlodipine Aspartic Acid Impurity
- Amlodipine Lactose Adduct (Amlodipine N-Lactoside)
- Amlodipine Deschloro Impurity
- Methyl benzenesulfonate
- ethyl benzenesulfonate
- Isopropyl Benzenesulfonate
- 4-(2-Phthalimidoethoxy)acetoacetic Acid Ethyl Ester
- Ethyl 3-aminocrotonate
- 5-ethyl 7-methyl 6-(2-chlorophenyl)-8-methyl-3,4,6,7-tetrahydro-2H-benzo[b][1,4]oxazine-5,7-dicarboxylate
- 4-(2-Chlorophenyl)-2,6-bis[[2-(1,3-dihydro-1,3-dioxo-2H-isoindol-2-yl)ethoxy]methyl]-1,4-dihydro-3,5-pyridinedicarboxylic Acid Ethyl Methyl Ester
- N-(2-Hydroxyethyl) phthalimide
- Amodiaquine Dichloro Impurity (Hydroxychloroquine EP Impurity G/ ChloroquineRelated Compound A)
- N-(4-Hydroxyphenyl) acetamide
- Amoxicillin EP Impurity B/ Amoxicillin Related Compound B (L-Amoxicillin)
- Amoxicillin EP Impurity C/ Amoxicillin Related Compound C (Amoxicillin Diketopiperazine)
- Amoxicillin EP Impurity G/ Amoxicillin Related Compound G
- Amoxicillin EP Impurity H/ Amoxicillin Related Compound H
- Amoxicillin EP Impurity I/ Amoxicillin Related Compound I (Cefadroxil EP Impurity A/ Cefoperazone Impurity 3)
- Amoxicillin EP Impurity J/ Amoxicillin Related Compound J (Amoxicillin Dimer)
- Amoxicillin EP Impurity K
- Amoxicillin EP Impurity L (6-APA Amoxicillin Amide)
- Amoxicillin Trimer
- N-Pivaloyl Amoxicillin
- Ampicillin EP Impurity A (Amoxicillin EP Impurity A
- 2-Methyl-pyrazolo[1,5-a]pyrimidine-6-carboxylic acid
- 2-(tert- Butoxycarbonyloxyimino)phenylacetonitrile
- (S)-1-(2-Chloroacetyl) pyrrolidine-2-carbonitrile
- Anagrelide Related Compound A
- Anagrelide Related Compound B
- Anagrelide Related Compound C (Anagrelide open ring ethyl ester)
- Anagrelide open ring methyl ester
- 2,3-Dichloro-6-nitrobenzyl Alcohol
- Anastrozole EP Impurity A
- Anastrozole EP Impurity F (Lisinopril EP Impurity B, Tizanidine EP Impurity I, Sultamicillin EP Impurity B)
- Anastrozole EP Impurity H/ Anastrozole Related Compound A
- 2,6-di-tert-butyl-4-(2- hydroxyethyl)phenol
- 1-(4-amino phenyl)-3-morpholino-5,6-dihydropyridin-2 (1H)-one
- Ethyl 2-chloro-2-(2 -(4-methoxyphenyl)hydrazono)acetate chloro ester
- 1-(4-iodophenyl)-3-morpholino-5,6- dihydropyridin-2(1H)-one
- Apremilast EP Impurity B (Apremilast 3-Acetamido phthalic acid Impurity)
- Apremilast EP Impurity H (Apremilast Des Acetamidophthalic acid Impurity)
- Apremilast 2-Acetamido Benzoic Acid Imurity
- [1(S)-Phenylethoxy]-Aprepitant/ Aprepitant S,R,S-Isomer
- Aripiprazole EP Impurity A/ Aripiprazole Dihydroquinolinone Impurity
- Aripiprazole EP Impurity B/ Aripiprazole Related Compound C
- Aripiprazole EP Impurity C (Aripiprazole 3-Deschloro Impurity)
- Aripiprazole EP Impurity D (Aripiprazole 2-Deschloro Impurity)
- Aripiprazole EP Impurity E/ Aripiprazole Related Compound G (Dehydro Aripiprazole)
- Aripiprazole EP Impurity F/ Aripiprazole Related Compound F (Aripiprazole N-Oxide)
- Aripiprazole EP Impurity G (Aripiprazole Dimer)
- Aripiprazole Related Compound H
- Aripiprazole Hydroxybutylpiperazine Impurity
- Aripiprazole Related Compound B (Aripiprazole Hydroxybutoxyquinoline Impurity)
- Aripiprazole Diquinoline Butanediol Impurity
- Aripiprazole N,N-Dioxide
- Aripiprazole Impurity-1
- Aripiprazole Impurity 2
- Aripiprazole Bromo Impurity/ 7-(4-bromobutoxy)quinolin-2(1H)-one
- Aripiprazole Quinolinone Impurity
- Aripiprazole Chlorophenyl piperazine Impurity
- Aripiprazole 3,4-Dichloro Impurity
- Aripiprazole Bromobutoxyquinoline Impurity
- Aripiprazole 3,4-Dichlorophenyl Piperazine
- Aripiprazole Butanoic Acid Impurity
- Aripiprazole Desethylene Impurity
- Aripiprazole Iodobutoxyquinoline Impurity
- Aripiprazole Methoxybutoxyquinoline Impurity
- Aripiprazole N-Bromobutyl Impurity
- Aripiprazole N-Isomer
- Aripiprazole Quinolinediol Impurity
- Chlorobutoxy Dihydroquinolinone
- Di-[2-(2,3-dichlorophenyl) aminoethyl]amine
- 1,4- Dibromobutane
- Thiobenzhyd rol
- Artemether Ph. Int. Impurity A (Tricabonly compound)
- Artemether Related Compound A (Artemisinin Dihydro Impurity)
- Artesunate IP Impurity B (Artemisinin)
- Ascorbic Acid EP Impurity C
- Ascorbic Acid EP Impurity D
- Asenapine Tetradehydro Impurity
- Asenapine N-Desmethyl Impurity
- Asenapine N-Oxide
- rel-(3aR,12bR)-11-Chloro-2,3,3a,12b-tetrahydro-2-methyl-1H-dibenz [2,3:6,7]oxepino[4,5-c]pyrrol-1-one
- N-Desmethyl Asenapine
- Aspartame EP Impurity A/ Aspartame Related Compound A
- Aspartame EP Impurity B/ Aspartame Related Compound B (Aspartame Acid)
- Aspartame EP Impurity C (Nateglinide EP Impurity D)
- (2S,3S)-3-Boc-amino-1,2-epoxy-4- phenylbutane
- (2R,3S)-3-(tert-Boc)amino-1,2-epoxy-4- phenylbutane
- Atenolol EP Impurity A
- Atenolol EP Impurity B/ Atenolol Related Compound A
- Atenolol EP Impurity C
- 7-Hydroxy-3,4-dihydro quinolin-2(1H)-one
- 1-(2,3-Dichlorophenyl) piperazine hydrochloride
- 7-[4-[4-(2-Chlorophenyl)-1-piperazinyl]butoxy] -3,4-dihydro-2(1H)-quinolinone
- 7-[4-[4-(3-Chlorophenyl)-1-piperazinyl]butoxy]- 3,4-dihydro-2(1H)-quinolinone
- 7-(4-(4-(2,3-dichlorophenyl) piperazin-1-yl)butoxy)-3,4-quinolin-2(1H)-one
- 4-(2,3-Dichlorophenyl)-1-[4-(2-oxo-1,2,3,4-tetrahydro quinolin-7-yloxy)butyl] piperazine-1-oxide
- 1,1'-(Ethane-1,1-diyl)bis(2,3-dichloro-4{4-[3,4-dihydro quin olin-2(1H)-one-7-yloxy butyl] piperazin-1-yl}benzene)(U/D)
- 7-(4-Hydroxy-butoxy)-3,4- dihydroquinolin-2(1H)-one
- 7-(4-Bromobutoxy)-3,4- dihydroquinolin-2-one
- 7,7'-[Butane-1,4,diylbis(oxy) ]bis[3,4-dihydroquinolin-2 (1H) -one
- 1-(3,4-Dichloro phenyl) piperazine.HCl
- 4-Hydroxy benzoic acid
- 4-Hydroxy Isopthalic acid
- 2-((2-Acetoxybenzoyl)oxy) benzoic acid
- 2-((2-Hydroxybenzoyl)oxy) benzoic acid
- 2-Acetoxybenzoic anhydride
- N-[3-[(4-Amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl]furan-2-carboxamide hydrochloride
- 4-Amino-2-chloro-6,7- dimethoxyquinazoline
- N-(3-(4-Amino-6,7-dimethoxyquinazolin-2-ylamino)propyl)-N-methyltetrahydrofuran- 2-carboxamide
- N-(4-Amino-6,7-dimethoxyquinazolin-2-yl)-N-methylpropane-1,3-diamine hydrochloride
- N-[3-[(4-Amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl]formamide hydrochloride
- N-Methyl-3-phenoxy-3-phenylpropan-1-amine hydrochloride
- (R)-(+)-N-Methyl-3-phenyl- 3-hydroxypropylaMine
- (3S)-N-Methyl-3-(2-methylphenoxy)-3-phenylpropan-1-amine hydrochloride
- (3R)-N-Methyl-3-(3-methylphenoxy)-3-phenylpropan-1-amine hydrochloride
- (3R)-N-Methyl-3-(4-methylphenoxy)-3-phenylpropan-1-amine hydrochloride
- L-(+)-Mandelic Acid
- 3,5-Bis(1-cyano-1-methylethyl)benzyl Bromide
- 4-Methylbenzenesulfonic acid monohydrate
- 2-[4-[(2RS)-2,3-dihydroxypropoxy] phenyl]acetamide
- 2-[4-[[(2RS)-oxiran-2-yl]methoxy]phenyl] acetamide
- 2-[4-[(2RS)-2-Hydroxy-3-[(1-methylethyl)amino]propoxy]phenyl]acetic acid
- 2-(4-(3-(Ethylamino)-2-hydroxypropoxy) phenyl)acetamide
- 4-(6-bis(2-hydroxyethyl)amino-3-methyl benzimidazoyl(2)butyric acid
- 4-(1-Methyl-5-morpholino-1H -benzimidazol-2-yl)butanoic acid
- 4-(5-(Bis(2-hydroxyethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acid ethyl ester
- 4-(5-((2-Chloro4-(5-((2-Chloroethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acidethyl)amino)-1-methyl-1H-benzo[d]imidazol-2-yl)butanoic acid
- 5-[(2-Chloroethyl)(2-hydroxyethyl)amino]-1-methyl-1H-benzimidazole-2-butanoic acid HCl
- Ethyl 5-[Bis(2-chloroethyl)amino]-1-methyl-1H-benzimidazole-2-butanoate
- 4-[5-[Bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid isopropyl ester
- N-[(1S)-2-Amino-2-oxo-1-(phenylmethyl)ethyl]-2- pyrazinecarboxamide
- (S)-3-Phenyl-2-[(pyrazine-2-carbonyl) amino]propionic Acid
- (S)-N-(1-(isopentylaMino)-1-oxo-3-phenylpropan-2-yl) pyrazine-2-carboxaMide
- (2S)-N-[(1R)-1-hydroxy-3-methylbutyl]-3-phenyl-2-(pyrazin-2-ylformamido) propanamide
- (2S)-N-[(1S)-1-hydroxy-3-methylbutyl]-3-phenyl-2-(pyrazin-2-ylformamido)propanamide;(S)-Hydroxy Des(boric Acid) Bortezomib
- (RS)-1-(4-Hydroxymethylphenoxy)-3- isopropylaminopropan-2-ol
- (2RS)-3-[4-((2-Isopropoxyethoxy)methyl) phenoxy]-1,2-propanediol
- 4-[((2RS)-2-Hydroxy-3-(isopropylamino)-propyl)oxy]benzaldehyde hydrochloride
- 4-[(2-Isopropoxyethoxy)methyl] phenol
- (2RS)-1-(Isopropylamino)-3- (4-methylphenoxy)propan-2-ol
- 4- Hydroxybenzaldehyde
- 4-[[3-(1-Methylethyl)-2-oxo-5-oxazolidinyl]methoxy] benzaldehyde
- 4-(2,3-Epoxypropoxy) carbazole
- 2-(2-Methoxyphenoxy)ethylamine hydrochloride
- 1-(p-Chloro--phenylbenzyl) piperazine
- 4-[(4-Chlorophenyl)phenylmethyl]-1-piperazineacetic Acid
- 2-[2-[4-[(2-Chlorophenyl)phenylmethyl]-1-piperazinyl]ethoxy]acetic Acid Dihydrochloride
- 1,4-bis((4-chlorophenyl)(phenyl)methyl) piperazine
- RS)-2-[2-[2-[4-[(4-Chlorophenyl)phenylmethyl]piperazin-1-yl]ethoxy] ethoxy]acetic acid
- 2-[2-[4-(Diphenylmethyl)-1-piperazinyl]ethoxy]acetic Acid Hydrochloride
- 4-[(R)-(4-Chlorophenyl)phenylmethyl]-1-piperazine ethanol Hydrochloride
- [2-[4-(4-Chlorophenyl)phenylmethyl]-1-oxido-1-piperazinyl]ethoxy]acetic Acid
- (RS)-2-[2-[4-[(4-Chlorophenyl)phenylmethyl]piperazin-1-yl]ethoxy]acetic acid ethyl ester dihydrochloride
- (RS)-2-[2-[4-[(4-Chlorophenyl)phenylmethyl]piperazin-1-yl]ethoxy]acetic acid methyl ester dihydrochloride
- 3-(2-Chloro-10H-phenothiazin-10-yl)-N,N-dimethylpropan-1-amine S-oxide
- 2-Chloro-10-[3-[[3-(dimethylamino)propyl]methylamino] propyl]phenothiazine
- N,N-Dimethyl-10H-phenothiazine-10-propanamine hydrochloride
- 2-Chloro-N-methyl-10H-phenothiazine-10-propanamine hydrochloride
- 2-Chloro-10H- phenothiazine
- (R)-1-(Naphthalen-1-yl)ethanamine HCl
- (R)-N-Benzyl-1-(naphthalen-1-yl)ethanamine HCl
- (R)-N-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)-N-(3-(3-(trifluoromethyl)phenyl) propyl)propan-1-amine
- R)-N-(1-(Naphthalen-1-yl)ethyl)-3-m-tolylpropan-1- amine hydrochloride
- (R)-N-(1-(Naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl) phenyl)propan-1-amine N-oxide
- 3-(3-Trifluoromethylphenyl)propyl Methanesulfonate
- 3-[3-(Trifluoromethyl) phenyl]propan-1-ol
- 7-chloro-1-cyclopropyl-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
- 1-Cyclopropyl-6-fluoro-7-(piperazin-1-yl) quinolin-4(1H)-one
- 1-cyclopropyl-6-fluoro-7-(4-formylpiperazin-1-yl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
- 11,11'-(Piperazine-1,4-diyl)bis(8-chloro- 5H-dibenzo[b,e][1,4]diazepine)
- 8-Chloro-11-(1-piperazinyl)-5H-dibenzo[b,e][1,4]diazepine Hydrochloride
- 2-Chloro-N-(2-chloroethyl)ethanamine Hydrochloride
- 3-(2-Chloroethyl)octahydro-2-hydroxy-1,3,6,2-oxadiazaphosphonine 2-Oxide
- 3-Aminopropyl dihydrogen phosphate
- 3-[[2-[(2-Chloroethyl)amino]ethyl]amino]propyl Monophosphate Dihydrochloride
- 3-Aminopropan- 1-ol
- 1-(2,6-Dichlorophenyl)-1,3- dihydro-2H-indol-2-one
- 2-[(2,6-Dichlorophenyl)amino] benzaldehyde
- [2-[(2,6-Dichlorophenyl)amino]phenyl] methanol
- 1,3-Dihydro-2H-indol-2- one
- N-(4-Chlorophenyl)-2-(2,6-dichlorophenyl) acetamide
- Ethyl [2-[(2,6-dichlorophenyl)amino]phenyl]acetate
- Methyl [2-[(2,6-dichlorophenyl)amino]phenyl]acetate
- O-(2,6-Dichlorophenylamino)-phenylglyoxalic acid
- N-(2,6-Dichlorophenyl)-N-phenyl-N- chloro
- 5-Chloroacetyl-6-chloro-1,3-dihydro- 2H-indole-2-one
- 6-Methyl-2-(4-methylphenyl)-imidazo[1,2-a]pyridine-3- carboxaldehyde
- 4-Methyl-N-(5-methyl-2-pyridinyl) benzamide
- 5,5-[[2-(3,4-Dimethoxyphenyl)ethyl]imino]bis[2-(3,4-dimethoxyphenyl)-2-(1-methylethyl) pentane nitrile] hydrochloride
- 2RS)-2-(3,4-Dimethoxyphenyl)-5-[[2-(3,4-dimethoxyphenyl)ethyl]amino]-2-(1-methylethyl)pentanenitrile monohydrochloride
- (2RS)-2-(3,4-Dimethoxyphenyl)-2-[2-[[2-(3,4-dimethoxyphenyl)ethyl] (methyl)amino]ethyl]-3-methylbutanenitrile monohydrochloride
- 3,4- Dimethoxybenzaldehyde
- (2RS)-2-(3,4-Dimethoxyphenyl)-5-(methyl amino)-2-(1-methylethyl) pentanenitrile monohydrochloride
- (3,4-Dimethoxyphenyl) methanol
- 3-Chloro-N-[2-(3,4-dimethoxyphenyl)ethyl]-N-methylpropan- 1-amine
- 2-(3,4-Dimethoxyphenyl)-N,N-dimethylethanamine Hydrochloride
- 2-(3,4-Dimethoxyphenyl)-N-methyl ethanamine monohydrochloride
- N,N-Bis[2-(3,4-dimethoxyphenyl)ethyl]-N,N-dimethylpropane-1,3-diamine dihydrochloride
- 1-(2-Ethoxycarbonylbenzofuran-5-yl) piperazine
- Indoles & Oxindoles
- 5- Acetyloxindole
- (3-Acetyl-7-ethyl-1h-indol-1-yl)acetic acid
- 1-Acetyl-1h-indole- 6-carbonitrile
- 2- Acetylindole
- 1-Acetyl-5-bromoindolin- 3-one
- 1-Acetyl- 3-bromoindole
- 1-Acetyl-4-chloro- 1H-indazole
- N-Acetyl-3- hydroxyindole
- (R)-N-Acetyl- 6-fluoro-trp-ome
- 1-Acetylindoline-5-carboxylic acid
- 2-(3-Acetyl-1h-indol-1-yl)propanoic acid
- 1-Acetyl-5-iodo-1h-indol-3-yl acetate
- 1-Acetylindolin- 2-one
- N-Acetylindole- 3-carboxaldehyde
- 4-Acetyl- 7-azaindole
- 1-Acetyl-5-amino-2,3-dihydro- 1H-indole
- N1-(1-Acetyl-2,3-dihydro-1h-indol-5-yl)-4-chloro- 3-nitrobenzamide
- 3-Acetylamino-1h-indole-2-carboxylic acid
- N-Acetylindoline-2-carboxylic acid
- 2-Acetamido-3-(7-chloro-1h-indol-3-yl)propanoic acid
- 1-Acetylindole-2-carboxylic acid
- 1-Acetyl-3-bromo- 4-chloroindole
- 3-Acetyl-7(1H) -azaindole
- (R)-N-Acetyl-5-fluoro-trp- ome
- 3- Acetoxyindole
- 3-Acetyl-1h-indole- 6-carbonitrile
- 1-Acetyl-3, 3-dimethylindoline
- N-Acetyl-5-bromo- 3-hydroxyindole
- (R)-N-Acetyl- 6-chloro-trp-ome
- 1-Acetyl- 3-acetoxyindole
- 1-Acetyl-5-fluoro-4-iodo- 7-azaindole
- 1-Acetyl-2-methylindolin-5-amine hydrochloride
- (R)-N-Acetyl-5-chloro-trp- ome
- (R)-N-Acetyl- 5-fluoro-trp-ome
- 5- Acetylindole
- (3R)-2-Acetyl-2,3,4,9-tetrahydro-1h-beta-carboline-3-carboxylic acid
- N-Acetyl- 5-methyl-dl-tryptophan
- 1-Acetyl-5-bromo-7- nitroindoline
- 1-Acetyl-5-nitro- 1H-indazole
- 7-Acetylamino-4-fluoro-1h- indole
- 5-Acetyl-1h-indole-2-carboxylic acid
- 1-Acetyl-6-fluoro-1h-indol-3-yl acetate
- 1-Acetyl- 6-aminoindoline
- 2-([(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]amino)-3-phenylpropanoic acid
- (1-Acetyl-2,3-dihydro-1h-indol-5-yl)boronic acid
- 1-Acetyl-4-bromo- 1H-indazole
- 3-Acetyl-4-chloro- 7-azaindole
- (S)-1-Acetyl-2,3-dihydro-1h-indole-2-carboxylic acid
- Acetic acid, 2-[(1,3-dihydro-1,3-dioxo-2h-isoindol-2-yl)oxy]-, 2,5-dioxo-1-pyrrolidinyl ester
- 2-(4-Acetylphenyl)-1h-isoindole-1,3(2h)- dione
- 4-([(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]amino)butanoic acid
- 4- Acetoxyindole
- 1-Acetyl-6-chloro- 1H-indazole
- (2R)-1-Acetyl-2,3-dihydro-1h-indole-2-carboxylic acid
- 1- Acetylindoline
- N-Acetyl-DL- Tryptophan
- N-Acetyl-5-methoxy- l-tryptophan
- 2-[2-(Acetylamino)phenyl]-n-[2-(1h-indol-3-yl)ethyl]- 2-oxoacetamide
- 1-Acetyl-5-bromo-4-chloro-1h-indol-3-yl acetate
- (3-Acetyl-2-methyl-indol-1-yl)-acetic acid
- 5-Acetyl-3-methyl-1h-indole-2-carboxylic acid
- 1-Acetyl-2-methylindoline-5-sulphonyl chloride
- 1-Acetyl-2-methyl- 5-nitroindoline
- 1-Acetyl- 5-bromoindole
- 1-Acetyl- 3-indolinone
- N-Acetyl- 3-hydroxyindole
- 3-(3-Acetyl-1h-indol-1-yl)propanoic acid
- Chemical Name: ([(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]amino)(phenyl)acetic acid
- (3R)-2-Acetyl-1-phenyl-2,3,4,9-tetrahydro-1h-beta-carboline-3-carboxylic acid
- (3-Acetyl-1h-indol-1-yl)acetic acid
- 2-(4-Acetylphenyl)-1h-isoindole-1, 3(2h)-dione
- 1-Acetyl-5-bromo-1h-indole-2, 3-dione
- 1-Acetyl- 5-nitroindoline
- 3- Acetylindole
- 1- Acetylindole
- 1-Acetyl-7-amino-2,3-dihydro- 1h-indole
- N-Acetyl-D- Tryptophan
- (R)-N-Acetyl-6-fluoro-trp- ome
- 7- Acetoxyindole
- 1- Acetylisatin
- 1-Acetyl-5-bromo-4-chloro- pseudoindoxyl
- 3-[(1-Acetyl-2,3-dihydro-1h-indol-5-yl)sulfonyl]propanoic acid
- 1-Acetyl-2,3-dihydro-1H-indole-5-sulfonyl chloride
- 2-Hydroxy benzoic acid
- Nitro Compounds
- Sulfonamides
- 1,2-Dimethyl-1h-imidazole-5-sulphonyl chloride
- Dansyl fluoride
- 3,4- Difluorobenzenesulfonamide
- N-Allyl-4-aminobenzene sulfonamide
- 3- Cyanobenzenesulfonamide
- 2,5-Dimethoxybenzenesulfonyl chloride
- 2- Cyanobenzenesulfonamide
- Alpha -(p-toluenesulfonyl)-4- fluorobenzylisonitrile
- N,4-Dimethyl-n-(oxiran-2-ylmethyl) benzenesulfonamide.
- 1-(2-Cyanophenyl)-N- pentylmethanesulfonamide
- 2-Allyloxy-1,1,2,2-tetrafluoroethane sulfonyl fluoride
- 1-(2-Cyanophenyl)-N- cyclohexylmethanesulfonamide
- Alpha - toluenesulfonylchloride
- 3-(5-Diethylsulfamoyl-1-ethyl-1h-benzoimidazol-2-yl)-propionic acid
- N-(4-Acetylphenyl) methane sulfonamide
- Cyclopropylmethane sulfonamide
- N,N-Dimethyl 3-bromo-5-methylbenzenesulfonamide
- 4-Cyanobenzene-1- sulfonamide
- 4-(Dimethylamino)- 3-nitrobenzenesulfonamide
- 3-(2,3-Dihydro-indole-1-sulfonyl)-benzoic acid
- 3-(3,4-Dihydro-2h-quinoline-1-sulfonyl)-4-methoxy-benzoic acid
- N,N-Dimethyl 4-fluorobenzenesulfonamide
- N-Cyclohexyl 3-boronobenzenesulfonamide
- (3-Cyanophenyl)methanesulfonyl chloride
- (Dimethylsulfamoyl) methylamine
- 4-Amino-N-cyclopropylbenzene sulfonamide
- Difluoromethanesulfonyl chloride
- N-(3-Acetylphenyl) benzene sulfonamide
- 2,6-Dimethylmorpholine-4-sulfonyl chloride
- 3,5-Difluorobenzenesulfonyl chloride
- 4-(Difluoro-methanesulfonyl)-benzoic acid
- 4-Amino -n- utylbenzene sulfonamide
- N-Cyclohexyl-2- nitrobenzenesulfonamide
- N,N-Diethyl 5-bromo-2-methoxybenzenesulfonamide
- 4-Acetylbenzenesulfonyl chloride
- 3,4-Dimethyl-(4H)1,4-benzothiazine-1,1- dioxide
- 4-cyano-N- methylbenzenesulfonamide
- 2-Amino-6-chloro-n-(2,4-dimethylphenyl)benzene sulfonamide
- N-(3-Acetylphenyl) methane sulfonamide
- N-(2-Acetylphenyl) benzene sulfonamide
- N-Cyclopropyl 3-nitrobenzenesulfonamide
- 4-Acetamidobenzene sulfonamide
- 4-Acetamido-3-chlorobenzenesulfonyl chloride
- 2-Acetamido-5-ethylbenzenesulfonyl chloride
- 5-Acetonyl-2-methoxybenzene sulfonamide
- 4-(Acetylamino)benzenesulfonyl fluoride
- 5-(Acetylamino)-2-methoxybenzenesulfonyl chloride
- 2-(Acetylamino)-5-methylbenzenesulfonyl chloride
- 4-Acetylamino-2-methyl-benzenesulfonyl chloride
- 4-Acetylbenzene sulfonamide
- 3-Acetyl- 5-chlorothiophene- 2-sulfonamide
- 1-Acetyl-2,3-dihydro -1H-indole -5-sulfonyl chloride
- 4-Acetyl-n- (4-fluorobenzyl) benzenesulfonamide
- 4-Acetyl-n -(2-furylmethyl)benzene sulfonamide
- 4-Acetyl-n-(2-furylmethyl) benzenesulfonamide
- N-(5-Acetyl-2-hydroxy-phenyl)- methanesulfonamide
- 3-(4-Acetyl-piperazine-1-sulfonyl)-benzoic acid
- 4-(4-Acetylpiperazinosulfonyl) Bromobenzene
- Acetyl sulfamethoxazole
- 1-Adamantaneethyl sulfonamide
- 4-(Adamantan-1-yl)phenyl trifluoromethane sulfonate
- N-1-Adamantyl-4-aminobenzene sulfonamide
- N-[2-(1-Adamantyloxy)propyl]methane sulfonamide
- alfa-(4-Toluenesulfonyl)- 3-methoxybenzylisocyanid
- alfa-(4-Toluenesulfonyl)-4- methoxybenzylisocyanide
- 4-[(Allylamino) sulfonyl]benzoic acid
- N-Allyl-4-isothiocyanato benzene sulfonamide
- N-Allylmethane sulfonamide
- N-(Allyloxy)-2-nitrobenzene sulfonamide
- 1-Allyl-N-tert-butyl cyclopropane-1-sulfonamide
- N-Allyl -p -toluenesulfonamide
- Alpha-sulfophenylacetic acid
- Alpha -toluenesulfonamide
- Alpha- toluenesulfonamide
- Alpha- (p-toluenesulfonyl)- 4- fluorobenzylisonitrile
- N-(4-(2-Aminoacetyl)-5-methoxy-2-phenoxyphenyl)methanesulfonamide hydrochloride
- 2-Aminobenzene sulfonamide
- 3-Aminobenzene sulfonamide
- 4-aminobenzene -1- sulfonamide
- 2-Aminobenzene sulfonic acid (1-methylethylidene) di-4,1-phenylene ester
- (4-Amino-benzenesulfonylamino) -acetic acid
- 2-(3-Amino-benzenesulfonylamino)-benzoic acid
- 3-Aminobenzene-1-sulfonyl fluoride
- 2- [4-(2-Aminobenzenesulfonyl) piperazin-1-yl] ethanol
- 1-Amino benzimidazole-2 -sulfonic acid
- 2-Amino- 1,3-benzothiazole- 6-sulfonamide
- 4-Amino-n- benzylbenzene sulfonamide
- 4-Amino-n- benzyl-n- methylbenzenesulfonamide
- 2-Amino-5-bromobenzene sulfonamide
- N -(2-Amino-3-bromophenyl) methanesulfonamide
- 6-Amino-5-bromopyridine-3-sulfonic acid
- 2-Amino-N-butyl benzene sulfonamide
- 4-Amino-n-butylbenzene sulfonamide
- 2-Amino -N- butylbenzene sulfonamide hydrochloride
- N-(4-Aminobutyl)-5-chloro-2- naphthalenesulfonamide hydrochloride
- 3-Amino-N-butyl- 4-methylbenzene sulfonamide
- 4-Amino-n-butyl-n-methylbenzene sulfonamide
- N-(4-Aminobutyl)-2-naphthalene sulfonamide hydrochloride
- 4-Amino-6-chloro-1,3-benzenedi sulfonamide
- 4-Amino-6-chloro-benzene-1,3-disulfonyl dichloride
- 2-Amino-5-chlorobenzene sulfonamide
- 3-Amino-4-chloro-n-(2-chloro-phenyl)-benzene sulfonamide
- 5-Amino-2-chloro-n-(4-chloro-phenyl)-benzene sulfonamide
- 3-Amino-4-chloro-n,n-diethyl-benzene sulfonamide
- 5-Amino-2-chloro-n,n-diethyl-benzene sulfonamide
- 5-Amino-2-chloro-n,n-dimethyl-benzene sulfonamide
- 4-Amino-2-chloro-5-hydroxybenzen sulfonamide
- 3-Amino-n-(2-chloro-phenyl)-benzene sulfonamide
- 3-Amino-n-(4-chloro-phenyl)-benzene sulfonamide
- 4-Amino-n- (3-chlorophenyl) benzene sulfonamide
- 4-Amino-n-(4-chloro-phenyl)-benzene sulfonamide
- 3-Amino-n-(2-chloro-phenyl)-4-hydroxy-benzene sulfonamide
- 3-Amino-n-(3-chloro-phenyl)-4-hydroxy-benzene sulfonamide
- N-(2-Amino-5-chlorophenyl)methane sulfonamide
- 3-Amino-n-(3-chloro-phenyl)-4-methyl-benzene sulfonamide
- 3-Amino-n-(4-chloro-phenyl)-4-methyl-benzene sulfonamide
- 5-Amino-n-(2-chloro-phenyl)-2-morpholin-4-yl-benzene sulfonamide
- benzene sulfonamide
- Sulfachloropyridazine sodium
- 3-Amino-N-cyclohexyl-4-methylbenzene sulfonamide
- 4-Amino-N-cyclohexyl-N-methyl-benzene sulfonamide
- 3-Amino-N-cyclopropyl-4-methylbenzene sulfonamide
- 4-Amino-n-(cyclopropylmethyl)benzene sulfonamide
- 5-Amino-N-cyclopropyl-2-methylbenzene sulfonamide
- 2-Chloro-5-(trifluoromethyl)benzenesulfonyl chloride
- 4-Chloro-3-(trifluoromethyl)benzenesulfonyl chloride
- 4-Chloro-2-(trifluoromethyl)benzenesulphonyl chloride
- 3-Chloro-n,n,4-trimethylbenzene-1- sulfonamide
- 2-Cyanobenzenesulfonyl chloride
- 3-Cyanobenzenesulfonyl chloride
- 4-Cyanobenzenesulfonyl chloride
- N'-[(4-Cyanobenzene)sulfonyl]-N,N- dimethylmethanimidamide
- 2-cyanobenzenesulfonyl fluoride
- 3-cyanobenzenesulfonyl fluoride
- 4-cyanobenzenesulfonyl fluoride
- N-(2-(4'-Cyano-[1,1'-biphenyl]-4-yl)propyl)propane-2- sulfonamide
- 3-Cyano-N- cyclopropylbenzenesulfonamide
- 3-Cyano-N,N- dimethylbenzenesulfonamide
- 4-Cyano-N,N- dimethylbenzenesulfonamide
- 2- cyanoethanesulfonamide
- 3-cyano-N-ethylbenzene-1- sulfonamide
- 2-Cyano-5-fluorobenzene-1-sulfonyl chloride
- 3-Cyano-4-fluorobenzenesulfonyl chloride
- N-(2-Cyano-4-fluoro-phenyl)- methanesulfonamide
- N-(4-cyano-2-fluorophenyl) methanesulfonamide
- 3-Cyano-4-fluoro-n-(thiazol-2-yl) benzenesulfonamide
- N-(4-Cyano-2-iodo-5-(trifluoromethyl)phenyl) methanesulfonamide
- 3-Cyano-N- isopropylbenzenesulfonamide
- 2-Cyano-5-methoxybenzene-1-sulfonyl chloride
- 3-Cyano-N- methylbenzenesulfonamide
- 2-Cyano-5-methylbenzenesulfonyl chloride
- 3-Cyano-n- phenylbenzenesulfonamide
- 1-(4-Cyanophenyl)-N- cyclohexylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- cyclopropylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- cyclopropylmethanesulfonamide
- 1-(2-Cyanophenyl)-N,N- diethylmethanesulfonamide
- 1-(4-Cyanophenyl)-N,N- diethylmethanesulfonamide
- 1-( 2-Cyanophenyl)-N,N-dimethylmethanesulfonamide
- 1-( 4-Cyanophenyl)-N,N-dimethylmethanesulfonamide
- 1-(2-Cyanophenyl )-N-ethylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- ethylmethanesulfonamide
- N-(2-Cyanophenyl)-N- ethylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- isopropylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- isopropylmethanesulfonamide
- N-(2-Cyanophenyl)-N- isopropylmethanesulfonamide
- 1-(2-Cyanophenyl) methanesulfonamide
- 1-(4-Cyanophenyl) methanesulfonamide
- (2-Cyanophenyl)methanesulfonyl chloride
- (4-Cyanophenyl)methanesulfonyl chloride
- 1-(2-Cyanophenyl)-N-(2-methoxyethyl) methanesulfonamide
- 1-(4-Cyanophenyl)-N-(2-methoxyethyl) methanesulfonamide
- 1-(2-Cyanophenyl)-N-methoxy-N- methylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- methylmethanesulfonamide
- 1-(4-Cyanophenyl)-n- methylmethanesulfonamide
- 1-(2-Cyanophenyl)-N-(2-methylpropyl) methanesulfonamide
- 1-(4-Cyanophenyl)-N-(2- methylpropyl)methanesulfonamide
- 1-(2-Cyanophenyl)-N- phenylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- phenylmethanesulfonamide
- 1-(2-Cyanophenyl)-N- propylmethanesulfonamide
- 1-(4-Cyanophenyl)-N- propylmethanesulfonamide
- ([(4-Cyanophenyl)sulfonyl]amino) acetic acid
- 4- Cyanophenylthiourea
- N-Cyano-N-phenyl-p- toluenesulfonamide
- N-(5-(5-Cyanothiophen-2-yl)-2-methylphenyl)-4- methylbenzenesulfonamide
- Cyclamic acid
- N,N'-(1S,2S)-1,2-Cyclohexanediylbis[2-hydroxy-7,7-dimethyl-(1r,1'r,2s,2's,4s,4's)-bicyclo[2.2.1]heptane-1 -methanesulfonamide]
- Cyclohexane sulfonamide
- Cyclohexanone tosylhydrazone
- N-Cyclohexyl 2-aminobenzenesulfonamide
- N-Cyclohexyl 3-aminobenzenesulfonamide
- N-Cyclohexyl 4-aminobenzenesulfonamide
- 3-(Cyclohexylamino)-2-hydroxy-1-propanesulfonic acid
- 3-(Cyclohexylamino)propane-1-sulfonic acid
- 4-Cyclohexylbenzenesulfonyl chloride
- N-Cyclohexyl 4- boronobenzenesulfonamide
- N-Cyclohexyl 3- bromobenzenesulfonamide
- N-Cyclohexyl 4- bromobenzenesulfonamide
- N-Cyclohexyl 3-bromo-4- methylbenzenesulfonamide
- N-Cyclohexyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N-Cyclohexyl-n-(1,1-dioxidotetrahydrothien-3-yl)amine hydrochloride
- N-Cyclohexyl 4-fluorobenzenesulfonamide
- N-Cyclohexyl-3-hydroxybenzene-1- sulfonamide
- N-Cyclohexyl-4-hydroxybenzene-1- sulfonamide
- N-cyclohexyl-4-hydroxy-N-[(4-methoxyphenyl)methyl]benzene-1- sulfonamide
- N-Cyclohexyl-4- isothiocyanatobenzenesulfonamide
- 1-Cyclohexyl-3-({4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-1,2,3,4-tetrahydroisoquinolin-2-yl) ethyl]benzene}sulfonyl)urea
- N-Cyclohexyl-4-methyl-3- nitrobenzenesulfonamide
- N-Cyclohexyl 3-nitrobenzenesulfonamide
- N-Cyclohexyl-4- nitrobenzenesulfonamide
- N-Cyclohexyl-2-oxo-3- oxazolidinesulfonamide
- 3-(Cyclohexylsulfamoyl)benzoic acid
- 4-(N-Cyclohexylsulfamoyl)-2-trifluoromethylphenylboronic acid
- N- Cyclohexyltaurine
- N- Cyclohexyl-p-toluenesulfonamide
- 4- (Cyclopentanesulfonyl)aniline
- 2-(Cyclopentanesulfonyl)aniline hydrochloride
- Cyclopentanesulfonyl chloride
- 4-(Cyclopentylsulfanyl) benzaldehyde
- 3-(Cyclopentylsulfonyl) aniline
- 1-Cyclopentyl-1-tosylmethyl isocyanide
- 1,1-Cyclopropane dimethanol cyclic sulfite
- Cyclopropane sulfonamide
- Cyclopropanesulfonyl chloride
- N-Cyclopropyl 2-aminobenzenesulfonamide
- N-Cyclopropyl 3-aminobenzenesulfonamide
- 4-[(Cyclopropylamino)sulfonyl]benzoic acid
- N-Cyclopropyl 3-boronobenzenesulfonamide
- N-Cyclopropyl 4-boronobenzenesulfonamide
- N-Cyclopropyl 4-bromobenzenesulfonamide
- N-Cyclopropyl 4-bromo-3-methylbenzenesulfonamide
- N-Cyclopropyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N-Cyclopropyl 2-bromo-4-trifluoromethoxybenzenesulfonamide
- N-Cyclopropyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N-cyclopropyl-2-(1,3-dioxoisoindol-2-yl) ethanesulfonamide
- N-Cyclopropyl 4-fluorobenzenesulfonamide
- N-Cyclopropyl-3-hydroxybenzene-1- sulfonamide
- N-Cyclopropyl-4-hydroxybenzene-1- sulfonamide
- N-cyclopropyl-3-hydroxy-N-[(4-methoxyphenyl)methyl]benzene-1- sulfonamide
- N-cyclopropyl-4-hydroxy-N-[(4-methoxyphenyl)methyl]benzene-1 -sulfonamide
- 3-(Cyclopropylmethanesulfonyl) aniline
- 1-(Cyclopropylmethyl)cyclopropane-1- sulfonamide
- N-Cyclopropyl-2-methyl-5- nitrobenzenesulfonamide
- N-Cyclopropyl-4-methyl-3- nitrobenzenesulfonamide
- 3-(Cyclopropylmethyl) sulfonylazetidine
- N-Cyclopropyl-2- nitrobenzenesulfonamide
- N-Cyclopropyl-4- nitrobenzenesulfonamide
- N-Cyclopropyl-1-(3-nitrophenyl) methanesulfonamide
- 2-(4-Cyclopropylpiperazine-1-sulfonyl) aniline
- Dabrafenib mesylate
- N,N-Diallyl-4 -methylbenzenesulfonamide
- 3,4- Diaminobenzenesulfonamide
- 3,4-Diaminobenzenesulfonamide dihydrochloride
- 3,4-Diaminobenzenesulfonic acid
- 4,4'-Diaminobiphenyl-2,2'-disulfonic acid hydrate
- 2,5-Diamino-n-(2-chlorophenyl) benzenesulfonamide
- 3,4-Diamino-n-(2-chloro-phenyl) -benzenesulfonamide
- 3,4-Diamino-n-(4-chloro-phenyl)- benzenesulfonamide
- 3,4-Diamino-n,n-diethyl- benzenesulfonamide
- 3,4-Diamino-N,N -dimethylbenzenesulfonamide
- 3,4-Diamino-n-(1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)- benzenesulfonamide
- (2R,4R)-1-[(2S)-5-[(Diaminomethylidene)amino]-2-[(3R)-3-methyl-1,2,3,4-tetrahydroquinoline-8-sulfonamido]pentanoyl]-4-methylpiperidine-2-carboxylic acid
- 3-[({2-[(Diaminomethylidene)amino]-1,3-thiazol-4-yl}methyl)sulfanyl]-N- sulfamoylpropanimidamid
- 3,4-Diamino-n-(3-trifluoromethyl-phenyl)- benzenesulfonamide
- Dibenzene sulfonimide
- Dibenzo[b,d]furan-2-sulfonyl chloride
- N,N-Dibenzyl-1 -phenylmethanesulfonamide
- 2,4-Dibromobenzenesulfonyl chloride
- 2,5-Dibromobenzenesulfonyl chloride
- 1,2-Dibromo-4- methanesulfonylbenzene
- 1,3-Dibromo-5- methanesulfonylbenzene
- 3,4- Dibromosulfolane
- 4,5-Dibromothiophene-2-sulfonyl chloride
- 2,4 -Dichlorobenzenesulfonamide
- 3,5- Dichlorobenzenesulfonamide
- 2-(3,4-Dichloro-benzenesulfonylamino)-benzoic acid
- 2,3-Dichlorobenzenesulfonyl chloride
- 2,4-Dichlorobenzenesulfonyl chloride
- 3,5-Dichlorobenzenesulfonyl chloride
- 3',4'-Dichloro[1,1'-biphenyl]-4-sulfonyl chloride
- 1,4-Dichloro-2- [(2-chloroethane)sulfonyl]benzene
- 2,4-Dichloro-7, 8-dihydro-5H-S,S-di-oxoisothiopyrano[4,3-d]pyrimidine
- 2,2-Dichloro-N-[(1R,2S)-3-fluoro-1-hydroxy-1 -(4-methanesulfonylphenyl)propan-2-yl]acetamide
- 2,6-Dichloro-3- methanesulfonylpyridine
- 2,4-Dichloro-6-methylbenzenesulphonyl chloride
- 1,2-Dichloro-4- methylsulfonylbenzene
- 1,4-Dichloro-2-[(2-chloroethane)sulfonyl] benzene
- 2,4-Dichloro-7,8-dihydro-5H-S,S-di-oxoisothiopyrano[4,3-d] pyrimidine
- 2,2-Dichloro-N-[(1R,2S)-3-fluoro-1-hydroxy-1-(4-methanesulfonylphenyl)propan-2-yl] acetamide
- N,N-Diethyl 2-aminobenzenesulfonamide
- N,N-Diethyl 3-aminobenzenesulfonamide
- N,N-Diethyl 4-aminobenzenesulfonamide
- 5-[(Diethylamino)sulfonyl]-2-morpholin-4-ylbenzoic acid
- 1-(4-[(Diethylamino)sulfonyl]phenyl)-5-oxopyrrolidine-3-carboxylic acid
- N,N-Diethyl-4,4'-bipiperidine-1- sulfonamide
- N,N-Diethyl 2-boronobenzenesulfonamide
- N,N-Diethyl 3-boronobenzenesulfonamide
- N,N-Diethyl 4-boronobenzenesulfonamide
- N,N-Diethyl 5-borono-2-methoxybenzenesulfonamide
- N,N-Diethyl 3-borono-4-methylbenzenesulfonamide
- N,N-Diethyl 3-bromobenzenesulfonamide
- N,N-Diethyl 4-bromobenzenesulfonamide
- N,N-Diethyl 3-bromo-4-methylbenzenesulfonamide
- N,N-Diethyl 3-bromo-5-methylbenzenesulfonamide
- N,N-Diethyl 4-bromo-3-methylbenzenesulfonamide
- N,N-Diethyl 2-Bromo-4-trifluoromethoxybenzenesulfonamide
- N,N-Diethyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N,N-Diethyl 4-bromo-3-trifluoromethylbenzenesulfonamide
- N,N-Diethyl-4- fluorobenzenesulfonamide
- N,N-Diethyl-3-hydrazinocarbonyl- benzenesulfonamide
- N,N-Diethyl-2-hydroxybenzene-1- sulfonamide
- N,N-Diethyl-3-hydroxybenzene-1- sulfonamide
- N,N-Diethyl-4-hydroxybenzene-1- sulfonamide
- N,N-Diethyl-4 -isothiocyanatobenzenesulfonamide
- N,N-Diethyl-4-methyl-3 -nitrobenzenesulfonamide
- N,N-Diethyl-7-methyl-3-oxo-3,4-dihydro-2h-1,4- benzoxazine-6-sulfonamide
- N,N-Diethyl-2- nitrobenzenesulfonamide
- N,N-Diethyl 3-nitrobenzenesulfonamide
- N,N-Diethyl 4-nitrobenzenesulfonamide
- N,N-Diethyl-3-nitro-4-piperazin-1 -ylbenzenesulfonamide
- N,N-Diethyl(sodiosulfanyl) carbothioamide
- 3-Diethylsulfamoylbenzoic acid
- 4-Diethylsulfamoyl-benzoic acid
- [4-(diethylsulfamoyl)-3-methylphenyl]boronic acid
- 4-(N,N-Diethylsulfamoyl)-2-methylphenylboronic acid
- 3-[4-(Diethylsulfamoyl)phenyl]propanoic acid
- N,N-Diethyl-p- toluenesulfonamide
- Diethyl (tosyloxy)methylphosphonate
- 2,4 -Difluorobenzenesulfonamide
- 2,5- Difluorobenzenesulfonamide
- 2,6- Difluorobenzenesulfonamide
- 3,5- Difluorobenzenesulfonamide
- 2,5-Difluorobenzenesulfonyl chloride
- 2,6-Difluorobenzenesulfonyl chloride
- 3,4-Difluorobenzenesulfonyl chloride
- 2,2-Difluoroethyl trifluoromethanesulfonate
- N-(2,4-Difluoro-3-formylphenyl) propane-1-sulfonamide
- Difluoromethanesulfonamide .
- 3-(Difluoromethoxy)benzenesulfonyl chloride
- 1,3-Difluoro-5 -methylsulfonylbenzene
- 2,4-Difluoro-5-nitrobenzenesulfonyl chloride
- 3-(2,4-Difluorophenylsulfonylamino)-2-methoxypyridine-5-boronic acid pinacol ester
- 2,3-Dihydro-benzo[1,4]dioxine-6-sulfonic acid amide
- 6-(2,3-Dihydro-benzo[1,4]dioxine-6-sulfonylamino)-hexanoic acid
- 2,3-Dihydro-1-benzofuran-5- sulfonamide
- 2,3-Dihydro-1-benzofuran-5-sulfonyl chloride
- 3,4-Dihydro-2h-1,4-benzothiazine 1,1-dioxide
- 2,3-Dihydro-1,1-dioxo-1,2- benzisothiazole
- (4R,6S)-5,6-Dihydro-4-hydroxy-6-methylthieno[2,3-b]thiopyran-7,7- dioxide
- 5-(2,3-Dihydro-indole-1-sulfonyl)-2-methoxy-benzoic acid
- 3,4-Dihydroisoquinoline-2(1h)-sulfonyl chloride
- 2-((R)-4-((3R,5S,7S,8R,9S,10S,13R,14S,17R)-3,7-Dihydroxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)pentanamido)ethane-1-sulfonic acid
- N,N-Diisopropyl 4-(4-Bromopyrazol-1-yl)benzenesulfonamide
- N,N-Diisopropyl 4-fluorobenzenesulfonamide
- N,N-Diisopropyl-4- isothiocyanatobenzenesulfonamide
- 2,4- Dimethanesulfonylbenzaldehyde
- 2,5-dimethanesulfonylbenzoic acid
- 3,4- Dimethanesulfonylbenzonitrile
- 1,2 -Dimethanesulfonyl-4-nitrobenzene
- 1,2 -Dimethanesulfonylpyrrole
- 1,4-Dimethanesulfonyl- 2-(trifluoromethyl)benzene
- 2,5- Dimethoxybenzenesulfonamide
- 2,4-Dimethoxybenzenesulfonyl chloride
- N-(2,2-Dimethoxyethyl)-4 -methylbenzenesulfonamide
- 2-(2,5- Dimethoxyphenylthio)benzaldehyde
- 2-({[(4,6-Dimethoxypyrimidin-2-yl)carbamoyl]amino}sulfonyl)-N,N-dimethylpyridine-3- carboxamide
- N-((4,6-Dimethoxypyrimidin-2-yl)carbamoyl)-3-(ethylsulfonyl)pyridine-2- sulfonamide
- 2,3-Dimethoxy-5-sulfamoylbenzoic acid
- 4-(Dimethylamino) benzenesulfonamide
- N,N-Dimethyl 4-aminobenzenesulfonamide
- 2-(Dimethylamino)-N,N-dimethyl-5- nitrobenzenesulfonamide
- 5-Dimethylamino-1- naphthalenesulfonamide
- 4-(Dimethylamino)-3- nitrobenzenesulfonamide
- 4-[(Dimethylamino)sulfonyl]benzoic acid
- ((3-[(Dimethylamino)sulfonyl]benzoyl)amino)acetic acid
- 3-(5-[(Dimethylamino)sulfonyl]-1-ethyl-1h-benzimidazol-2-yl)propanoic acid
- 5-[(Dimethylamino)sulfonyl]-2-[(2-hydroxyethyl)amino]benzoic acid
- 2,5-Dimethyl- benzenesulfonamide
- N,4- Dimethylbenzenesulfonamide
- N,N- Dimethylbenzenesulfonamide
- 2,5-Dimethylbenzenesulfonyl chloride
- 3,5-Dimethylbenzenesulfonyl chloride
- N,N-Dimethyl benzoimidazole-1-sulfonamide
- N,N-Dimethyl-4,4'-bipiperidine-1- sulfonamide
- N,N-Dimethyl 2-boronobenzenesulfonamide
- N,N-Dimethyl 3-boronobenzenesulfonamide
- N,N-Dimethyl 4-boronobenzenesulfonamide
- N,N-Dimethyl 3-borono-4-methylbenzenesulfonamide
- N,N-Dimethyl 2-bromobenzenesulfonamide
- N,N-Dimethyl 3-bromobenzenesulfonamide
- N,N-Dimethyl 4-bromobenzenesulfonamide
- N,N-Dimethyl 5-bromo-2-methoxybenzenesulfonamide
- N,N-Dimethyl 3-bromo-4-methylbenzenesulfonamide
- N,N-Dimethyl 4-(4-bromopyrazol-1-yl)benzenesulfonamide
- N,N-Dimethyl 5-bromopyridine-3-sulfonamide
- N,N-Dimethyl 3-bromo-5-trifluoromethylbenzenesulfonamide
- N,N-Dimethyl 4,5-diiodo-1H-imidazole-1-sulfonamide
- 3-(3,5-Dimethyl-1,1-dioxo-1,2-dihydro-1lambda6-[1,2,6]thiadiazin-4-yl)-propionic acid
- Dimethyl 2,2'-dithiobisbenzoate
- N,N-Dimethyl imidazole-1-sulfonamide
- N,N-Dimethyl 4-iodo-1H-imidazole-1-sulfonamide
- 3,5-Dimethylisoxazole-4- sulfonamide
- 3,5-Dimethylisoxazole-4-sulfonyl chloride
- 4,6-Dimethyl-2- methylsulfonylpyrimidine
- 4-(2,6-Dimethyl-morpholine-4-sulfonyl)-benzoic acid
- 2,5-Dimethyl-1-[3-(morpholine-4-sulfonyl)-phenyl]-1h-pyrrole-3- carbaldehyde
- N,N-Dimethylnicotinamide- 2-sulfonamide
- N,2-Dimethyl-5- nitrobenzenesulfonamide
- N,4-Dimethyl-3 -nitrobenzenesulfonamide
- N,N-Dimethyl 3-nitrobenzenesulfonamide
- N,N-Dimethyl-4- nitrobenzenesulfonamide
- N,N-Dimethyl-1-(3-nitrophenyl) methanesulfonamide
- N-(1,5-Dimethyl-3-oxo-2-phenyl-2,3-dihydro-4-pyrazolyl)butane-1- sulfonamide
- 4,N-Dimethyl-n-phenyl- benzenesulfonamide
- N-(2,4-Dimethyl-phenyl)-4-hydrazino-3-nitro- benzenesulfonamide
- (2,4-Dimethylphenyl)(isocyano)methyl 4-methylphenyl sulfone
- N-(2,6-Dimethylphenyl)-4- methylbenzenesulfonamide
- 4-(3,4-Dimethyl-phenylsulfamoyl)-benzoic acid
- (2,4-Dimethylphenyl) thiourea
- 3,5 -Dimethylphenylthiourea
- N,N-Dimethylpiperidine-4- sulfonamide
- {4-[(2,2-dimethylpropoxy)sulfonyl]-3-methylphenyl}boronic acid
- 4-(3,5-Dimethyl-pyrazol-1-yl)- benzenesulfonamide
- N,N-Dimethylpyridine-3- sulfonamide
- N,N-Dimethylpyrrolidine-3- sulfonamide
- N,N-Dimethylpyrrolidine-3-sulfonamide hydrochloride
- N,N -Dimethylsulfamide
- 4 -(Dimethylsulfamoyl)benzamide
- 4-(Dimethylsulfamoyl)benzene-1-sulfonyl chloride
- 3-Dimethylsulfamoylbenzoic acid
- 4-[(Dimethylsulfamoyl)methyl]benzoic acid
- thiazoles
- 2-(2-Aminobenzo[d]thiazol-6-yl)acetonitrile hydrobromide
- 2-Amino-4-bromo- 7-fluorobenzothiazole
- 2-Amino-6-bromo -4-fluorobenzothiazole
- N- (5-Acetyl-1,3-thiazol-2-yl)-2,2-dimethylpropanamide
- 2-(5-acetyl-4-methyl-1,3-thiazol-2-yl) guanidine
- 2-Amino- 6-benzothiazolol hydrochloride
- N- (2-Amino-benzothiazol-6-yl)-acetamide
- 4-(1-Adamantyl) -2-methylthiazole
- 2-Acetamido-4- (4-hydroxyphenyl)thiazole
- 1- (2-Aminobenzo[d]thiazol-6-yl)ethanone
- 1- (2-Amino-benzothiazol-4-yl)-ethanone
- 2-Amino- 5-bromobenzothiazole 3-oxide
- 2-Amino- 5-bromo-4-cyclopropylthiazole
- 2-Amino- 4-bromo-6-fluorobenzothiazole
- 2-Amino- benzothiazol-6-ol
- 2-Acetylamino-benzothiazole-6-carboxylic acid
- N-Allylbenzothiazolium bromide
- 2-Amino- 5-benzothiazolecarboxylic acid
- (2-Amino-benzothiazol-6-yl)-acetic acid ethyl ester acetate
- 2-(2-Aminobenzo[d] thiazol-6-yl)acetonitrile
- (2-Amino-benzothiazol-6-yl)-acetic acid ethyl ester
- (2-Amino-benzothiazol-6-yl)-acetic acid methyl ester
- 2-Amino- 5-bromobenzothiazole
- 2-Amino-5-bromo- 4-isopropylthiazole
- (Z)-2-Amino-alpha-[1-(tert-butoxycarbonyl)]-1-methylethoxyimino-4-thiazolacetic acid
- (2-Aminobenzothiazol-6-yl) acetic acid
- 6- Aminobenzothiazole
- 4-(1-Adamantyl)- 1,3-thiazol-2-amine
- 2-Acetylamino-4-(chloromethyl) thiazole
- 2- Amino-benzothiazole-6-carbonitrile
- 2-Amino-4-bromo-6- fluorobenzothiazole
- 5-Acetyl- 2,4-dimethylthiazole
- 2-Amino- 5-bromo-4-t-butylthiazole
- 6-Amino-7- bromobenzothiazole
- 2-Amino-6-bromo-1,3- benzothiazole
- 2-Acetamido-5-bromo -4-ethylthiazole
- Abafungin .
- 2-Acetamido-5-bromo-4- ethylthiazole
- 2-Acetamido-4-(4-bromophenyl) thiazole
- 2-Acetamido- 5-bromothiazole
- 2-Acetamido- 5-chlorothiazole
- 2-Acetamido-4- (2-hydroxyphenyl)thiazole
- 2-Acetamido -4-methylthiazole
- 2-Acetamido- 5-methylthiazole
- 2-Acetamido-4-methylthiazole-5-sulfonyl chloride
- 2-Acetamido -5-nitrothiazole
- 2- Acetamidothiazole
- 2-acetamido-1,3-thiazole-5-carboxylic acid
- (2-acetamido-1,3-thiazol-4-yl)acetic acid
- Acetone-benzothiazolyl -2-hydrazone
- Acetone-benzothiazoly l-2-hydrazone
- 2-(Acetylamino)-1,3-benzothiazole-6-carboxylic acid
- 2-acetamido-1,3-benzothiazole-6-carboxylic acid
- [2-(([4-(Acetylamino)phenyl]sulfonyl)amino)-1,3-thiazol-4-yl]acetic acid
- 2-Acetylamino-5-thiazolecarboxylic acid methyl ester
- 2-Acetylamino-thiazole-5-sulfonyl chloride
- 2- Acetylbenzothiazole
- 6- Acetylbenzothiazole
- N-(5-Acetyl-4-methyl- 1,3-thiazol-2-yl)acetamide
- 1 -(5-Acetyl-4-methylthiazol-2-yl)imidazolidin-2-one
- 2- (1-Acetylpyrrolidin-2-yl)-5-bromo-4-methyl-1,3- thiazole
- 2- Acetylthiazole
- 2-Acetylthiazole-5-carboxylic acid
- 4-Acetyl-thiazole-2-carboxylic acid ethyl ester
- Acotiamide hydrochloride
- Acotiamide hydrochloride trihydrate
- T16Ainh- a01
- N-Allyl-4-(4-methylphenyl)- 1,3-thiazol-2-amine
- Alpha-(2-thiazolylamino)- o-cresol
- 2- Aminobenzo[d ]thiazole-6-carbaldehyde
- 2 Aminobenzo[d]thiazole-5-carbonitrile
- 6-Amino- 2-benzothiazolecarbonitrile
- 2-Aminobenzothiazole- 6-carboxamide
- 2-Aminobenzothiazole- 6-carboxylic acid
- 2-Aminobenzo[d] thiazole-7-carboxylic acid
- 2-Amino- 1,3-benzothiazole-6-carboxylic acid hydrochloride
- 2-Amino-benzothiazole -6-carboxylic acid 2-methoxy-ethyl ester
- 2-Amino benzothiazole- 5-carboxylic acid methyl ester
- 2-Amino-benzothiazole- 6-carboxylic acid methyl ester
- 2-Amino- 1,3-benzothiazole-6-sulfonamide
- 2-Amino- 1,3-benzothiazol-7-ol
- 2- Aminobenzo[d]thiazol-5-ol
- Laboratory Chemicals
- Home Page
- Company Profile
- Contact Us
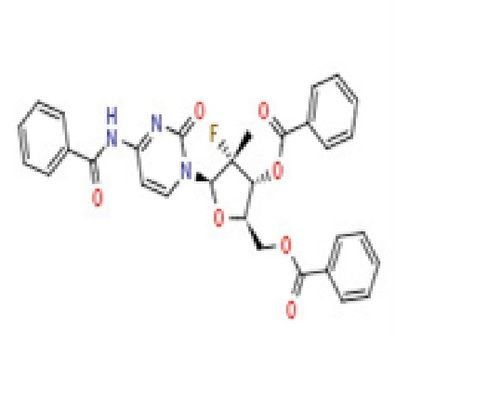
(2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidine 3',5'-dibenzoate
Product Details:
- Appearance White to off-white crystalline powder
- Poisonous NO
- Grade Research/Pharmaceutical Grade
- Properties Highly pure, nucleoside derivative, modified base with fluorine and methyl substitution
- Melting Point 170 - 175C
- Application Used in nucleoside analogue synthesis, pharmaceutical R&D, and as an intermediate in antiviral drug development
- Physical Form Solid
- Click to view more
-
Share Your Product:
(2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidine 3',5'-dibenzoate Price And Quantity
- 100 INR/Number
- 1 GRM Kilograms
(2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidine 3',5'-dibenzoate Product Specifications
- (2R)-N-Benzoyl-2-deoxy-2-fluoro-2-methylcytidine 3,5-dibenzoate
- Pharmaceutical Intermediate
- Not determined
- 622.61 g/mol
- Store at 2-8C, protected from light and moisture
- Crystalline
- Gram per cubic centimeter(g/cm3)
- Solid
- Soluble in DMSO, slightly soluble in methanol and ethanol
- Odorless
- 2 years under recommended storage conditions
- 98% (HPLC)
- 29349900
- Suitable as a synthetic intermediate in laboratory research
- C34H27FN4O7
- 863329-66-2
- See image or refer to C34H27FN4O7
- Research/Pharmaceutical Grade
- NO
- White to off-white crystalline powder
- Highly pure, nucleoside derivative, modified base with fluorine and methyl substitution
- Used in nucleoside analogue synthesis, pharmaceutical R&D, and as an intermediate in antiviral drug development
- 170 - 175C
Product Description
Chemical Name: (2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidine3',5'-dibenzoate
Synonyms : (2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-Methylcytidine3',5'-dibenzoate |
Cytidine,N-benzoyl-2'-deoxy-2'-fluoro-2'-methyl-, 3',5'-dibenzoate,(2'R)-
|
Cytidine, N-benzoyl-2'-deoxy-2'-fluoro-2'-methyl-,3',5'-dibenzoate, (2'R)- |
(2'R)-N-benzoyl-2'-deoxy-2'-fluoro-2'-methyl-3',5'-dibenzoateCytidine |
[(2R,3R,4R,5R)-5-(4-benzamido-2-oxopyrimidin-1-yl)-3-benzoyloxy-4-fluoro-4-methyloxolan-2-yl]methylbenzoate |
Cytidine,N-benzoyl-2'-deoxy-2'-fluoro-2'-methyl-,3',5'-dibenzoate,(2'R)-
CAS No : 817204-32-3
Purity : 97%
Molecular Formula :C31H26FN3O7
Molecular Weight: 571.6
Appearance: Solid
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

Other Products in 'Fluorides' category
Thank You!
Thank You for your valuable time. We have received your details and will get back to you shortly.
- Kesarkunj, Rm No. 9, Vasanji Lalji Road, Kandivali (W), Mumbai - 400067, Maharashtra, India
- Phone : 08071630254
- Send Inquiry
GST : 27AWSPS9814L1ZF
- Mr Nilesh Shah (Director)
- Mobile : 08071630254
- Mr Dharmik Shah
+919987787082
Developed and Managed by Infocom Network Private Limited.


For an immediate response, please call this
number 08071630254

Price:


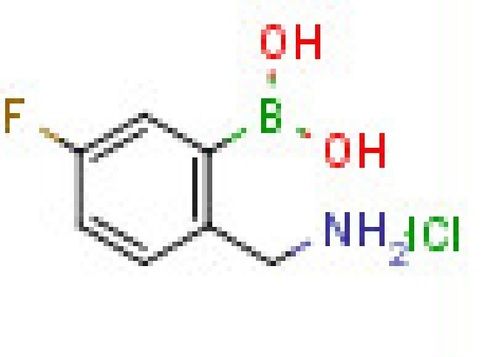
 Call Me Free
Call Me Free